
Filed Pursuant to Rule 424(b)(3)
File No. 333-266287
PROSPECTUS
48,260,954 Subordinate Voting Shares

This prospectus relates to the sale or other disposition from time to time of up to 48,260,954 subordinate voting shares, no par value (Subordinate Voting Shares), consisting of: (i) 7,491,952 Subordinate Voting Shares issuable upon exercise of outstanding options issued under the Company's equity plan, (ii) 661,607 Subordinate Voting Shares issued upon the exercise of options issued under the Company’s equity plan, (iii) 1,538,326 Subordinate Voting Shares issued as restricted stock awards under the Company's equity plan, (iv) 715,846 Subordinate Voting Shares issued upon exercise of warrants issued in connection with services rendered to the Company; (v) 1,535,000 Subordinate Voting Shares issuable upon exercise of outstanding warrants issued in connection with services provided to the Company; (vi) 6,900,000 Subordinate Voting Shares issuable upon exercise of outstanding warrants issued in connection with the Company's 10% Senior Secured Notes; (vii) 1,311,555 Subordinate Voting Shares issued upon exercise of warrants issued in connection with the Company's 10% Senior Secured Notes; (viii) 11,575,000 Subordinate Voting Shares issuable upon exercise of outstanding warrants issued in private placement transactions; (ix) 198,468 Subordinate Voting Shares issued upon the exercise of warrants issued in private placement transactions; and (x) 16,333,200 Subordinate Voting Shares issued in private placement and acquisition transactions held by the selling shareholders named in this prospectus. We are not selling any Subordinate Voting Shares under this prospectus and will not receive any of the proceeds from the sale of Subordinate Voting Shares by the selling shareholders. This is the first public offering of our securities in the United States.
We have four classes of authorized shares: Subordinate Voting Shares, Multiple Voting Shares, Super Voting Shares and Preferred Shares. The terms and conditions of the Subordinate Voting Shares, Multiple Voting Shares and Super Voting Shares are identical except with respect to voting and conversion rights. The terms and conditions of the Preferred Shares will be determined by our board of directors, if, as and when such Preferred Shares are issued. Currently only Subordinate Voting Shares are issued and outstanding. See “Description of Capital Stock.”
The selling shareholders may sell or otherwise dispose of the Subordinate Voting Shares covered by this prospectus in a number of different ways and at varying prices. The prices at which the selling shareholders may sell the Subordinate Voting Shares will be determined by the prevailing market price for the Subordinate Voting Shares or in negotiated transactions. We provide more information about the selling shareholders and how they may sell or otherwise dispose of their Subordinate Voting Shares in the sections entitled “Selling Shareholders” and “Plan of Distribution” on pages 29 and 113, respectively, of this prospectus. The selling shareholders will pay all brokerage fees and commissions and similar expenses. We will pay all expenses (except brokerage fees and commissions and similar expenses) relating to the registration of the Subordinate Voting Shares with the Securities and Exchange Commission.
Our Subordinate Voting Shares are listed on the Canadian Securities Exchange (the CSE) under the symbol “JUSH” and quoted on the OTCQX Best Market under the symbol “JUSHF.” The last reported sale price of our Subordinate Voting Shares on the CSE on August 12, 2022 was C$2.43 per share and on the OTCQX Best Market on August 12, 2022 was $1.89 per share.
Investing in our Subordinate Voting Shares involves risks that are described in the “Risk Factors” section beginning on page 9 of this prospectus.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined if this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
The date of this prospectus is August 12, 2022.
TABLE OF CONTENTS
Unless the context otherwise requires, the terms “Jushi,” “the Company,” “we,” “us” and “our” in this prospectus refer to Jushi Holdings Inc. and its subsidiaries, and “this offering” refers to the offering contemplated by this prospectus.
Neither we nor the selling shareholders authorized anyone to provide any information or to make any representations other than those contained in this prospectus or in any free writing prospectus prepared by or on behalf of us or to which we have referred you. We take no responsibility for, and can provide no assurance as to the reliability of, any other information that others may give you. This prospectus is an offer to sell only the shares offered hereby, but only under the circumstances and in the jurisdictions where it is lawful to do so. The information contained in this prospectus or in any applicable free writing prospectus is current only as of the date of such prospectus, regardless of its time of delivery or any sale of Subordinate Voting Shares. Our business, financial condition, results of operations and prospects may have changed since that date. We are not, and the selling shareholders are not, making an offer of these securities in any jurisdiction where such offer is not permitted.
i
We have not done anything that would permit a public offering of the Subordinate Voting Shares or possession or distribution of this prospectus in any jurisdiction where action for that purpose is required, other than in the United States. Persons outside the United States who come into possession of this prospectus must inform themselves about, and observe any restrictions relating to, the offering of Subordinate Voting Shares and the distribution of this prospectus outside of the United States.
Unless otherwise indicated, information contained in this prospectus concerning our industry and the markets in which we operate, including our general expectations and market position, market opportunity and market share, is based on information from our own management estimates and research, as well as from industry and general publications and research, surveys and studies conducted by third parties. Management estimates are derived from publicly available information, our knowledge of our industry and assumptions based on such information and knowledge, which we believe to be reasonable. Our management estimates have not been verified by any independent source, and we have not independently verified any third-party information. In addition, assumptions and estimates of our and our industry’s future performance are necessarily subject to a high degree of uncertainty and risk due to a variety of factors, including those described in “Risk Factors.” These and other factors could cause our future performance to differ materially from our assumptions and estimates. See “Risk Factors” and “Cautionary Note Regarding Forward-Looking Statements.”
It is important for you to read and consider all of the information contained in this prospectus in making your investment decision. To understand the offering fully and for a more complete description of the offering, you should read this entire document carefully.
ii
|
This summary highlights information contained elsewhere in this prospectus. This summary does not contain all of the information that you should consider before investing in our Subordinate Voting Shares. You should read the following summary together with the more detailed information appearing in this prospectus, including our financial statements and related notes, and the information set forth under the sections titled “Risk Factors,” “Cautionary Note Regarding Forward-Looking Statements” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” before making an investment decision. |
Company Overview
We are a vertically integrated, multi-state cannabis operator engaged in retail, distribution, cultivation, and processing operations in both medical and adult-use markets. Our management team is focused on building a diverse portfolio of cannabis assets through opportunistic investments and pursuing application opportunities in attractive limited license markets. We have targeted assets in highly populated, limited license medical markets that are on a path toward adult-use legalization, including Pennsylvania and Ohio, markets that are in the process of transitioning to adult-use, namely Virginia, and limited license, fast-growing, large adult-use markets, such as Illinois, Nevada and Massachusetts, and certain municipalities of California.
Our Subordinate Voting Shares are listed for trading on the Canadian Securities Exchange (the CSE) under the ticker symbol “JUSH” and quoted in the United States Over the Counter Stock Market (OTCQX) under the ticker symbol “JUSHF.”
Key Markets Overview
Pennsylvania Operations:
We currently operate a total of eighteen medical dispensaries under the BEYOND/HELLO™ brand in Pennsylvania. We also operate an 81,000 sq. ft. cannabis cultivation and processing facility in Scranton, Pennsylvania, through our subsidiary Pennsylvania Medical Solutions, LLC (PAMS). The PAMS facility has recently completed a significant expansion that increased the size of the facility to 123,000 sq. ft. The details relating to the expansion of PAMS can be found in the “Business” section of this prospectus.
Illinois Operations:
We currently operate four adult-use BEYOND/HELLO™ dispensaries in Illinois: two in Sauget (one with co-located medical) and two in Bloomington-Normal (one with co-located medical).
1
In August 2021, Northern Cardinal Ventures, LLC (Northern Cardinal), was awarded a conditional retail dispensary license in Illinois via the state’s lottery process. Jushi is an operational and 49% equity member in Northern Cardinal. Pending regulatory approvals, that dispensary will become the fifth BEYOND/HELLO™ dispensary in Illinois and is designated for the Peoria Bureau of Labor Statistics region in Illinois. The new retail dispensary licensing process for all applicants is currently stayed, pursuant to a court order issued July 2021 in connection with litigation against the state which Jushi is not a party to.
Virginia Operations:
Through our subsidiary Dalitso LLC (Dalitso), we operate a pharmaceutical processor facility in Manassas, Virginia (the Manassas Facility), which allows us to cultivate, process, dispense and deliver medical cannabis to registered patients in Virginia. The Manassas Facility is approximately 93,000 sq. ft., of which approximately 30,000 sq. ft. is built out for operations. In May 2021, we began phase one of the expansion of the existing facility, which added approximately 63,000 sq. ft. of cultivation, manufacturing and processing capacity and was completed in the second quarter of 2022. We also operate two medical BEYOND/HELLO™ dispensaries in Virginia: one in Prince William County near the City of Manassas and one in Sterling, and have plans to open an additional four dispensaries, subject to local zoning and state regulatory approvals.
Massachusetts Operations:
In September 2021, Jushi completed the acquisition of Nature’s Remedy of Massachusetts, Inc. and certain of its affiliates (collectively, Nature’s Remedy), a vertically integrated single state operator in Massachusetts. Nature’s Remedy currently operates two adult-use dispensaries (with one co-located medical), in Millbury, Massachusetts and Tyngsborough, Massachusetts, and a 50,000 sq. ft. cultivation and production facility in Lakeville, Massachusetts with approximately 33,000 sq. ft. of high-quality indoor flower canopy and state-of-the-art extraction and manufacturing capabilities. Our entrance into the Massachusetts market marks the seventh state where we operate cannabis assets and the third state where we are vertically integrated (i.e., operating retail, cultivation, and processing facilities).
California Operations:
We currently operate an adult-use and medical licensed dispensary in each of Santa Barbara, Palm Springs, and Grover Beach, California, and plan to open an additional dispensary in Culver City, California. Our Palm Springs dispensary’s operations, which were voluntarily suspended while the dispensary underwent a significant renovation, resumed in the third quarter of 2022.
Nevada Operations:
Through our subsidiary, Franklin Bioscience NV, LLC (FBS NV), we currently operate cultivation, production and distribution facilities in North Las Vegas, Nevada.
On March 16, 2022, we acquired 100% of the equity interest of an entity operating an adult-use and medical retail dispensary in Las Vegas, Nevada (Apothecarium). We are in the process of changing its name to Beyond Hello™. The Apothecarium acquisition, together with the purchase of FBS NV, enables us to provide significant branding exposure for our high-quality product lines, including The Bank, The Lab, Tasteology and Sèchè.
On April 6, 2022, we closed on our acquisition to acquire NuLeaf, Inc. together with its subsidiaries and related companies (collectively, NuLeaf), a Nevada-based vertically integrated operator. NuLeaf operates two adult-use and medical retail dispensaries in Las Vegas, Nevada, and one adult-use and medical retail dispensary in Lake Tahoe, Nevada, in addition to a 27,000 sq. ft. cultivation facility in Sparks, Nevada, as well as a 13,000 sq. ft. processing facility in Reno, Nevada.
Ohio Operations:
In July 2021, we closed on the acquisition of Ohio Green Grow LLC and OhiGrow, LLC (OhiGrow), one of 34 licensed cultivators in Ohio, inclusive of an approximately 10,000 sq. ft. cultivation facility and 1.35 acres of land.
2
In August 2021, we closed on the acquisition of Franklin Bioscience OH, LLC, (FBS OH), a licensed medical cannabis processor in Ohio. FBS OH operates an 8,000 sq. ft., state-of-the-art processing facility in Columbus, Ohio.
On May 16, 2022, our 100% owned subsidiary, Campbell Hill Ventures, LLC (Campbell Hill Ventures), was awarded a provisional medical marijuana dispensary license by the Ohio Medical Marijuana Control Program and is awaiting certification of licenses by the Ohio Board of Pharmacy. Pending regulatory approvals, this marks our first retail location in Ohio, and establishes our fifth vertically integrated state, accompanying Pennsylvania (by way of its affiliated subsidiaries), Virginia, Massachusetts, and Nevada.
Recent Developments
Grand Reopening of Beyond Hello™ Palm Springs
On July 11, 2022, we announced the grand reopening of our Beyond Hello™ Palm Springs retail location. The redesigned retail location features a new desert-inspired aesthetic, some of the most sought-after locally sourced cannabis and artisan crafts, along with a new art exhibit from award-winning photographer, Jushi Chief Creative Director Andreas (Dre) Neumann.
Management Changes
On July 12, 2022, we appointed our President Louis Jon Barack as Interim Chief Financial Officer, replacing Ed Kremer who resigned as our Chief Financial Officer on the same date. Additionally, we announced on that same date that James Cabral, formerly our Senior Vice President of Finance, was promoted to Chief Accounting Executive. Mr. Kremer’s resignation was not the result of any disagreement on any matter relating to our operations, policies or practices or any financial or accounting matters.
Debuted Line of Concentrate Products Using Hydrocarbon Extraction
On June 29, 2022, we announced the debut of our first line of concentrates made using hydrocarbon extraction by our award-winning brand The Lab™, which is famous for delivering high-quality, precision vape products and concentrates. The Lab™ Live Resin is the second of several single-source concentrate product lines to be launched. Initially, we will exclusively carry The Lab™ Live Resin 500mg full-spectrum 0.5 gram 510 cartridges at BEYOND/HELLO™ retail locations in Pennsylvania. We plan to roll out our hydrocarbon-extracted line at partner dispensaries across Pennsylvania in the coming months, as well as in additional states such as Massachusetts, Virginia and Nevada.
CEO Purchased Subordinate Voting Shares
On June 27, 2022, we announced that Chief Executive Officer (CEO), Chairman, and Founder, James Cacioppo, purchased 100,000 Subordinate Voting Shares of the Company in the open market for an approximate amount of $151,000. Mr. Cacioppo holds in the aggregate approximately 16.9% of the issued and outstanding Subordinate Voting Shares on a non-diluted basis.
Opened 33rd Retail Location Nationwide and Fourth Dispensary in Nevada
On June 8, 2022, we expanded our retail presence with the opening of our 33rd dispensary nationally and fourth dispensary in Nevada with NuLeaf Las Vegas The Strip. Following the opening of NuLeaf Las Vegas The Strip, our retail operations in Nevada consists of three adult-use and medical dispensaries in Las Vegas and one adult-use and medical dispensary in Lake Tahoe.
Awarded Provisional Medical Marijuana Dispensary License in Ohio
On May 16, 2022, our 100% owned subsidiary, Campbell Hill Ventures, was awarded a provisional medical marijuana dispensary license by the Ohio Medical Marijuana Control Program in the Tri-State area of Cincinnati. The new store will operate under Jushi’s retail brand, BEYOND/HELLO™, and is expected to open by early Q1 2023.
Expanded Product Offerings with the Launch of First Line of Solventless Cannabis Extracts
On May 16, 2022, we announced the debut of our first line of solventless live rosin extracts by our award-winning brand, The LabTM. The new top-shelf product line, includes a 0.5g vape extract cartridge available now and 1g jarred concentrates coming soon for purchase exclusively at BEYOND/HELLO™ store locations in Pennsylvania under the name, The Lab™ Live RSN. The Lab™ Live Rosin is expected to launch at our retail locations in Massachusetts, Nevada and Virginia, pending regulatory approvals.
Opened 3rd Adult-Use and Medical Dispensary in California
On May 13, 2022, we expanded our retail footprint in California with the opening of an adult-use and medical retail dispensary in Grover Beach, California, BEYOND/HELLO™ Grover Beach (Grover Beach). This is our 32nd retail location nationally and third adult-use and medical retail dispensary in California.
Completed Acquisition of Vertically Integrated Operator in Nevada
On April 6, 2022, we closed on the previously announced agreement to acquire NuLeaf for $53.6 million. At close, NuLeaf operated two adult-use and medical retail dispensaries in Las Vegas and Lake Tahoe, in addition to a 27,000 sq. ft. cultivation facility in Sparks, Nevada and a 13,000 sq. ft. processing facility in Reno, Nevada. NuLeaf subsequently opened a third licensed retail dispensary, located directly on Las Vegas Boulevard in Las Vegas, on June 8, 2022.
Completed Acquisition of Las Vegas, Nevada Dispensary
On March 17, 2022, we closed on the previously announced agreement to acquire Apothecarium, an operating adult-use and medical retail dispensary in Las Vegas, Nevada.
Private Placement with Strategic Asset Manager and Long-Term Strategic Investors
On January 26, 2022, we completed a non-brokered private placement offering of an aggregate of 2,717,392 Subordinate Voting Shares at a price of $3.68 per share to Graticule Asset Management Asia for gross proceeds of $10.0 million. On January 31, 2022, we completed a non-brokered private placement offering of an aggregate of 1,000,000 Subordinate Voting Shares at a price of $3.68 per share to Kenneth Rosen and group for gross proceeds of $3.7 million.
Acquisition Facility
On October 20, 2021, we entered into definitive documentation in respect of a $100 million Senior Secured Credit Facility (the Acquisition Facility) with Roxbury, LP acting as agent to SunStream Bancorp Inc. (Sunstream), a joint venture sponsored by Sundial Growers Inc. (NASDAQ:SNDL). The Acquisition Facility is subject to certain customary negative covenants until the 10% Senior Secured Notes mature or are refinanced, including covenants that restrict the Company’s and its subsidiaries’ ability to pay dividends or make distributions, incur or guarantee additional indebtedness or conduct sales and other dispositions of certain properties or assets. Upon the maturity or refinancing of the 10% Senior Secured Notes, the Company and its subsidiaries who are parties to the the Acquisition Facility will be subject to additional customary negative covenants that, among other things, restrict the Company’s and its applicable subsidiaries’ ability to create liens, make investments, consummate acquisitions, mergers, reorganizations or similar transactions, enter into sale-leaseback transactions, prepay subordinated debt, pay dividends or make distributions, change the line of business and enter into transactions with affiliates.
As of the date of this prospectus, we have drawn down $65.0 million from under the Acquisition Facility to fund the cash portion of the recently completed acquisitions of Nature’s Remedy, Apothecarium and NuLeaf. We will consider drawing down additional amounts under the Acquisition Facility in the future for potential strategic expansion opportunities in both core and developing markets. Funds drawn under the Acquisition Facility bear an interest rate of 9.5% per annum, payable quarterly, and mature on October 20, 2026. We will be able to borrow amounts under the Acquisition Facility until May 20, 2023 and will have a two-year interest-only period before partial amortization begins on a quarterly basis. We also may increase the total commitment of the Acquisition Facility by an aggregate amount of up to an additional $25 million, subject to certain conditions. The Acquisition Facility is secured by a first lien over certain of our assets and on a pari passu basis with current senior indebtedness on existing assets that are collateralized under our current 10% Senior Secured Notes.
3
Corporate Information
Our principal executive offices are located at 301 Yamato Road, Suite 3250, Boca Raton, Florida 33431 and our telephone number is (561) 617-9100. We maintain a website at http://www.jushico.com. The information contained on, or accessible through, our website is not part of this prospectus. Our periodic and current reports are available, free of charge, after the material is electronically filed with, or furnished to, the Canadian securities regulators on SEDAR, at www.sedar.com. The offering contemplated by this prospectus is the first public offering of our securities in the United States (U.S.). Because we have not previously registered a class of securities under Section 12 of the Exchange Act, we have not historically been required to file reports on Forms 10-K, 10-Q or 8-K. We have filed with the Securities and Exchange Commission (SEC) a registration statement on Form S-1, including exhibits and schedules, under the Securities Act of 1933, as amended (the Securities Act) with respect to the Subordinate Voting Shares to be sold in this offering. This prospectus constitutes a part of the registration statement and, following the effectiveness of the registration statement, we will become subject to the full informational and periodic reporting requirements of the Securities Exchange Act of 1934, as amended (the Exchange Act). For further information about us and our Subordinate Voting Shares, you may refer to the registration statement. You may read, without charge, all or any portion of the registration statement or any reports, statements or other information we file with the SEC on the internet website maintained by the SEC at http://www.sec.gov.
Summary of Risk Factors
Our business is subject to numerous risks and uncertainties, including those highlighted in the section titled “Risk Factors” immediately following this prospectus summary. Some of these risks are:
Risks
Related to our Business and Industry
| • | Our ability to grow our medical and adult-use cannabis product offerings and dispensary services may be limited. |
| • | If we cannot manage our growth, it could have a material adverse effect on our business, financial condition and results of operations. |
| • | We have a history of sustained losses and negative cash flow from operations. |
| • | We expect to incur significant ongoing costs and obligations related to our investment in infrastructure, growth, regulatory compliance and operations. |
| • | The market for the Subordinate Voting Shares may be limited for holders of our securities who live in the U.S. |
| • | The COVID-19 pandemic could adversely affect our business, financial condition and results of operations. |
| • | We face increasing competition that may materially and adversely affect our business, financial condition and results of operations. |
| • | We may not be able to accurately forecast our operating results and plan our operations due to uncertainties in the cannabis industry. |
| • | We are highly dependent on certain key personnel. |
| • | We face inherent risks of liability claims related to the use of our products. |
| • | Our medical marijuana business may be impacted by consumer perception of the cannabis industry, which we cannot control or predict. |
| • | Product recalls could result in a material and adverse impact on our business, financial condition and results of operations. |
| • | We are subject to security risks related to our products as well as our information and technology systems. |
| • | We have a substantial level of indebtedness, and we may not be able to generate sufficient cash to service all of our indebtedness and may be forced to take other actions to satisfy our obligations under our indebtedness, which may not be successful. The terms of our indebtedness may also impair our ability to respond to changing business and economic conditions and may seriously harm our business. | |
| • | The potential impact to the Company of management's determination regarding substantial doubt about its ability to continue to operate as a going concern. | |
| • | We are subject to labor risks and a dispute with our employees or labor unions could have an adverse effect on our results of operations. |
Risks Related to the Regulatory Environment
| • | Cannabis is illegal under U.S. federal law. |
| • | The regulation of cannabis in the U.S. is uncertain. |
4
| • | Anti-Money Laundering Laws in the U.S. may limit access to funds from banks and other financial institutions. |
| • | Potential regulation by the U.S. Food and Drug Administration could have a material adverse effect on our business, financial condition and results of operations. |
| • | As a cannabis company, we may be subject to heightened scrutiny in Canada and the U.S. that could materially adversely impact the liquidity of the Subordinate Voting Shares. |
| • | As a cannabis business, we are subject to certain tax provisions that have a material adverse effect on our business, financial condition and results of operations. |
| • | We are subject to limits on our ability to own the licenses necessary to operate our business, which will adversely affect our ability to grow our business and market share in certain states. |
| • | Our property is subject to risk of civil asset forfeiture. |
| • | We may be at a higher risk of U.S. Internal Revenue Service (the IRS) audit. |
| • | We could be subject to criminal prosecution or civil liabilities under The Racketeer Influenced Corrupt Organizations Act (RICO). |
Risks
Related to our Subordinate Voting Shares
| • | Return on Subordinate Voting Shares is not guaranteed. |
| • | Raising additional capital may cause dilution to our shareholders. |
| • | Sales of substantial amounts of Subordinate Voting Shares by our existing shareholders in the public market may have an adverse effect on the market price of the Subordinate Voting Shares. |
| • | The market price for the Subordinate Voting Shares has been and is likely to continue to be volatile. |
| • | There may not be sufficient liquidity in the markets for our Subordinate Voting Shares. |
| • | We will be subject to increased expenses as a result of being a U.S. reporting company. |
| • | We have identified material weaknesses in our internal control over financial reporting which, if not corrected, could affect the reliability of our consolidated financial statements and have other adverse consequences. |
Implications of Being an Emerging Growth Company
As a company with less than $1.07 billion in revenue during our last fiscal year, we qualify as an “emerging growth company” as defined in the Jumpstart Our Business Startups Act of 2012 (the JOBS Act). As an emerging growth company, we may take advantage of specified reduced reporting and other requirements that are otherwise applicable generally to public companies. These provisions include:
| • | being permitted to provide only two years of audited financial statements, in addition to any required unaudited interim financial statements, with correspondingly reduced “Management’s Discussion and Analysis of Financial Condition and Results of Operations” disclosure; |
| • | an extended transition period for complying with new or revised accounting standards under Section 102(b)(1) of the JOBS Act; |
| • | reduced disclosure about our executive compensation arrangements in our periodic reports, proxy statements and registration statements; |
| • | exemptions from the requirements to hold a non-binding advisory vote on executive compensation or seek shareholder approval of golden parachute arrangements not previously approved; and |
| • | an exemption from the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act of 2002, as amended, (the Sarbanes-Oxley Act) in the assessment of our internal control over financial reporting. |
In addition, the JOBS Act provides that an emerging growth company can take advantage of an extended transition period for complying with new or revised accounting standards. This allows an emerging growth company to delay the adoption of certain accounting standards until those standards would otherwise apply to private companies.
5
We expect to take advantage of some or all of the reduced reporting and other requirements that will be available to us as long as we qualify as an emerging growth company. We will, in general, remain an emerging growth company for up to five full fiscal years following the effectiveness of the registration statement of which this prospectus forms a part. We will cease to be an emerging growth company and become ineligible to rely on the above exemptions, if we:
| • | have $1.07 billion or more in annual revenue in a fiscal year; |
| • | issue more than $1.0 billion of non-convertible debt during any three-year period; or |
| • | become a “large accelerated filer” as defined in Rule 12b-2 promulgated under the Exchange Act, which would occur as of the end of our fiscal year where: (i) we have filed at least one annual report pursuant to the Exchange Act; (ii) we have been a company reporting with the SEC for at least 12 months; and (iii) the market value of shares of our Subordinate Voting Shares that are held by non-affiliates equals or exceeds $700.0 million as of the last business day of our most recently completed second fiscal quarter. |
| The Offering |
| Subordinate Voting Shares offered: | The selling shareholders may offer from time to time up to an aggregate of 48,260,954 Subordinate Voting Shares consisting of: (i) 7,491,952 Subordinate Voting Shares issuable upon exercise of outstanding options issued under the Company's equity plan, (ii) 661,607 Subordinate Voting Shares issued upon the exercise of options issued under the Company’s equity plan, (iii) 1,538,326 Subordinate Voting Shares issued as restricted stock awards under the Company's equity plan, (iv) 715,846 Subordinate Voting Shares issued upon exercise of warrants issued in connection with services rendered to the Company; (v) 1,535,000 Subordinate Voting Shares issuable upon exercise of outstanding warrants issued in connection with services provided to the Company; (vi) 6,900,000 Subordinate Voting Shares issuable upon exercise of outstanding warrants issued in connection with the Company's 10% Senior Secured Notes; (vii) 1,311,555 Subordinate Voting Shares issued upon exercise of warrants issued in connection with the Company's 10% Senior Secured Notes; (viii) 11,575,000 Subordinate Voting Shares issuable upon exercise of outstanding warrants issued in private placement transactions; (ix) 198,468 Subordinate Voting Shares issued upon the exercise of warrants issued in private placement transactions; and (x) 16,333,200 Subordinate Voting Shares issued in private placement and acquisition transactions. |
Subordinate Voting Shares outstanding as of August 4, 2022: |
As of August 4, 2022, 195,849,084 Subordinate Voting Shares were issued and outstanding. As of August 4, 2022, no Subordinate Shares were issuable upon conversion of Super Voting Shares, Multiple Voting Shares or Preferred Shares. |
Subordinated Voting Shares outstanding after giving effect to the exercise of options and warrants for the Subordinated Voting Shares offered by the selling shareholders hereby: |
223,351,036 |
| Use of proceeds: | We will not receive any of the proceeds from the sale of Subordinate Voting Shares by the selling shareholders in this offering. To the extent exercised for cash, we will receive the applicable exercise price for any options or warrants exercised that may be sold in this offering. We will not receive any proceeds from the exercise of any of such options or warrants that are exercised on a net or “cashless” basis in accordance with their terms. |
6
| Risk Factors: | You should read the “Risk Factors” section beginning on page 9 and other information included in this prospectus for a discussion of factors to consider carefully before deciding to invest in our Subordinate Voting Shares. |
| Stock exchange listing: | The Subordinate Voting Shares are listed on the Canadian Securities Exchange under the symbol “JUSH” and trade on the OTCQX Best Market under the symbol “JUSHF.” |
| Description of Capital Stock: | We have four classes of authorized shares: Subordinate Voting Shares, Multiple Voting Shares, Super Voting Shares and Preferred shares. Currently only Subordinate Voting Shares are issued and outstanding. The terms and conditions of the Subordinate Voting Shares, Multiple Voting Shares, and Super Voting Shares are identical except with respect to voting and conversion rights. The terms and conditions of the Preferred Shares will determined by our board of directors, if, as and when such Preferred Shares are issued. See “Description of Capital Stock.” |
7
Summary Consolidated Financial Data
The following tables summarize our consolidated financial and other data. We derived our summary consolidated statements of operations and comprehensive income data for the three months ended March 31, 2022 and 2021, and the years ended December 31, 2021 and 2020 and our summary consolidated balance sheet data as of March 31, 2022, December 31, 2021 and 2020 from our consolidated financial statements included elsewhere in this prospectus. Our historical results are not necessarily indicative of the results that may be expected in the future. You should read the following financial information together with the information under the section titled “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and our consolidated financial statements and related notes included elsewhere in this prospectus.
| For
the Three Months Ended March 31, (unaudited) | For the Year Ended December 31, | |||||||||||||||
| (amounts expressed in thousands of U.S. dollars, unless otherwise stated) | 2022 | 2021 | 2021 | 2020 | ||||||||||||
| Revenue, net | $ | 61,888 | $ | 41,675 | $ | 209,292 | $ | 80,772 | ||||||||
| Cost of goods sold | (42,776 | ) | (22,934 | ) | (125,898 | ) | (42,431 | ) | ||||||||
| Gross profit | $ | 19,112 | $ | 18,741 | $ | 83,394 | $ | 38,341 | ||||||||
| Operating expenses | $ | 37,308 | $ | 21,911 | $ | 119,159 | $ | 54,895 | ||||||||
| Net income (loss) and comprehensive income(loss) attributable to Jushi shareholders | $ | (19,757 | ) | $ | (30,876 | ) | $ | 20,251 | $ | (210,607 | ) | |||||
| Net income (loss) and comprehensive income (loss) per share attributable to Jushi shareholders - basic | $ | (0.11 | ) | $ | (0.20 | ) | $ | 0.12 | $ | (1.94 | ) | |||||
| Weighted average shares outstanding - basic | 183,226,027 | 157,176,375 | 170,292,035 | 108,485,158 | ||||||||||||
| Net income (loss) and comprehensive income (loss) per share attributable to Jushi shareholders - diluted | $ | (0.16 | ) | $ | (0.20 | ) | $ | (0.42 | ) | $ | (1.94 | ) | ||||
| Weighted average shares outstanding - diluted | 207,838,906 | 157,176,375 | 201,610,251 | 108,485,158 | ||||||||||||
| March
31, 2022 (unaudited) | December 31, 2021 | December 31, 2020 | ||||||||||
| Consolidated Balance Sheet Data: | ||||||||||||
| Cash and cash equivalents | $ | 75,717 | $ | 94,962 | $ | 85,857 | ||||||
| Working capital (1) | $ | (19,706 | ) | $ | 70,430 | $ | 89,345 | |||||
| Total assets | $ | 650,644 | $ | 649,141 | $ | 335,665 | ||||||
| Total liabilities | $ | 457,487 | $ | 468,158 | $ | 333,242 | ||||||
| Total shareholders’ equity | $ | 193,157 | $ | 180,983 | $ | 2,423 | ||||||
| (1) | We define working capital as current assets less current liabilities. |
8
Investing in our Subordinate Voting Shares involves a high degree of risk. Before you decide to invest in our Subordinate Voting Shares, you should consider carefully the risks described below, together with the other information contained in this prospectus, including our financial statements and the related notes appearing at the end of this prospectus. We believe the risks described below are the risks that are material to us as of the date of this prospectus. If any of the following risks actually occur, our business, results of operations and financial condition would likely be materially and adversely affected. In these circumstances, the market price of our Subordinate Voting Shares could decline, and you may lose part or all of your investment.
Risks Related to Our Business and Industry
The cannabis industry is relatively new.
We are operating in a relatively new industry and market. In addition to being subject to general business risks, we must continue to build brand awareness in this industry and market share through significant investments in our strategy, production capacity, quality assurance and compliance with regulations. Research in Canada, the U.S. and internationally regarding the medical benefits, viability, safety, efficacy and dosing of cannabis or isolated cannabinoids, such as cannabidiol (CBD), and tetrahydrocannabinol (THC) remains in relatively early stages. Few clinical trials on the benefits of cannabis or isolated cannabinoids have been conducted. Future research and clinical trials may draw opposing conclusions to statements contained in the articles, reports and studies currently favored, or could reach different or negative conclusions regarding the medical benefits, viability, safety, efficacy, dosing or other facts and perceptions related to medical cannabis, which could adversely affect social acceptance of cannabis and the demand for our products and dispensary services.
Accordingly, there is no assurance that the cannabis industry and the market for medicinal and/or adult-use cannabis will continue to exist and grow as currently anticipated or function and evolve in a manner consistent with management’s expectations and assumptions. Any event or circumstance that adversely affects the cannabis industry, such as the imposition of further restrictions on sales and marketing or further restrictions on sales in certain areas and markets could have a material adverse effect on our business, financial condition and results of operations.
Our ability to grow our medical and adult-use cannabis product offerings and dispensary services may be limited.
As we introduce or expand our medical and adult-use cannabis product offerings and dispensary services, we may incur losses or otherwise fail to enter certain markets successfully. Our expansion into new markets may place us in competitive and regulatory environments with which we are unfamiliar and involve various risks, including the need to invest significant resources and the possibility that returns on those investments will not be achieved for several years, if at all. In attempting to establish new product offerings or dispensary services, we may incur significant expenses and face various other challenges, such as expanding our work force and management personnel to cover these markets and complying with complicated cannabis regulations that apply to these markets. In addition, we may not successfully demonstrate the value of these product offerings and dispensary services to consumers, and failure to do so would compromise our ability to successfully expand these additional revenue streams.
We may acquire other companies or technologies.
Our success will depend, in part, on our ability to grow our business in response to the demands of consumers and other constituents within the cannabis industry as well as competitive pressures. In some circumstances, we may determine to do so through the acquisition of complementary businesses rather than through internal development. The identification of suitable acquisition candidates can be difficult, time-consuming, and costly, and we may not be able to successfully complete identified acquisitions. In addition, we may not realize the expected benefits from completed acquisitions. The risks we face in connection with acquisition include:
| • | diversion of management time and focus from operating our business to addressing acquisition integration challenges; |
| • | coordination of research and development and sales and marketing functions; |
| • | retention of employees from the acquired company; |
| • | cultural challenges associated with integrating employees from the acquired company into our organization; | |
| • | integration of the acquired company’s accounting, management information, human resources, and other administrative systems; |
| • | the need to implement or improve controls, procedures, and policies at a business that prior to the acquisition may have lacked effective controls, procedures and policies; |
| • | potential write-offs of intangible assets or other assets acquired in transactions that may have an adverse effect on our operating results in a given period; |
| • | liability for activities of the acquired company before the acquisition, including patent and trademark infringement claims, violations of laws, commercial disputes, tax liabilities, and other known and unknown liabilities; and |
9
| • | litigation or other claims in connection with the acquired company, including claims from terminated employees, consumers, former shareholders, or other third parties. |
Our failure to address these risks or other problems encountered in connection with any future acquisitions or investments could cause us to fail to realize the anticipated benefits of these acquisitions or investments, cause us to incur unanticipated liabilities, and harm our business generally. Future acquisitions could also result in the incurrence of debt, contingent liabilities, amortization expenses, or the impairment of goodwill, any of which could harm our financial condition.
We may issue additional Subordinate Voting Shares in connection with such transactions, which would dilute our other shareholders’ interests in us. The presence of one or more material liabilities of an acquired company that are unknown to us at the time of acquisition could have a material adverse effect on our business, results of operations, prospects and financial condition. A strategic transaction may result in a significant change in the nature of our business, operations and strategy. In addition, we may encounter unforeseen obstacles or costs in implementing a strategic transaction or integrating any acquired business into our operations.
If we cannot manage our growth, it could have a material adverse effect on our business, financial condition and results of operations.
We may be subject to growth-related risks, including capacity constraints and pressure on our internal systems and controls. Our ability to manage growth effectively will require us to continue to implement and improve our operational and financial systems and to expand, train and manage our employee base. Our inability to successfully manage our growth may have a material adverse effect on our business, financial condition, results of operations or prospects.
We have a history of sustained losses and negative cash flow from operations, and we expect to incur significant ongoing costs and obligations related to our investment in infrastructure, growth, regulatory compliance and operations and may not be able to achieve profitability.
We have sustained net losses from operations and negative cash flow from operating activities in the past and may incur such losses and negative operating cash flow in the future. We expect to incur significant ongoing costs and obligations related to our investment in infrastructure and growth and for regulatory compliance, which could have a material adverse impact on our results of operations, financial condition and cash flows. In addition, future changes in regulations, more vigorous enforcement thereof or other unanticipated events could require extensive changes to our operations, increase our compliance costs or give rise to material liabilities, which could have a material adverse effect on our business, results of operations and financial condition. Our efforts to grow our business may be more costly than expected, and we may not be able to increase our revenue enough to offset these higher operating expenses. We may incur significant losses in the future for a number of reasons, including unforeseen expenses, difficulties, complications and delays, and other unknown events. If we are unable to achieve and sustain profitability, the market price of our securities may significantly decrease.
We expect to incur significant ongoing costs and obligations related to our investment in infrastructure, growth, regulatory compliance and operations.
We expect to incur significant ongoing costs and obligations related to our investment in infrastructure and growth and for regulatory compliance, which could have a material adverse impact on our results of operations, financial condition and cash flows. In addition, future changes in regulations, more vigorous enforcement thereof or other unanticipated events could require extensive changes to our operations, increase our compliance costs or give rise to material liabilities, which could have a material adverse effect on our business, results of operations and financial condition. Our efforts to grow our business may be more costly than expected, and we may not be able to increase our revenue enough to offset these higher operating expenses. We may incur significant losses in the future for a number of reasons, including unforeseen expenses, difficulties, complications and delays, and other unknown events. If we are unable to achieve and sustain profitability, the market price of our securities may significantly decrease.
10
The market for the Subordinate Voting Shares may be limited for holders of our securities who live in the U.S.
Given the heightened risk profile associated with cannabis in the U.S., capital markets participants may be unwilling to assist with the settlement of trades for U.S. resident securityholders of companies with operations in the U.S. cannabis industry, which may prohibit or significantly impair the ability of securityholders in the U.S. to trade our securities. In the event residents of the U.S. are unable to settle trades of our securities, this may affect the pricing of such securities in the secondary market, the transparency and availability of trading prices and the liquidity of these securities.
The COVID-19 pandemic could adversely affect our business, financial condition and results of operations.
The COVID-19 pandemic has severely restricted the level of economic activity around the world, including where we operate in the U.S. In response to the COVID-19 pandemic the governments of many countries, states, provinces, municipalities and other geographic regions have taken preventative or protective actions, such as imposing restrictions on travel and business operations, ordering temporary closures of businesses and advising or requiring individuals to limit or forego their time outside of their homes. Numerous businesses have temporarily closed voluntarily or closed permanently. Although some preventative or protective actions have been eased or lifted in varying degrees by different governments of various countries, states and municipalities, COVID-19, including new and highly contagious variants of COVID-19, continues to spread quickly throughout the world. Notwithstanding widespread vaccine availability within the U.S., the emergence of COVID-19 variants and slowing vaccination rates in certain localities have resulted in increased infection rates and has caused, and may continue to cause, several jurisdictions to reinstitute certain COVID-19 restrictions. Additional waves of increased COVID-19 infections as well as COVID-19 related restrictions imposed by various governmental authorities (including, for example, requirements to show proof of vaccination), could negatively impact our supply chain, as well as traffic and sales volume for our products, which in turn could have an adverse effect on our business, financial condition and results of operations.
While many closures or restrictions have eased as the spread of COVID-19 and infection rates decline, if the pandemic persists, including if and when new variants of the virus emerge, closures or other restrictions on the conduct our business operations or of third party manufacturers, suppliers or vendors could disrupt our supply chain. Quarantines, shelter-in-place and similar government orders, or the perception that such orders, shutdowns or other restrictions on the conduct of business operations could occur, could impact personnel or the availability or cost of materials, which could in turn disrupt our supply chain. In addition, as a result of COVID-19, we have in certain cases implemented work-from-home policies for certain employees, and the effects of our work-from-home policies may negatively impact productivity, disrupt access to books and records, increase cybersecurity risks and the risk of inadvertent disclosure of confidential information and disrupt our business. In addition, the continued spread of COVID-19 could result in under-utilization of our assets as a result of disruptions in labor supply, as well as delays and increased costs in construction and expansion projects.
The global impact of the COVID-19 pandemic continues to evolve rapidly, and the extent of its effect on our operational and financial performance will depend on future developments, which are highly uncertain, including the duration, scope and severity of the pandemic, the development and availability of effective treatments and vaccines, further actions taken by governments and other third parties to contain or mitigate its impact, the direct and indirect economic effects of the pandemic and related containment measures, and new information that will emerge concerning the severity and impact of COVID-19 and new variants of the virus, among others. Even after the COVID-19 pandemic subsides, our businesses could also be negatively impacted should the effects of the COVID-19 pandemic lead to changes in consumer behavior, such as reductions in discretionary spending. In addition, a severe or prolonged recession resulting from the COVID-19 pandemic would likely materially affect our business and the value of our Subordinated Voting Shares.
We expect to be subject to taxation in both Canada and the U.S., which could have a material adverse effect on our financial condition and results of operations.
We are a Canadian corporation, and as a result generally would be classified as a non-U.S. corporation under the general rules of U.S. federal income taxation. Section 7874 of U.S. Internal Revenue Code of 1986, as amended (the Code), however, contains rules that can cause a non-U.S. corporation to be taxed as a U.S. corporation for U.S. federal income tax purposes. Under Section 7874 of the Code, a corporation created or organized outside of the U.S. will nevertheless be treated as a U.S. corporation for U.S. federal income tax purposes, which is referred to as an inversion, if each of the following three conditions are met: (i) the non-U.S. corporation acquires, directly or indirectly, or is treated as acquiring under applicable U.S. Treasury regulations, substantially all of the assets held, directly or indirectly, by a U.S. corporation or constituting a U.S. trade or business, (ii) after the acquisition, the former shareholders of the acquired U.S. corporation hold at least 80% (by vote or value) of the shares of the non-U.S. corporation by reason of holding shares of the acquired U.S. corporation or acquired trade or business, and (iii) after the acquisition, the non-U.S. corporation’s expanded affiliated group does not have substantial business activities in the non-U.S. corporation’s country of organization or incorporation when compared to the expanded affiliated group’s total business activities.
11
Pursuant to Section 7874 of the Code, we are classified as a U.S. corporation for U.S. federal income tax purposes and are subject to U.S. federal income tax on our worldwide income. Regardless of any application of Section 7874 of the Code, however, we expect to be treated as a Canadian resident company for purposes of the Canadian Income Tax Act, as amended. As a result, we will be subject to taxation both in Canada and the U.S., which could have a material adverse effect on our financial condition and results of operations.
We are a holding company and our ability to pay dividends or make other distributions to shareholders may be limited.
We are a holding company and essentially all of our assets are the capital stock of our subsidiaries. We currently conduct substantially all of our business through our subsidiaries, which currently generate substantially all of our revenues. Consequently, our cash flows and ability to complete current or desirable future growth opportunities are largely dependent on the earnings of our subsidiaries and the distribution of those earnings to Jushi Holdings Inc. The ability of our subsidiaries to pay dividends and other distributions will depend on those subsidiaries’ operating results and will be subject to applicable laws and regulations that require that solvency and capital standards be maintained by a subsidiary company and contractual restrictions contained in the instruments governing any current or future indebtedness of our subsidiaries. In the event of a bankruptcy, liquidation or reorganization of our subsidiaries, holders of indebtedness and trade creditors of that subsidiary may be entitled to payment of their claims from that subsidiary’s assets before we or our shareholders would be entitled to any payment or residual assets.
We face increasing competition that may materially and adversely affect our business, financial condition and results of operations.
We face competition from companies that may have greater capitalization, access to public equity markets, longer operating histories and more manufacturing, retail and marketing experience than us. As we execute our growth strategy, operators in markets we enter in the future will become direct competitors, and we are likely to continue to face increasing and intense competition from these companies. Increased competition by larger and better financed competitors could materially and adversely affect our business, financial condition and results of operations.
If the number of users of adult-use and medical marijuana in the U.S. increases, the demand for products will increase. Consequently, we expect that competition will become more intense as current and future competitors begin to offer an increasing number of diversified products to respond to such increased demand. To remain competitive, we will require a continued investment in research and development, marketing, sales and client support. We may not have sufficient resources to maintain sufficient levels of investment in research and development, marketing, sales and client support efforts to remain competitive, which could materially and adversely affect our business, financial condition and results of operations.
The cannabis industry is undergoing rapid growth and substantial change, which have resulted in an increase in competitors, consolidation and the formation of strategic relationships. Acquisitions or other consolidating transactions could harm us in a number of ways, including losing customers, revenue and market share, or forcing us to expend greater resources to meet new or additional competitive threats, all of which could harm our operating results. As competitors enter the market and become increasingly sophisticated, competition in our industry may intensify and place downward pressure on prices for our products and services, which could result in impairment of our asset values and negatively impact our profitability.
12
We may not be able to accurately forecast our operating results and plan our operations due to uncertainties in the cannabis industry.
Because U.S. federal and state laws prevent widespread participation in and otherwise hinder market research in the medical and adult-use cannabis industry, the third-party market data available to us is limited and unreliable. Accordingly, we must rely largely on our own market research to forecast sales as detailed forecasts are not generally obtainable from other sources at this early stage of the cannabis industry. Our market research and projections of estimated total retail sales, demographics, demand, and similar consumer research, are based on assumptions from limited and unreliable market data, and generally represent the personal opinions of our management team as of the date of this prospectus. A failure in the demand for our products to materialize as a result of competition, technological change or other factors could have a material adverse effect on our business, results of operations, financial condition or prospects.
We are subject to risks related to growing an agricultural product.
Our business involves the growing of cannabis, an agricultural product. Such business is subject to the risks inherent in the agricultural business, such as losses due to infestation by insects or plant diseases and similar agricultural risks. Although much of our growing is expected to be completed indoors, there can be no assurance that natural elements will not have a material adverse effect on our future production.
We are highly dependent on certain key personnel.
We depend on key managerial personnel, including James Cacioppo, our Chief Executive Officer and Chairman, for our continued success, and our anticipated growth may require additional expertise and the addition of new qualified personnel. Qualified individuals within the cannabis industry are in high demand and we may incur significant costs to attract and retain qualified management personnel, or be unable to attract or retain personnel necessary to operate or expand our business. The loss of the services of existing personnel or our failure to recruit additional key managerial personnel in a timely manner, or at all, could harm our business development programs and our ability to manage day-to-day operations, attract collaboration partners, attract and retain other employees, and generate revenues, and could have a material adverse effect on our business, financial condition and results of operations.
We face inherent risks of liability claims related to the use of our products.
As a distributor of products designed to be ingested by humans, we face an inherent risk of exposure to product liability claims, regulatory action and litigation if our products cause or are alleged to have caused significant loss or injury. We may be subject to various product liability claims, including, among others, that our products caused injury or illness, include inadequate instructions for use or include inadequate warnings concerning possible side effects or interactions with other substances. A product liability claim or regulatory action against us, whether or not successful, could result in materially increased costs, adversely affect our reputation with our clients and consumers generally, and have a material adverse effect on our results of operations and financial condition.
We may become party to litigation from time to time in the ordinary course of business which could adversely affect our business. Should any litigation in which we become involved be determined against us, such a decision could adversely affect our ability to continue operating and the market price for the Subordinate Voting Shares. Even if we achieve a successful result in any litigation in which we are involved, the costs of litigation and redirection of our management’s time and attention could have an adverse effect on our results of operations and financial condition.
Consumer preferences may change, and we may be unsuccessful in acquiring or retaining consumers and keeping pace with changing market developments. This could result in lower than expected demand for our products, which could adversely affect our revenues.
As a result of constantly changing consumer preferences, consumer products often attain financial success for a limited period of time. Even if our products achieve financial success, there can be no assurance that we are able to maintain that success or that those products will enable us to continue to be profitable. Our success will be significantly dependent upon our ability to develop new and improved product lines and adapt to consumer preferences. Even if we are successful in introducing new products or further developing our current products, the failure of those products to gain consumer acceptance or the failure to update our products in ways that our customers expect could cause a decline in our products’ popularity and impair our brand. In addition, we may be required to invest significant amounts of capital in the creation of new product lines, brands, marketing campaigns, packaging and other product features—none of which are guaranteed to be successful. Failure to introduce new features and product lines and to achieve and sustain market acceptance, or our inability to satisfy consumer preferences, could adversely affect our ability to generate sufficient revenue in order to maintain profitability.
13
The cannabis industry is in its early stages of development and it is likely that we, and our competitors, will seek to introduce new products in the future. We may not be successful in developing effective and safe new products, anticipating shifts in social trends and consumer demands, bringing such products to market in time to be effectively commercialized, or obtaining any required regulatory approvals, which, together with any capital expenditures made in the course of such product development and regulatory approval processes, may have a material adverse effect on our business and results of operations.
Our medical marijuana business may be impacted by consumer perception of the cannabis industry, which we cannot control or predict.
We believe that the medical marijuana industry is highly dependent upon consumer perception regarding the safety, efficacy and quality of medical marijuana distributed to those consumers. Consumer perception of our products may be significantly influenced by scientific research or findings, regulatory investigations, litigation, media attention and other publicity regarding the consumption of medical marijuana products. There can be no assurance that future scientific research, findings, regulatory proceedings, litigation, media attention or other research findings or publicity will be favorable to the medical marijuana market or any particular product, or consistent with earlier publicity. Future research reports, findings, regulatory proceedings, litigation, media attention or other publicity that are perceived as less favorable than, or that question, earlier research reports, findings or publicity could have a material adverse effect on the demand for our products and our business, results of operations, financial condition and cash flows.
Product recalls could result in a material and adverse impact on our business, financial condition and results of operations.
Manufacturers and distributors of products are sometimes subject to the recall or return of their products for a variety of reasons, including product defects, such as contamination, unintended harmful side effects or interactions with other substances, packaging safety and inadequate or inaccurate labelling disclosure. If any of our products are recalled due to an alleged product defect or for any other reason, we could be required to incur the unexpected expense of the recall and any legal proceedings that might arise in connection with the recall. We may lose a significant amount of sales and may not be able to replace those sales at an acceptable margin or at all. In addition, a product recall may require significant management attention. Although we have detailed procedures in place for testing our products, there can be no assurance that any quality, potency or contamination problems will be detected in time to avoid unforeseen product recalls, regulatory action or lawsuits. Additionally, if one of our significant brands were subject to recall, the image of that brand and Jushi generally could be harmed. Any recall could lead to decreased demand for our products and could have a material adverse effect on our results of operations and financial condition. Additionally, product recalls may lead to increased scrutiny of our operations by regulatory agencies, requiring further management attention and potential legal fees and other expenses.
If securities or industry analysts do not publish research, or publish inaccurate or unfavorable research, about our business or our market, our share price and trading volume could decline.
The trading market for our Subordinate Voting Shares will depend, in part, on the research and reports that securities or industry analysts publish about us or our business, our market or our competitors. We do not have any control over these analysts. If one or more of the analysts who cover us downgrade our Subordinated Voting Shares or publish inaccurate or unfavorable research about our business or industry, the trading price of our shares would likely decline. In addition, if our operating results fail to meet the forecast of analysts, our share price would likely decline. If one or more of these analysts cease coverage of our company or fail to publish reports on us regularly, demand for our shares could decrease, which might cause our share price and trading volume to decline.
We are subject to security risks related to our products as well as our information and technology systems.
Given the nature of our product and its limited legal availability, we are at significant risk of theft at our facilities. We implement security measures to counteract this threat, but there is no guarantee that these measures will be sufficient. A breach of our security measures at one of our facilities could expose us to additional liability and to potentially costly litigation, increase expenses relating to the resolution and future prevention of these breaches and may deter potential patients from choosing our products.
14
In addition, we collect and store personal information about our patients and we are responsible for protecting that information from privacy breaches. We store certain personally identifiable information and other confidential information of our customers on our systems and applications. Though we maintain robust, proprietary security protocols, we may experience attempts by third parties to obtain unauthorized access to the personally identifiable information and other confidential information of our customers. This information could also be otherwise exposed through human error or malfeasance. The unauthorized access or compromise of this personally identifiable information and other confidential information could have a material adverse impact on our business, financial condition and results of operations.
A privacy breach may occur through procedural or process failure, information technology malfunction, or deliberate unauthorized intrusions. Theft of data for competitive purposes, particularly patient lists and preferences, is an ongoing risk whether perpetrated via employee collusion or negligence or through deliberate cyber-attack. Any such theft or privacy breach would have a material adverse effect on our business, financial condition and results of operations.
Our operations depend and will depend, in part, on how well we protect our networks, equipment, information technology (IT), systems and software against damage from a number of threats, including, natural disasters, intentional damage and destruction, fire, power loss, hacking, computer viruses, vandalism and theft. Our operations also depend and will continue to depend on the timely maintenance, upgrade and replacement of networks, equipment, IT systems and software, as well as preemptive expenses to mitigate the risks of failures. Any of these and other events could result in information system failures, delays and/or increase in capital expenses. The failure of information systems or a component of information systems could, depending on the nature of any such failure, adversely impact our reputation and results of operations.
We have a substantial level of indebtedness, and we may not be able to generate sufficient cash to service all of our indebtedness and may be forced to take other actions to satisfy our obligations under our indebtedness, which may not be successful. The terms of our indebtedness may also impair our ability to respond to changing business and economic conditions and may seriously harm our business.
We had $158.8 million of indebtedness, excluding leases and property, plant, and equipment financing obligations of $6.7 million, as of March 31, 2022. We have incurred significant indebtedness under our 10% Senior Secured Notes, Acquisition Facility (as defined herein) and certain acquisition-related promissory notes to fund working capital and other cash needs and to fund acquisitions. We expect to incur additional indebtedness in the future, particularly if we use remaining borrowing availability under the Acquisition Facility to finance all or a portion of any future acquisitions.
Our debt service cost for the 10% Senior Secured Notes is approximately $1.9 million per calendar quarter and our debt service cost for the Acquisition Facility is approximately $1.6 million per calendar quarter (and, following the first quarter of 2024, will increase in connection with certain amortization payments we are required to make). The 10% Senior Secured Notes and the Acquisition Facility are secured by all material assets and owned equity of the Company and certain of its wholly-owned direct and indirect subsidiaries, subject to certain exclusions including cannabis, cannabis-related, hemp and hemp-related permits and licenses, most real property, accounts receivable, inventory, and assets and equity interests that cannot be collateralized pursuant to law or contractual obligation.
In addition, the terms of our existing debt instruments require, and any debt instruments we enter into in the future may require, that we comply with certain restrictions and covenants. These covenants and restrictions, as well as any significant increase in our indebtedness, could adversely impact us for a number of reasons, including:
| • | resulting in an event of default if we fail to satisfy our obligations under our outstanding debt or fail to comply with the financial or other restrictive covenants contained in the agreements governing our other indebtedness, which event of default could result in all of our debt becoming immediately due and payable and could permit our lenders and noteholders to foreclose on the assets securing any such debt; |
| • | increasing our vulnerability to general economic and industry conditions; |
| • | requiring a substantial portion of our cash flow from operations to be dedicated to the payment of principal and interest on our indebtedness, therefore reducing our ability to use our cash flow to fund our operations, capital expenditures and future business opportunities; |
| • | limiting our ability to obtain additional financing for working capital, capital expenditures, debt service requirements, acquisitions and general corporate or other purposes; and |
| • | limiting our ability to adjust to changing market conditions and placing us at a competitive disadvantage compared to our competitors who have less debt. |
Our ability to make scheduled payments on or to refinance our debt obligations depends on our financial condition and operating performance, which is subject to prevailing economic and competitive conditions and to certain financial, business and other factors beyond our control. We cannot assure that we will generate a level of cash flows from operating activities sufficient to permit us to pay the principal, premium, if any, and interest on our indebtedness.
15
If our cash flows and capital resources are insufficient to fund our debt service obligations or if we are unable to refinance existing indebtedness on favorable terms, we may be forced to reduce or delay capital expenditures, sell assets, seek additional capital or restructure or refinance our indebtedness. These alternative measures may not be successful and thus render us unable to meet our scheduled debt service obligations. In the absence of such operating results and resources, we could face substantial liquidity problems and might be required to dispose of material assets or operations to meet our debt service and other obligations, the terms of our debt instruments may prohibit such dispositions. We may not otherwise be able to consummate those dispositions or be able to obtain the proceeds which we could realize from them and any such proceeds received may not be adequate to meet any debt service obligations then due, which would seriously harm our business and prospects.
To service our debt obligations, fund our operations and execute on our business plan, we require a significant amount of cash to meet our needs, which depends on many factors beyond our control, and management has determined that there is substantial doubt as to our ability to continue as a going concern for a period within the next twelve months based on the uncertainty about these factors.
We require substantial cash to service our debt and other obligations when due, fund our operations and execute on our business plan. Pursuant to accounting standards codification (ASC) 205 Presentation of Financial Statements, we are required to and do evaluate at each annual and interim period whether there are conditions or events, considered in the aggregate, that raise substantial doubt about our ability to continue as a going concern within twelve months after the date that our consolidated financial statements for such period are issued.
As reflected in our unaudited interim condensed consolidated financial statements for the fiscal quarter ended March 31, 2022 contained elsewhere in this prospectus, we have incurred losses from operations for the three months ended March 31, 2022, have an accumulated deficit of $262.2 million as of March 31, 2022, and have cash and cash equivalents of $75.7 million as of March 31, 2022. Additionally, as of March 31, 2022, we had an aggregate principal amount of $74.9 million of 10% Senior Secured Notes that will mature on January 15, 2023, which is within twelve months of the date that the unaudited interim condensed consolidated financial statements were issued for the first quarter ended March 31, 2022. Based on the definitions in the relevant accounting standards, management has determined that this condition raises substantial doubt about our ability to continue as a going concern. This evaluation does not consider the potential mitigating effect of management’s plans that have not been fully implemented. The unaudited interim condensed consolidated financial statements as of and for the three months ended March 31, 2022 have been prepared assuming we will continue as a going concern, and do not include any adjustments that might result from the outcome of this uncertainty.
We are pursuing strategies to obtain the required additional funding primarily to fund the 10% Senior Secured Notes and future operations. These strategies may include, but are not limited to: (i) ongoing efforts with certain lenders to refinance the 10% Senior Secured Notes; (ii) deferral of certain expenditures, including capital projects, and reallocation of funds for debt repayment, if the need arose; (iii) alternative sources of financing, including debt through secured borrowings and equity through a base shelf prospectus, which allows us to offer up to C$500 million in securities in Canada through the end of 2023. However, there can be no assurance that we will be able to refinance the 10% Senior Secured Notes, generate positive results from operations, or obtain additional liquidity when needed or under acceptable terms, if at all. If we are unable to repay or refinance the 10% Senior Secured Notes when due, the holders of the 10% Senior Secured Notes may pursue certain remedies relating to the collateral securing such indebtedness and obligations or pursue other remedies in accordance with the Indenture and the documents relating to such collateral. Additionally, such a default in repayment of the 10% Senior Secured Notes would likely permit the lenders under our Acquisition Facility and certain other debt obligations to declare all indebtedness and obligations thereunder immediately due and payable. Any such default in payment of our debt obligations and pursuit of the remedies of the respective lenders thereunder would have a material adverse effect on us and would likely adversely affect the market price of our Subordinate Voting Shares.
We are subject to labor risks and a dispute with our employees or labor unions could have an adverse effect on our results of operations.
Labor unions are working to organize workforces in the cannabis industry in general. As of June 30, 2022, approximately 119 of our employees are covered by collective bargaining agreements with labor unions, and it is possible that employees in certain other facilities or dispensaries will be organized in the future, which could lead to work stoppages or increased labor costs and adversely affect our business, profitability and our ability to reinvest into the growth of our business. Labor unions may also limit our flexibility in dealing with our workforce. Work stoppages and instability in our union relationships could delay the production and sale of our products, which could strain relationships with customers and cause a loss of revenues which would adversely affect our operations.
Reliance on Third-Party Suppliers, Manufacturers and Contractors; Reliance on Key Inputs
The Company's business is dependent on a number of key inputs from third-parties and their related costs including raw materials and supplies related to its cultivation and manufacturing operations, as well as electricity, water and other local utilities. Due to the uncertain regulatory landscape for regulating cannabis in the U.S., the Company's third-party suppliers, manufacturers and contractors may elect, at any time, to decline or withdraw services necessary for the Company's operations. Any significant interruption or negative change in the availability or economics of the supply chain for key inputs from third-parties could materially impact the business, financial condition and operating results of the Company. Some of these inputs may only be available from a single supplier or a limited group of suppliers in the future. If the Company becomes reliant upon a sole source supplier and that supplier was to go out of business or suspend services, the Company might be unable to find a replacement for such source in a timely manner or at all. Similarly, if any future sole source supplier were to be acquired by a competitor, that competitor may elect not to sell to the Company in the future. Additionally, any supplier could at any time suspend or withdraw services. Any inability to secure required supplies and services or to do so on appropriate terms could have a materially adverse impact on the Company's business, financial condition and operating results.
We rely on key utility services.
Our business is dependent on a number of key inputs and their related costs, including raw materials and supplies related to our growing operations, as well as electricity, water and other local utilities. Our cannabis growing operations consume and will continue to consume considerable energy, which makes us vulnerable to rising energy costs. Accordingly, rising or volatile energy costs may, in the future, adversely impact our business and our ability to operate profitably. Additionally, any significant interruption or negative change in the availability or economics of the supply chain for our key inputs could materially impact our business, financial condition and operating results. If we are unable to secure required supplies and services on satisfactory terms, it could have a materially adverse impact on our business, financial condition and operating results.
Inflation could pose a risk to our business.
A continued upward rate of inflation could influence the profits that we generate from our business. When the rate of inflation rises, the operational costs of running our company also increases, such as labor costs, raw materials and public utilities, thus affecting our ability to provide our serves at competitive prices. An increase in the rate of inflation could force our customers to search for other products, causing us to lose business and revenue.
Risks Related to the Regulatory Environment
Cannabis is illegal under U.S. federal law.
In the U.S., cannabis is largely regulated at the state level. Each state in which we operate (or are currently proposing to operate) authorizes, as applicable, medical and/or adult-use cannabis production and distribution by licensed or registered entities, and numerous other states have legalized adult-use of cannabis in some form. However, under U.S. federal law, the possession, use, cultivation, and transfer of cannabis and any related drug paraphernalia is illegal, and any such acts are criminalized under the Controlled Substances Act (CSA). Cannabis is a Schedule I controlled substance under the CSA, and is thereby deemed to have a high potential for abuse, no accepted medical use in the U.S., and a lack of safety for use under medical supervision. The concepts of “medical cannabis,” “retail cannabis” and “adult-use cannabis” do not exist under U.S. federal law. Although we believe that our business activities are compliant with applicable state and local laws in the U.S., strict compliance with state and local cannabis laws would not provide a defense to any federal proceeding which may be brought against us. Any such proceedings may result in a material adverse effect on us. We derive 100% of our revenues from the cannabis industry. The enforcement of applicable U.S. federal laws poses a significant risk to us.
Violations of any U.S. federal laws and regulations could result in significant fines, penalties, administrative sanctions, or settlements arising from civil proceedings conducted by either the U.S. federal government or private citizens. We may also be subject to criminal charges under the CSA, and if convicted could face a variety of penalties including, but not limited to, disgorgement of profits, cessation of business activities or divestiture. Any of these penalties could have a material adverse effect on our reputation and ability to conduct our business, our holding (directly or indirectly) of medical and adult-use cannabis licenses in the U.S., our financial position, operating results, profitability or liquidity or the market price of our publicly-traded shares. In addition, it is difficult for us to estimate the time or resources that would be needed for the investigation, settlement or trial of any such proceedings or charges, and such time or resources could be substantial.
16
The regulation of cannabis in the U.S. is uncertain.
Our activities are subject to regulation by various state and local governmental authorities. Our business objectives are contingent upon, in part, compliance with regulatory requirements enacted by these governmental authorities and obtaining all regulatory approvals necessary for the sale of our products in the jurisdictions in which we operate. Any delays in obtaining or failure to obtain necessary regulatory approvals would significantly delay our development of markets and products, which could have a material adverse effect on our business, results of operations and financial condition. Furthermore, although we believe that our operations are currently carried out in accordance with all applicable state and local rules and regulations, no assurance can be given that new rules and regulations will not be enacted or that existing rules and regulations will not be applied in a manner that could limit or curtail our ability to distribute or produce marijuana. Amendments to current laws and regulations governing the importation, distribution, transportation and/or production of marijuana, or more stringent implementation thereof could have a substantial adverse impact on us.
Government inquiries and investigations could harm our business or reputation.
As the regulatory framework for cannabis continues to evolve in the U.S., government officials often exercise broad discretion in deciding how to interpret and apply applicable local, state and federal laws or regulations. In the future, we may receive formal and informal inquiries from or become subject to investigations by various governmental regulatory authorities regarding our business and compliance with federal, state and local laws, regulations, or standards. Any determination or allegation that our products, operations or activities, or the activities of our employees, contractors or agents, are not in compliance with existing laws, regulations or standards, could adversely affect our business in a number of ways. Even if such inquiries or investigations do not result in the imposition of fines, interruptions to our business, loss of suppliers or other third-party relationships, terminations of necessary licenses and permits, the existence of those inquiries or investigations alone could create negative publicity that could harm our business or reputation.
We are constrained by law in our ability to market our products in the jurisdictions in which we operate.
State and local jurisdictions enforce extensive and detailed requirements applicable to cannabis products in their jurisdiction. In addition, the Federal Trade Commission (the FTC) regulates advertising of consumer products generally, imposes requirements regarding the use and content of testimonials and endorsements, and also requires that advertising claims be adequately substantiated. As such, our brand and portfolio of products must be specifically tailored, and our marketing activities carefully structured, to comply with the state and local regulations, as well as the FTC’s rules and regulations. These restrictions may preclude us from effectively marketing our products and competing for market share, or impose costs on us that cannot be absorbed through increased selling prices for our products.
Anti-Money Laundering Laws in the U.S. may limit access to funds from banks and other financial institutions.
In February 2014, the Treasury Department Financial Crimes Enforcement Network (FinCEN) issued guidance (which is not law) with respect to financial institutions providing banking services to cannabis businesses, including burdensome due diligence expectations and reporting requirements. While the guidance advised prosecutors not to focus their enforcement efforts on banks and other financial institutions that serve marijuana-related businesses, so long as they meet certain conditions, this guidance does not provide any safe harbors or legal defenses from examination or regulatory or criminal enforcement actions by the U.S. Department of Justice (the DOJ), FinCEN, or other federal regulators. Because of this and the fact that the guidance may be amended or revoked at any time, most banks and other financial institutions have not been willing to provide banking services to cannabis-related businesses. In addition to the foregoing, banks may refuse to process debit card payments and credit card companies generally refuse to process credit card payments for cannabis-related businesses. As a result, we may have limited or no access to banking or other financial services in the U.S., and may have to operate our U.S. business on an all-cash basis. If we are unable or limited in our ability to open or maintain bank accounts, obtain other banking services or accept credit card and debit card payments, it may be difficult for us to operate and conduct our business as planned. Although, we are actively pursuing alternatives that ensure our operations will continue to be compliant with the FinCEN guidance (including requirements related to disclosures about cash management and U.S. federal tax reporting), we may not be able to meet all applicable requirements.
17
We are also subject to a variety of laws and regulations in the U.S. that involve money laundering, financial recordkeeping and proceeds of crime, including the Currency and Foreign Transactions Reporting Act of 1970 (commonly known as the Bank Secrecy Act), as amended by Title III of the Uniting and Strengthening America by Providing Appropriate Tools Required to Intercept and Obstruct Terrorism Act of 2001 and any related or similar rules, regulations or guidelines, issued, administered or enforced by governmental authorities in the U.S.
In the event that any of our operations or related activities in the U.S. were found to be in violation of money laundering legislation or otherwise, those transactions could be viewed as proceeds of crime under one or more of the statutes noted above or any other applicable legislation. This could restrict or otherwise jeopardize our ability to declare or pay dividends or effect other distributions.
The re-classification of cannabis or changes in U.S. controlled substance laws and regulations could have a material adverse effect on our business, financial condition and results of operations.
If cannabis is re-classified as a Schedule II or lower controlled substance under the CSA, the ability to conduct research on the medical benefits of cannabis would most likely be more accessible; however, if cannabis is re-classified as a Schedule II or lower controlled substance, the resulting re-classification would result in the need for approval by the U.S. Food and Drug Administration (the FDA) if medical claims are made about our medical cannabis products. As a result of such a re-classification, the manufacture, importation, exportation, domestic distribution, storage, sale and use of such products could become subject to a significant degree of regulation by the U.S. Drug Enforcement Administration (the DEA). In that case, we may be required to be registered to perform these activities and have the security, control, recordkeeping, reporting and inventory mechanisms required by the DEA to prevent drug loss and diversion. Obtaining the necessary registrations may result in delay of the manufacturing or distribution of our products. The DEA conducts periodic inspections of registered establishments that handle controlled substances. Failure to maintain compliance could have a material adverse effect on our business, financial condition and results of operations. The DEA may seek civil penalties, refuse to renew necessary registrations, or initiate proceedings to restrict, suspend or revoke those registrations. In certain circumstances, violations could lead to criminal proceedings.
Potential regulation by the FDA could have a material adverse effect on our business, financial condition and results of operations.
Should the U.S. federal government legalize cannabis, it is possible that the FDA would seek to regulate it under the Food, Drug and Cosmetics Act of 1938. Additionally, the FDA may issue rules and regulations, including good manufacturing practices related to the growth, cultivation, harvesting and processing of medical cannabis. Clinical trials may be needed to verify efficacy and safety of our medical cannabis products. It is also possible that the FDA would require that facilities where medical-use cannabis is grown register with the agency and comply with certain federally prescribed regulations. In the event that some or all of these regulations are imposed, the impact on the cannabis industry is uncertain and could include the imposition of new costs, requirements, and prohibitions. If we are unable to comply with the regulations or registration as prescribed by the FDA, it may have an adverse effect on our business, operating results, and financial condition.
We could be materially adversely impacted due to restrictions under U.S. border entry laws.
Because cannabis remains illegal under U.S. federal law, those investing in Canadian companies with operations in the U.S. cannabis industry could face detention, denial of entry or lifetime bans from the U.S. as a result of their business associations with U.S. cannabis businesses. Entry into the U.S. happens at the sole discretion of U.S. Customs and Border Patrol (CBP) officers on duty, and these officers have wide latitude to ask questions to determine the admissibility of a non-U.S. citizen or foreign national. The government of Canada has warned travelers on its website that previous use of cannabis, or any substance prohibited by U.S. federal law, could mean denial of entry to the U.S. Business or financial involvement in the cannabis industry in the U.S. could also be reason enough for denial of entry into the U.S. On September 21, 2018, the CBP released a statement outlining its current position with respect to enforcement of the laws of the U.S. It stated that Canada’s legalization of cannabis will not change CBP enforcement of U.S. laws regarding controlled substances. According to the statement, because cannabis continues to be a controlled substance under U.S. law, working in or facilitating the proliferation of the marijuana industry in U.S. states where it is legal under state law may affect admissibility to the U.S. On October 9, 2018, the CBP released an additional statement regarding the admissibility of Canadian citizens working in the legal cannabis industry in Canada. CBP stated that a Canadian citizen working in or facilitating the proliferation of the legal cannabis industry in Canada who seeks to come into the U.S. for reasons unrelated to the cannabis industry will generally be admissible to the U.S.; however, if such person is found to be coming into the U.S. for reasons related to the cannabis industry, such person may be deemed inadmissible. As a result, the CBP has affirmed that employees, directors, officers and managers of and investors in companies involved in business activities related to cannabis in the U.S. (such as Jushi), who are not U.S. citizens face the risk of being barred from entry into the U.S. for life.
18
As a cannabis company, we may be subject to heightened scrutiny in Canada and the U.S. that could materially adversely impact the liquidity of the Subordinate Voting Shares.
Our existing operations in the U.S., and any future operations, may become the subject of heightened scrutiny by regulators, stock exchanges and other authorities in the U.S. and Canada.
Given the heightened risk profile associated with cannabis in the U.S., The Canadian Depository of Securities (CDS) may implement procedures or protocols that would prohibit or significantly impair the ability of CDS to settle trades for companies that have cannabis businesses or assets in the U.S.
On February 8, 2018, following discussions with the Canadian Securities Administrators and recognized Canadian securities exchanges, the TMX Group, the parent company of CDS, announced the signing of a Memorandum of Understanding, which we refer to as the TMX MOU, with Aequitas NEO Exchange Inc., the CSE, the Toronto Stock Exchange, and the TSX Venture Exchange. The TMX MOU outlines the parties’ understanding of Canada’s regulatory framework applicable to the rules, procedures, and regulatory oversight of the exchanges and CDS as it relates to issuers with cannabis-related activities in the U.S. The TMX MOU confirms, with respect to the clearing of listed securities, that CDS relies on the exchanges to review the conduct of listed issuers. As a result, there is no CDS ban on the clearing of securities of issuers with cannabis-related activities in the U.S. However, there can be no assurances given that this approach to regulation will continue in the future. If such a ban were to be implemented, it would have a material adverse effect on the ability of holders of the Subordinate Voting Shares to settle trades. In particular, the Subordinate Voting Shares would become highly illiquid until an alternative was implemented and investors would have no ability to effect a trade of the Subordinate Voting Shares through the facilities of a stock exchange.
We may not be able to locate and obtain the rights to operate at preferred locations.
In Massachusetts and other states, the local municipality has authority to choose where any cannabis establishment will be located. These authorized areas are frequently removed from other retail operations. Because the cannabis industry remains illegal under U.S. federal law, the disadvantaged tax status of businesses deriving their income from cannabis, and the reluctance of the banking industry to support cannabis businesses, it may be difficult for us to locate and obtain the rights to operate at various preferred locations. Property owners may violate their mortgages by leasing to us, and those property owners that are willing to allow use of their facilities may require payment of above fair market value rents to reflect the scarcity of such locations and the risks and costs of providing such facilities.
As a cannabis business, we are subject to certain tax provisions that have a material adverse effect on our business, financial condition and results of operations.
Under Section 280E of the Code “no deduction or credit shall be allowed for any amount paid or incurred during the taxable year in carrying on any trade or business if such trade or business (or the activities which comprise such trade or business) consists of trafficking in controlled substances (within the meaning of schedule I and II of the Controlled Substances Act) which is prohibited by Federal law or the law of any State in which such trade or business is conducted.” This provision has been applied by the IRS to cannabis operations, prohibiting companies engaged in such operations from deducting expenses directly associated with the sale of cannabis. Section 280E of the Code may have a lesser impact on cannabis cultivation and manufacturing operations than on sales operations. Section 280E of the Code and related IRS enforcement activity has had a significant impact on the operations of cannabis companies. As a result of Section 280E of the Code, an otherwise profitable business may, in fact, operate at a loss, after taking into account its U.S. income tax expenses.
We may not have access to U.S. bankruptcy protections available to non-cannabis businesses.
Because cannabis is a Schedule I controlled substance under the CSA, many courts have denied cannabis businesses federal bankruptcy protections, making it difficult for lenders to be made whole on their investments in the cannabis industry in the event of a bankruptcy. If we were to experience a bankruptcy, there is no guarantee that U.S. federal bankruptcy protections would be available to us, which would have a material adverse effect on us and may make it more difficult for us to obtain debt financing.
19
There is doubt regarding our ability to enforce contracts.
It is a fundamental principle of law that a contract will not be enforced if it involves a violation of law or public policy. Because cannabis remains illegal at a federal level in the U.S., judges in multiple states have on a number of occasions refused to enforce contracts for the repayment of money when the loan was used in connection with activities that violate U.S. federal law, even if there is no violation of state law. There remains doubt and uncertainty that we will be able to legally enforce our contracts. If we are unable to realize the benefits of or otherwise enforce the contracts into which we enter, it could have a material adverse effect on our business, financial condition and results of operations.
We are subject to limits on our ability to own the licenses necessary to operate our business, which will adversely affect our ability to grow our business and market share in certain states.
In certain states, the cannabis laws and regulations limit not only the number of cannabis licenses issued, but also the number of cannabis licenses that one person or entity may own in that state. For example, in Massachusetts, no person or entity may have an ownership interest in, or control over, more than three medical licenses or three adult-use licenses in any category, which include cultivation, product manufacturing, transport or retail. Such limitations on the acquisition of ownership of additional licenses within certain states may limit our ability to grow organically or to increase our market share in affected states.
We may not be able to adequately protect our intellectual property.
As long as cannabis remains illegal under U.S. federal law as a Schedule I controlled substance under the CSA, the benefit of certain federal laws and protections that may be available to most businesses, such as federal trademark and patent protection, may not be available to us. As a result, our intellectual property may never be adequately or sufficiently protected against the use or misappropriation by third parties. In addition, since the regulatory framework of the cannabis industry is in a constant state of flux, we can provide no assurance that we will ever obtain any protection for our intellectual property, whether on a federal, state or local level.
Our property is subject to risk of civil asset forfeiture.
Because the cannabis industry remains illegal under U.S. federal law, any property owned by participants in the cannabis industry that is either used in the course of conducting or comprises the proceeds of a cannabis business could be subject to seizure by law enforcement and subsequent civil asset forfeiture. Even if the owner of the property were never charged with a crime, the property in question could still be seized and subject to an administrative proceeding by which, with minimal process, it could become subject to forfeiture.
We may be at a higher risk of IRS audit.
We believe there is a greater likelihood that the Internal Revenue Service will audit the tax returns of cannabis-related businesses. Any such audit of our tax returns could result in our being required to pay additional tax, interest and penalties, as well as incremental accounting and legal expenses, which could be material.
We may be unable to obtain adequate insurance coverage.
We have obtained insurance coverage with respect to workers’ compensation, general liability, directors’ and officers’ liability, fire and other similar policies customarily obtained for businesses to the extent commercially appropriate; however, because we are engaged in and operate within the cannabis industry, there are exclusions and additional difficulties and complexities associated with our insurance coverage that could cause us to suffer uninsured losses, which could adversely affect our business, results of operations, and profitability. There is no assurance that we will be able to obtain insurance coverage at a reasonable cost or fully utilize such insurance coverage, if necessary.
20
We could be subject to criminal prosecution or civil liabilities under RICO.
RICO criminalizes the use of any profits from certain defined “racketeering” activities in interstate commerce. While intended to provide an additional cause of action against organized crime, due to the fact that cannabis is illegal under U.S. federal law, the production and sale of cannabis qualifies cannabis related businesses as “racketeering” as defined by RICO. As such, all officers, managers and owners in a cannabis related business could be subject to criminal prosecution under RICO, which carries substantial criminal penalties.
RICO can create civil liability as well: persons harmed in their business or property by actions which would constitute racketeering under RICO often have a civil cause of action against such “racketeers,” and can claim triple their amount of estimated damages in attendant court proceedings. Jushi or its subsidiaries, as well as its officers, managers and owners could all be subject to civil claims under RICO.
Risks Related to Owning Jushi’s Subordinate Voting Shares
Return on Subordinate Voting Shares is not guaranteed.
There is no guarantee that the Subordinate Voting Shares will earn any positive return in the short-term or long-term. A holding of Subordinate Voting Shares is speculative and involves a high degree of risk and should be undertaken only by holders whose financial resources are sufficient to enable them to assume such risks and who have no need for immediate liquidity in their investment. A holding of Subordinate Voting Shares is appropriate only for holders who have the capacity to absorb a loss of some or all of their holdings.
Raising additional capital may cause dilution to our shareholders.
Until such time, if ever, as we can generate substantial revenue, we may finance our cash needs through a combination of equity offerings, debt financings, marketing and distribution arrangements and other collaborations, strategic alliances and licensing arrangements or other sources. We do not currently have any committed external source of funds. In addition, we may seek additional capital due to favorable market conditions or strategic considerations, even if we believe that we have sufficient funds for our current or future operating plans.
To the extent that we raise additional capital through the sale of equity or convertible debt securities, your ownership interest will be diluted, and the terms of these securities may include liquidation or other preferences that adversely affect your rights as a common stockholder. Debt financing and preferred equity financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends. If we are unable to raise additional funds through equity or debt financings when needed, we may be required to delay, limit, reduce or terminate product candidate development or future commercialization efforts
Sales of substantial amounts of Subordinate Voting Shares by our existing shareholders in the public market may have an adverse effect on the market price of the Subordinate Voting Shares.
Sales of a substantial number of Subordinate Voting Shares in the public market could occur at any time. These sales, or the perception in the market that holders of a large number of shares intend to sell shares, or the availability of such securities for sale, could adversely affect the prevailing market prices for the Subordinate Voting Shares. As of August 4, 2022, we have an aggregate of 195,849,084 Subordinate Voting Shares issued and outstanding (excluding securities convertible into or exercisable for Subordinate Voting Shares). A decline in the market prices of the Subordinate Voting Shares could impair our ability to raise additional capital through the sale of securities should we desire to do so.
21
The market price for the Subordinate Voting Shares has been and is likely to continue to be volatile.
The market price for the Subordinate Voting Shares may be volatile and subject to wide fluctuations in response to numerous factors, many of which will be beyond our control, including, but not limited to, the following: (i) actual or anticipated fluctuations in our quarterly results of operations; (ii) recommendations by securities research analysts; (iii) changes in the economic performance or market valuations of companies in the cannabis industry; (iv) additions or departures of our executive officers and other key personnel; (v) release or expiration of transfer restrictions on our issued and outstanding shares; (vi) regulatory changes affecting the cannabis industry generally and our business and operations; (vii) announcements by us and our competitors of developments and other material events; (viii) fluctuations in the costs of vital production materials and services; (ix) changes in global financial markets and global economies and general market conditions, such as interest rates and pharmaceutical product price volatility; (x) significant acquisitions or business combinations, strategic partnerships, joint ventures or capital commitments by or involving us or our competitors; (xi) operating and share price performance of other companies that investors deem comparable to us or from a lack of market comparable companies; (xii) false or negative reports issued by individuals or companies who have taken aggressive short sale positions; and (xiii) news reports relating to trends, concerns, technological or competitive developments, regulatory changes and other related issues in our industry or target markets.
Financial markets have experienced significant price and volume fluctuations that have affected the market prices of equity securities of companies and that have often been unrelated to the operating performance, underlying asset values or prospects of those companies. Accordingly, the market price of the Subordinate Voting Shares may decline even if our operating results, underlying asset values or prospects have not changed.
These factors, as well as other related factors, may cause decreases in asset values that are deemed to be other than temporary, which may result in impairment losses. There can be no assurance that continuing fluctuations in price and volume will not occur. If such increased levels of volatility and market turmoil continue, our operations could be adversely impacted, and the trading price of the Subordinate Voting Shares could be materially adversely affected.
There may not be sufficient liquidity in the markets for our Subordinate Voting Shares.
Our Subordinate Voting Shares are listed for trading on the CSE under the trading symbol “JUSH” and quoted on the OTCQX Best Market under the symbol “JUSHF.” The liquidity of any market for the shares of our Subordinate Voting Shares will depend on a number of factors, including:
| • | the number of shareholders; |
| • | our operating performance and financial condition; |
| • | the market for similar securities; |
| • | the extent of coverage by securities or industry analysts; and |
| • | the interest of securities dealers in making a market in the shares. |
There can be no assurance that an active trading market for the Subordinate Voting Shares, will be sustained.
We will be subject to increased costs as a result of being a U.S. reporting company.
As a public issuer, we are subject to the reporting requirements and rules and regulations under the applicable Canadian securities laws and rules of any stock exchange on which our securities may be listed from time to time. In addition, following the effectiveness of the registration statement of which this prospectus forms a part, we will become subject to the reporting requirements of the Exchange Act, and the regulations promulgated thereunder. Additional or new regulatory requirements may be adopted in the future. The requirements of existing and potential future rules and regulations will increase our legal, accounting and financial compliance costs, make some activities more difficult, time-consuming or costly and may also place undue strain on our personnel, systems and resources, which could adversely affect our business, financial condition, and results of operations.
The provisions of our articles of incorporation requiring exclusive forum in the courts of the province of British Columbia and appellate courts therefrom for certain disputes may have the effect of discouraging lawsuits against us or our directors and officers.
Pursuant to section 28 of our articles of incorporation (the Articles), unless we approve or consent in writing to the selection of an alternative forum, the courts of the province of British Columbia and appellate courts therefrom shall be the sole and exclusive forum for: (a) any derivative action or proceeding brought on behalf of our Company, (b) any action asserting a claim of breach of a fiduciary duty owed by any director or officer of our Company to our Company, (c) any action asserting a claim arising pursuant to any provision of the Business Corporations Act (British Columbia) or the Notice of Articles or Articles of our Company (as either may be amended from time to time); or (d) any action asserting a claim otherwise related to the relationships among our Company, its affiliates and their respective shareholders, directors and/or officers, but this does not include claims related to the business carried on by our Company or such affiliates; provided however it is uncertain whether such provision would apply to actions arising under U.S. federal securities laws, and if it does, whether a British Columbia Court would enforce such provision since in accordance with Section 27 of the Exchange Act, United States federal courts shall have jurisdiction over all suits and any action brought to enforce any duty or liability created by the Exchange Act or the rules and regulations thereunder and that in accordance with Section 22 of the Securities Act, United States federal and state courts shall have concurrent jurisdiction over all suits brought to enforce any duty or liability created by the Securities Act or the rules and regulations thereunder.
The choice of forum provision may limit the ability of our shareholders to bring a claim in a forum that they find favorable for disputes with us or our directors, officers or other employees, and may discourage such lawsuits. If a British Columbia court ruled the choice of forum provision was inapplicable or unenforceable in an action, we may incur additional costs to resolve such action in other jurisdictions. Our shareholders will not be deemed, by operation of the choice of forum provision, to have waived our obligation to comply with all applicable United States federal securities laws and the rules and regulations thereunder.
22
We are an “emerging growth company” and will be able take advantage of reduced disclosure requirements applicable to emerging growth companies, which could make our Subordinate Voting Shares less attractive to investors.
We are an “emerging growth company,” as defined in the Jumpstart Our Business Startups Act of 2012 (the JOBS Act) and, for as long as we continue to be an emerging growth company, we intend to take advantage of certain exemptions from various reporting requirements applicable to other public companies but not to emerging growth companies, including, but not limited to, not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act, reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements, and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and shareholder approval of any golden parachute payments not previously approved. We could be an emerging growth company for up to five years, or until the earliest of (i) the last day of the first fiscal year in which our annual gross revenues exceed $1.07 billion, (ii) the date that we become a “large accelerated filer” as defined in Rule 12b-2 under the Exchange Act, which would occur if the market value of our common stock that is held by non-affiliates exceeds $700 million as of the last business day of our most recently completed second fiscal quarter, or (iii) the date on which we have issued more than $1 billion in non-convertible debt during the preceding three year period.
We intend to take advantage of these reporting exemptions described above until we are no longer an emerging growth company. Under the JOBS Act, emerging growth companies can also delay adopting new or revised accounting standards until such time as those standards apply to private companies.
We cannot predict if investors will find our Subordinate Voting Shares less attractive if we choose to rely on these exemptions. If some investors find our Subordinate Voting Shares less attractive as a result of any choices to reduce future disclosure, there may be a less active trading market for our Subordinate Voting Shares and the price of our Subordinate Voting Shares may be more volatile.
Our internal controls over financial reporting may not be effective, and our independent registered public accounting firm may not be able to certify as to their effectiveness, which could have a significant and adverse effect on our business and reputation.
We are not currently required to maintain an effective system of internal controls as defined by Section 404 of the Sarbanes-Oxley Act. As a U.S. public company, we will be required to evaluate our internal controls over financial reporting following the applicable phase-in period. When we cease to be an “emerging growth company” as defined by the JOBS Act, we will also be required to comply with the auditor attestation requirements of Section 404. As of the date of this prospectus, we have not completed an assessment, nor has our independent registered public accounting firm tested our systems of internal controls. We cannot be certain as to the timing of completion of our evaluation, testing and any remediation actions or the impact of the same on our operations. If we are not able to implement the requirements of Section 404 in a timely manner or with adequate compliance, our independent registered public accounting firm may issue an adverse opinion due to ineffective internal controls over financial reporting and we may be subject to sanctions or investigation by regulatory authorities, such as the SEC. In addition, we may be required to incur costs in improving our internal control system and the hiring of additional personnel. Any such action could negatively affect our results of operations and cash flows.
We have identified material weaknesses in our internal control over financial reporting which, if not corrected, could affect the reliability of our consolidated financial statements and have other adverse consequences.
In connection with the audit of our financial statements as of and for the years ended December 31, 2021 and 2020 and in the process of preparing our financial statements as of and for the three months ended March 31, 2022, the following material weaknesses in our internal control over financial reporting were identified: (i) insufficient accounting resources, inadequate level of precision in the performance of review controls, or effective communication, as it relates to: financial reporting, accounting and valuation for complex financial instruments, inventory, property plant and equipment, accruals, accounting for impairment and business combinations; and (ii) insufficient information technology general controls, as it relates to user access controls, change management, passwords, access controls reviews, backup and cybersecurity vulnerability.
A material weakness is a deficiency or combination of deficiencies in internal control over financial reporting such that there is a reasonable possibility that a material misstatement of the financial statements would not be prevented or detected on a timely basis.
23
Our management is in the process of developing a remediation plan. The material weaknesses will be considered remediated when our management designs and implements effective controls that operate for a sufficient period of time and management has concluded, through testing, that these controls are effective. Our management will monitor the effectiveness of its remediation plans and will make changes management determines to be appropriate.
If not remediated, these material weaknesses could result in material misstatements to our annual or interim consolidated financial statements that might not be prevented or detected on a timely basis, or in delayed filing of required periodic reports, which may adversely affect investor confidence in us and, as a result, our share price.
24
We will not receive any of the proceeds from the sale of the Subordinate Voting Shares by the selling shareholders. To the extent exercised for cash, we will receive the applicable exercise price for any options or warrants exercised for shares that may be sold in this offering. We will not receive any proceeds from the exercise of any of such options or warrants that are exercised on a net or “cashless” basis in accordance with their terms.
We have not declared dividends or distributions on Subordinate Voting Shares in the past. In addition, the Note Indenture governing the 10% Senior Secured Notes and the Acquisition Facility, as defined and described in more detail under the heading “Management’s Discussion and Analysis of Financial Condition and Results of Operations—Liquidity and Capital Resources—Description of Indebtedness,” contains covenants that, among other things, limit our ability to declare or pay dividends or make certain other payments. We currently intend to reinvest all future earnings to finance the development and growth of our business. As a result, we do not intend to pay dividends on Subordinate Voting Shares in the foreseeable future. Any future determination to pay dividends will be at the discretion of our board of directors and will depend on the financial condition, business environment, operating results, capital requirements, any contractual restrictions on the payment of dividends (including the Note Indenture) and any other factors that the board of directors deems relevant. Other than the Note Indenture or the Acquisition Facility, we are not bound or limited in any way to pay dividends in the event that the board of directors determined that a dividend was in the best interest of our shareholders.
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This prospectus contains forward-looking statements. In some cases, you can identify these statements by forward-looking words such as “may”, “will”, “would”, “could”, “should”, “believes”, “estimates”, “projects”, “potential”, “expects”, “plans”, “intends”, “anticipates”, “targeted”, “continues”, “forecasts”, “designed”, “goal”, or the negative of those words or other similar or comparable words. Any statements contained in this prospectus that are not statements of historical facts may be deemed to be forward-looking statements. We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our business, financial condition, results of operations and future growth prospects.
Forward-looking statements may relate to future financial conditions, results of operations, plans, objectives, performance or business developments. These statements speak only as of the date they are made and are based on information currently available and on the then-current expectations of the party making the statement and assumptions concerning future events, which are subject to a number of known and unknown risks, uncertainties and other factors that may cause actual results, performance or achievements to be materially different from that which was expressed or implied by such forward-looking statements, including, but not limited to, risks and uncertainties related to: the performance of our business and operations; the receipt and/or maintenance by us of required licenses and permits in a timely manner or at all; the intention to grow our business and operations; the expected growth in the number of the people using medical and/or adult-use cannabis products; expectations of market size and growth in the U.S.; the competitive conditions and increasing competition of the cannabis industry; applicable laws, regulations and any amendments thereof; our competitive and business strategies; our operations in the U.S., the characterization and consequences of those operations under federal U.S. law, and the framework for the enforcement of medical and adult-use cannabis and cannabis-related offenses in the U.S.; the completion of additional cultivation and production facilities; the general economic, financial market, regulatory and political conditions in which we operate; the U.S. regulatory landscape and enforcement related to cannabis, including political risks; anti-money laundering laws and regulation; other governmental and environmental regulation; public opinion and perception of the cannabis industry; U.S. border entry; heightened scrutiny of cannabis companies in Canada and the U.S.; the enforceability of contracts; reliance on the expertise and judgment of our senior management; proprietary intellectual property and potential infringement by third parties; the concentration of voting control in certain shareholders and the unpredictability caused by our capital structure; the management of growth; risks inherent in an agricultural business; risks relating to energy costs; risks associated to cannabis products manufactured for human consumption, including potential product recalls; reliance on key inputs, suppliers and skilled labor; cybersecurity risks; ability and constraints on marketing products; fraudulent activity by employees, contractors and consultants; tax and insurance related risks; risk of litigation; conflicts of interest; risks relating to certain remedies being limited and the difficulty of enforcement of judgments and effecting service outside of Canada; security risks; risks related to future acquisitions or dispositions; sales by existing shareholders; limited research and data relating to cannabis; the medical benefits, viability, safety, efficacy and social acceptance of cannabis; the availability of financing opportunities, the ability to make payments on existing indebtedness; the potential impact to the Company of management’s determination regarding substantial doubt about its ability to continue to operate as a going concern; risks associated with economic, political and social conditions; risks related to contagious disease, particularly COVID-19; dependence on management; and other risks described in this prospectus and described from time to time in documents filed by us with the SEC.
25
The forward-looking statements contained herein are based on certain key expectations and assumptions, including, but not limited to, with respect to expectations and assumptions concerning: (i) receipt and/or maintenance of required licenses and third party consents; and (ii) the success of our operations. These assumptions are based on estimates prepared by us using data from publicly available governmental sources, as well as from market research and industry analysis, and on data and knowledge of this industry that we believe to be reasonable. However, although generally indicative of relative market positions, market shares and performance characteristics, such data is inherently imprecise. While we are not aware of any misstatement regarding any industry or government data presented herein, the current marijuana industry involves risks and uncertainties and is subject to change based on various factors. Although we believe that the expectations and assumptions on which such forward-looking statements are based are reasonable, undue reliance should not be placed on the forward-looking statements contained herein because no assurance can be given that they will prove to be correct. Since forward-looking statements address future events and conditions, by their very nature they involve inherent risks and uncertainties. Actual results could differ materially from those currently anticipated due to a number of factors and risks. These include, but are not limited to, the risks described above and other factors beyond our control, as more particularly described under the headings “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and elsewhere in this prospectus. Consequently, all forward-looking statements made in this prospectus are qualified by such cautionary statements and there can be no assurance that the anticipated results or developments will actually be realized or, even if realized, that they will have the expected consequences to or effects on us. The cautionary statements contained or referred to in this prospectus should be considered in connection with any subsequent written or oral forward-looking statements that we and/or persons acting on our behalf may issue. We do not undertake any obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, other than as required by law.
You should read this prospectus and the documents that we have filed as exhibits to the registration statement of which this prospectus is a part completely and with the understanding that our actual future results may be materially different from what we expect. We qualify all of our forward-looking statements by these cautionary statements. We do not assume any obligation to update any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law.
26
We are not selling any Subordinate Voting Shares in this offering; however, if you purchase Subordinate Voting Shares in this offering, your ownership interest will be diluted to the extent of the difference between the public offering price paid per Subordinate Voting Share sold in this offering and our net tangible book value per Subordinate Voting Share after this offering. Dilution results from the fact that the public offering price per Subordinate Voting Share is substantially in excess of the net tangible book value per share attributable to the existing shareholders for our presently outstanding shares. Our net tangible book value per share represents the amount of our total tangible assets (total assets less intangible assets) less total liabilities, divided by the number of shares issued and outstanding.
As at March 31, 2022, we had a historical negative net tangible book value of $58.2 million, or $(0.31) per share, based on 189,728,625 shares outstanding as at such date. Dilution is calculated by subtracting net tangible book value per share from the public offering price paid per Subordinate Voting Share in this offering. Because the selling shareholders may sell or otherwise dispose of the Subordinate Voting Shares covered by this prospectus in a number of different ways and at varying prices, the amount of dilution will vary depending on the price paid for any such shares sold in this offering.
27
The following table provides our cash and cash equivalents (excluding restricted cash) and our capitalization as of March 31, 2022. You should read this table together with our consolidated financial statements and related notes appearing at the end of this prospectus and the sections of this prospectus titled “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and “Description of Capital Stock.”
| March 31, 2022 | ||||
| (in thousands) | ||||
| Cash and cash equivalents | $ | 75,717 | ||
| Debt | ||||
| 10% Senior Secured Notes | $ | 74,935 | ||
| Acquisition Facility | 40,000 | |||
| Acquisition-related promissory notes payable | 35,614 | |||
| Other debt | 14,965 | |||
| Total debt - principal amounts | $ | 165,514 | ||
| Less: debt issuance costs and original issue discounts | (22,542 | ) | ||
| Total debt - carrying amounts | $ | 142,972 | ||
| Finance lease liabilities | 101,736 | |||
| Total debt and finance leases | $ | 244,708 | ||
| Shareholders’ equity: | ||||
| Common stock, no par value; 189,728,625 issued and outstanding | $ | — | ||
| Additional paid-in capital | 456,719 | |||
| Accumulated loss | (262,175 | ) | ||
| Total Jushi shareholders’ equity | $ | 194,544 | ||
| Non-controlling interests | (1,387 | ) | ||
| Total equity | $ | 193,157 | ||
| Total capitalization | $ | 437,865 | ||
28
This prospectus relates to the possible resale by the Selling Shareholders of up to 48,260,954 Subordinate Voting Shares consisting of: (i) 7,491,952 Subordinate Voting Shares issuable upon exercise of outstanding options issued under the Company's equity plan, (ii) 661,607 Subordinate Voting Shares issued upon the exercise of options issued under the Company’s equity plan, (iii) 1,538,326 Subordinate Voting Shares issued as restricted stock awards under the Company's equity plan, (iv) 715,846 Subordinate Voting Shares issued upon exercise of warrants issued in connection with services rendered to the Company; (v) 1,535,000 Subordinate Voting Shares issuable upon exercise of outstanding warrants issued in connection with services provided to the Company; (vi) 6,900,000 Subordinate Voting Shares issuable upon exercise of outstanding warrants issued in connection with the Company's 10% Senior Secured Notes; (vii) 1,311,555 Subordinate Voting Shares issued upon exercise of warrants issued in connection with the Company's 10% Senior Secured Notes; (viii) 11,575,000 Subordinate Voting Shares issuable upon exercise of outstanding warrants issued in private placement transactions; (ix) 198,468 Subordinate Voting Shares issued upon the exercise of warrants issued in private placement transactions; and (x) 16,333,200 Subordinate Voting Shares issued in private placement and acquisition transactions. The Selling Shareholders may from time to time offer and sell any or all of the Subordinate Voting Shares set forth below pursuant to this prospectus. When we refer to the “Selling Shareholders” in this prospectus, we mean the persons listed in the table below, and the pledgees, donees, transferees, assignees, successors and other successors-in-interest who later come to hold any of the Selling Shareholders’ interest in the Subordinate Voting Shares other than through a public sale.
The following table sets forth, based on information currently known by us as of August 4, 2022, (i) the number of Subordinate Voting Shares held of record or beneficially by the Selling Shareholders as of such date (as determined below), (ii) the number of Subordinate Voting Shares that may be offered under this prospectus by the Selling Shareholders and (iii) any material relationships the Selling Shareholders may have had with us within the past three years. The beneficial ownership of the Subordinate Voting Shares set forth in the following table is determined in accordance with Rule 13d-3 under the Exchange Act, and the information is not necessarily indicative of beneficial ownership for any other purpose. Under Rule 13d-3, beneficial ownership includes any shares as to which the selling securityholders have sole or shared voting power or investment power and also any shares which each Selling Shareholder, respectively, has the right to acquire within 60 days of August 4, 2022 through the exercise of any stock option, warrant or other rights. The applicable percentage ownership for each Selling Shareholder listed below is based upon 195,849,084 Subordinate Voting Shares outstanding as of August 4, 2022.
We cannot advise you as to whether the Selling Shareholders will in fact sell any or all of such Subordinate Voting Shares. In addition, the Selling Shareholders may sell, transfer or otherwise dispose of, at any time and from time to time, the Subordinate Voting Shares in transactions exempt from the registration requirements of the Securities Act after the date of this prospectus. A Selling Shareholder may sell all, some or none of such shares in this offering. See “Plan of Distribution.”
Unless otherwise indicated in the footnotes below, the address of each Selling Shareholder is c/o Jushi Holdings Inc., 301 Yamato Road, Suite 3250, Boca Raton, FL 33431.
| Name of Selling Shareholder | Subordinate Voting Shares Owned before the Offering(1) | Subordinate Voting Shares to be Offered for the Selling Shareholder’s Account (2) | Subordinate Voting Shares Owned by the Selling Shareholder after the Offering | Percent of Subordinate Voting Shares to be Owned by the Selling Shareholder after the Offering | ||||||||||||
| James Cacioppo(3) | 36,313,465 | 36,846,665 | 1,466,800 | (12) | * | |||||||||||
| Louis Jon Barack(4) | 5,448,107 | 1,793,000 | 4,321,773 | 2.2 | % | |||||||||||
| Leonardo Garcia-Berg(5) | — | 100,000 | — | * | ||||||||||||
| Benjamin Cross(6) | 419,080 | 60,000 | 359,080 | * | ||||||||||||
| Stephen Monroe(7) | 227,636 | 73,952 | 153,684 | * | ||||||||||||
| Peter Adderton(8) | 169,080 | 60,000 | 109,080 | * | ||||||||||||
| Marina Hahn(9) | 37,301 | 20,000 | 17,301 | * | ||||||||||||
| Meanwhile Productions, LLC | 1,263,622 | 550,000 | 713,622 | * | ||||||||||||
| Navy Capital Green Co-Investment Fund, LLC | 3,272,037 | 872,037 | 2,400,000 | 1.2 | % | |||||||||||
| Navy Capital Green Fund, LP | 2,131,518 | 381,518 | 1,750,000 | * | ||||||||||||
| Ken Rosen and Lisa Rosen | 2,269,900 | 750,000 | 1,519,900 | * | ||||||||||||
| MM-LEC Holdings, LLC | 1,439,618 | 250,000 | 1,189,618 | * | ||||||||||||
| Gabriel Cohen | 80,600 | 80,600 | — | * | ||||||||||||
| Todd Green | 187,877 | 5,952 | 181,925 | * | ||||||||||||
| Green Ache'rs Consulting Limited | 316,623 | 316,623 | — | * | ||||||||||||
| Verdant Nevada LLC | 211,081 | 211,081 | — | * | ||||||||||||
| NuLeaf Capital Investors Group LLC | 2,564,306 | 2,564,306 | — | * | ||||||||||||
| NuLeaf CLV Capital Investors LLC | 488,889 | 488,889 | — | * | ||||||||||||
| Etienne Fontan | 874,324 | 874,324 | — | * | ||||||||||||
| Timothy Schick | 874,324 | 874,324 | — | * | ||||||||||||
| Sean Luse | 749,421 | 749,421 | — | * | ||||||||||||
| Greenvision LLC | 114,416 | 114,416 | — | * | ||||||||||||
| Colin Braid (10) | 102,000 | 58,000 | 44,000 | * | ||||||||||||
| Canaccord Genuity Corp.(11) | 670,201 | 165,846 | 504,355 | * | ||||||||||||
29
| * | Less than 1%. |
| (1) | The number of subordinate voting shares owned prior to the offering and that may be offered for resale includes 1,538,326 shares of common stock issuable upon settlement of restricted stock awards where settlement remains contingent upon satisfaction of vesting conditions. |
| (2) | Includes subordinate voting shares underlying unvested stock options. |
| (3) | Consists of: (i) 4,452,827 subordinate voting shares, 5,385,000 subordinate voting shares underlying stock options and 5,000,000 subordinate voting shares underlying warrants held by Mr. Cacioppo; (ii) 2,500,000 subordinate voting shares and 4,000,000 subordinate voting shares underlying warrants held by OEP Opportunities, L.P.; (iii) 1,400,000 subordinate voting shares and 2,775,000 subordinate voting shares underlying warrants held by One East Capital Advisors, LP; (iv) 2,303,350 subordinate voting shares and 4,175,000 subordinate voting shares underlying warrants held by One East Partners LP; (v) 795,488 subordinate voting shares held by ST2 LLC; (vi) 160,000 subordinate voting shares underlying warrants issued to One East Management Services, LLC; (vii) 3,000,000 subordinate voting shares underlying warrants issued to Serpentine Capital Management Fund II, LLC; and (viii) 900,000 subordinate voting shares underlying warrants issued to JAC Serpentine, LLC. Mr. Cacioppo directly or beneficially owns and controls each of the foregoing entities. Mr. Cacioppo is our Chief Executive Officer and Chairman. |
| (4) | Mr. Barack is our President, Corporate Secretary and Interim Chief Financial Officer. |
| (5) | Mr. Garcia-Berg is our Chief Operations Officer. |
| (6) | Mr. Cross is a member of our board of directors. |
| (7) | Mr. Monroe is a member of our board of directors. |
| (8) | Mr. Adderton is a member of our board of directors. |
| (9) | Ms. Hahn is a member of our board of directors. |
| (10) | Held by Canaccord Genuity Corp. in trust for Mr. Braid. |
| (11) | Includes 292,463 subordinated voting shares held for the benefit of an employee. Such selling shareholder disclaims beneficial ownership of 292,463 of such shares held for the benefit of an employee. Such selling shareholder is affiliated with Canaccord Genuity LLC, a registered broker-dealer. Canaccord Genuity LLC and affiliates have provided investment banking services to the Company from time to time. |
| (12) | Includes: (i) 166,800 subordinate voting shares and 1,000,000 options held by Mr. Cacioppo personally; and (ii) 300,000 subordinate voting shares held by One East Partners LP. |
30
MANAGEMENT’S DISCUSSION AND ANALYSIS OF
FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The following discussion of our financial condition and results of operations should be read together with “Summary Consolidated Financial Data,” and our consolidated financial statements and the related notes included elsewhere in this prospectus. This discussion contains forward-looking statements and involves numerous risks and uncertainties, including but not limited to those described in the “Risk Factors” section of this prospectus. Actual results may differ materially from those contained in any forward-looking statements. You should read “Cautionary Note Regarding Forward-Looking Statements” and “Risk Factors” contained in this prospectus.
Company Overview
We are a vertically integrated, multi-state cannabis operator engaged in retail, distribution, cultivation, and processing operations in both medical and adult-use markets. Our management team is focused on building a diverse portfolio of cannabis assets through opportunistic investments and pursuing application opportunities in attractive limited license jurisdictions. We have targeted assets in highly populated, limited license medical markets on a trajectory toward adult-use legalization, including Pennsylvania and Ohio, markets that are in the process of transitioning to adult-use, namely Virginia, and limited license, fast-growing, large adult-use markets, such as Illinois, Nevada and Massachusetts, and certain municipalities of California.
Refer to the “Business” section and to our consolidated financial statements and the related notes included elsewhere in this prospectus for additional information about us and recent developments.
Factors Affecting our Performance and Related Trends
COVID-19
The COVID-19 pandemic has severely restricted the level of economic activity around the world, including where we operate in the United States. In response to the COVID-19 pandemic the governments of many countries, states, provinces, municipalities and other geographic regions have taken preventative or protective actions, such as imposing restrictions on travel and business operations, ordering temporary closures of businesses and advising or requiring individuals to limit or forego their time outside of their homes. Numerous businesses have temporarily closed voluntarily or closed permanently. Although some preventative or protective actions have been eased or lifted in varying degrees by different governments of various countries, states and municipalities, COVID-19, including new and highly contagious variants of COVID-19, continues to spread quickly throughout the world. Notwithstanding widespread vaccine availability within the U.S., the emergence of COVID-19 variants and slowing vaccination rates in certain localities have resulted in increased infection rates and has caused, and may continue to cause, several jurisdictions to reinstitute certain COVID-19 restrictions. Additional waves of increased COVID-19 infections as well as COVID-19 related restrictions imposed by various governmental authorities (including, for example, requirements to show proof of vaccination), could negatively impact our supply chain, as well as traffic and sales volume for our products, which in turn could have an adverse effect on our business, financial condition and results of operations.
At the onset of the COVID-19 pandemic, the Company implemented new procedures at all operating locations to better protect the health and safety of our employees, medical patients, and customers across its network of dispensaries. Depending on the location, some of the initiatives include, but are not limited to: reducing the number of point-of-sale registers, restricting the number of people permitted in-store, restricting general store hours to permit access to those most susceptible to infection, and offering curbside pick-up. The Company has also directed a significant amount of traffic to its recently launched online informational tool and reservation platform, www.beyond-hello.com, which enables a medical patient or customer to view real-time pricing and product availability, and reserve products for convenient in-store pick-up at BEYOND/HELLO™ locations across Pennsylvania, Illinois, California, and Virginia.
Although the Company’s dispensaries and grower-processor facilities have remained open throughout the pandemic, the Company has experienced disruptions related to supply chains, labor supply and delays in expansion projects and, in certain states, cannabis license applications, and may experience further disruptions as a result of COVID-19. In particular:
| · | We may experience significant reductions or volatility in demand for our products as customers may not be able to purchase our products due to illness, quarantine or government or self-imposed restrictions placed on our dispensaries’ operations or reduction in operational hours due to labor shortages; |
| · | We may be unable to meet production demand if a particular grower-processor or section of the grower-processor is closed due to illness, quarantine or government or self-imposed restrictions placed on our facility operations or reduction in operational hours due to labor shortages; |
| · | Social distancing measures or changes in consumer spending behaviors due to COVID-19 may impact traffic in our dispensaries and such actions could result in a loss of sales and profit; |
| · | We may experience temporary or long-term disruptions in our supply chain; |
| · | Transportation delays and cost increases, closures or disruptions of businesses and facilities, or social, economic, political or labor instability, may impact our or our suppliers’ operations or our customers; and |
| · | We may experience impairments of assets in markets that are disproportionately impacted by these and other effects of the COVID-19 pandemic. |
To date, our financial condition and results of operations have not been materially impacted by COVID-19. The extent to which the COVID-19 pandemic impacts our future results will depend on future developments, which are highly uncertain and cannot be predicted with certainty, including possible future outbreaks of new strains of the virus and governmental and consumer responses to such future developments.
Competition and Pricing Pressure
The cannabis industry is subject to significant competition and pricing pressures, which is often market specific and may be transitory from period to period. We may experience significant competitive pricing pressures as well as competitive products and service providers in the markets in which we operate. Several significant competitors may offer products and/or services with prices that may match or are lower than ours. We believe that the products and services we offer are generally competitive with those offered by other cannabis companies. It is possible that one or more of our competitors could develop a significant research advantage over us that allows them to provide superior products or pricing, which could put us at a competitive disadvantage. Continued pricing pressure or shifts in customer preferences could adversely impact our customer base or pricing structure and have a material impact on our results of operations in future periods.
31
Components of Results of Operations
Revenue
Our significant revenue streams are retail and wholesale. Retail revenues are primarily from the sale of cannabis products at our medical and adult-use dispensaries. Wholesale revenues are from the cultivation, production and distribution of our cannabis products sold to medical and adult-use dispensaries.
Cost of Goods Sold and Gross Profit
Cost of goods sold includes costs related to the internal cultivation and production of cannabis products, the acquisition cost of finished goods from other licensed producers operating within the states we operate, and packaging costs. Costs to cultivate and produce our cannabis products are primarily comprised of: direct labor costs; direct materials including nutrients and supplies; and facilities costs including depreciation, repairs and maintenance, rent, utilities, property taxes, security and insurance. The primary factors that can impact gross profit include the mix and margins on products sold. Costs are also affected by various state regulations that limit the sourcing and procurement of cannabis product, which may create fluctuations in margins over comparative periods as the regulatory environment changes.
Operating Expenses
Selling, general and administrative expenses represent costs incurred at our corporate and administrative offices, primarily related to: compensation expenses; depreciation and amortization; professional fees and legal expenses; marketing, advertising and selling costs; facility-related expenses, including rent and security; insurance; software and technology expenses; and impairments.
We expect to continue to invest considerably in this area to support our expansion plans and to support the increasing complexity of the cannabis business. As we continue to expand and open additional dispensaries, we expect our sales and marketing expenses to continue to increase. Furthermore, we expect to continue to incur acquisition and deal costs related to our expansion plans, and we anticipate an increase in compensation expenses related to recruiting and hiring new talent, and in professional fees associated with becoming compliant with the Sarbanes-Oxley Act, SEC reporting requirements and other costs associated with being an SEC-registered company.
Other Income (Expense), Net
Other income (expense), net, consists of interest expense, net of interest income; net investment gains (losses); net gains on business combinations; gains (losses) on debt modification; and other.
Interest income relates primarily to interest earned on cash and cash equivalent balances in bank accounts. Interest expense consists primarily of interest on borrowings. Net gains on business combinations result from bargain purchases, the related tax effects and other related adjustments. Net investment gains (losses) comprise realized and unrealized gains on securities, equity investments and financial assets, such as notes or contingent asset receivables, if applicable. Gains (losses) on debt modifications relate to exchanged or extinguished debt and other related modifications.
Provision for Income Taxes
Provision for income taxes is calculated using the asset and liability method. Deferred income tax assets and liabilities are determined based on enacted tax rates and laws for the years in which the differences are expected to reverse. Deferred tax assets are reduced by a valuation allowance when, in the opinion of management, it is more likely than not that some portion or all of the deferred tax assets will not be realized.
As we operate in the cannabis industry, we are subject to the limits of Section 280E of the Code under which we are only allowed to deduct expenses directly related to the cost of producing or acquiring the products we sell.
32
Results of Operations
(amounts expressed in thousands of U.S. dollars, unless otherwise stated)
For
the Three Months (unaudited) | For the Year Ended December 31, | |||||||||||||||||||||||
| 2022 | 2021 | % Change | 2021 | 2020 | % Change | |||||||||||||||||||
| REVENUE, NET | $ | 61,888 | $ | 41,675 | 49 | % | $ | 209,292 | $ | 80,772 | 159 | % | ||||||||||||
| COST OF GOODS SOLD | (42,776 | ) | (22,934 | ) | 87 | % | (125,898 | ) | (42,431 | ) | 197 | % | ||||||||||||
| GROSS PROFIT | $ | 19,112 | $ | 18,741 | 2 | % | $ | 83,394 | $ | 38,341 | 118 | % | ||||||||||||
| OPERATING EXPENSES | $ | 37,308 | $ | 21,911 | 70 | % | $ | 119,159 | $ | 54,895 | 117 | % | ||||||||||||
| LOSS FROM OPERATIONS | $ | (18,196 | ) | $ | (3,170 | ) | 474 | % | $ | (35,765 | ) | $ | (16,554 | ) | 116 | % | ||||||||
| OTHER (EXPENSE) INCOME: | ||||||||||||||||||||||||
| Interest expense, net | $ | (10,116 | ) | $ | (6,835 | ) | 48 | % | $ | (30,610 | ) | $ | (15,333 | ) | 100 | % | ||||||||
| Fair value gains (losses) on derivatives | 14,309 | (9,358 | ) | 253 | % | 105,170 | (173,707 | ) | 161 | % | ||||||||||||||
| Other, net | (703 | ) | (3,376 | ) | (79 | )% | 8,309 | 3,702 | 124 | % | ||||||||||||||
| Total net other income (expense) | $ | 3,490 | $ | (19,569 | ) | 118 | % | $ | 82,869 | $ | (185,338 | ) | 145 | % | ||||||||||
| INCOME (LOSS) BEFORE PROVISION FOR INCOME TAXES | $ | (14,706 | ) | $ | (22,739 | ) | 35 | % | $ | 47,104 | $ | (201,892 | ) | 123 | % | |||||||||
| Provision for income taxes | (5,051 | ) | (8,312 | ) | (39 | )% | (29,625 | ) | (10,623 | ) | 179 | % | ||||||||||||
| NET INCOME (LOSS) AND COMPREHENSIVE INCOME (LOSS) | $ | (19,757 | ) | $ | (31,051 | ) | 36 | % | $ | 17,479 | $ | (212,515 | ) | 108 | % | |||||||||
| Net loss attributable to non-controlling interests | — | (175 | ) | (100 | )% | (2,772 | ) | (1,908 | ) | 45 | % | |||||||||||||
| NET INCOME (LOSS) AND COMPREHENSIVE INCOME (LOSS) ATTRIBUTABLE TO JUSHI SHAREHOLDERS | $ | (19,757 | ) | $ | (30,876 | ) | 36 | % | $ | 20,251 | $ | (210,607 | ) | 110 | % | |||||||||
| NET INCOME (LOSS) AND COMPREHENSIVE INCOME (LOSS) PER SHARE ATTRIBUTABLE TO JUSHI SHAREHOLDERS - BASIC | $ | (0.11 | ) | $ | (0.20 | ) | 45 | % | $ | 0.12 | $ | (1.94 | ) | 106 | % | |||||||||
| Weighted average shares outstanding - BASIC | 183,226,027 | 157,176,375 | 17 | % | 170,292,035 | 108,485,158 | 57 | % | ||||||||||||||||
| NET (LOSS) AND COMPREHENSIVE (LOSS) PER SHARE ATTRIBUTABLE TO JUSHI SHAREHOLDERS - DILUTED | $ | (0.16 | ) | $ | (0.20 | ) | 20 | % | $ | (0.42 | ) | $ | (1.94 | ) | 78 | % | ||||||||
| Weighted average shares outstanding - DILUTED | 207,838,906 | 157,176,375 | 32 | % | 201,610,251 | 108,485,158 | 86 | % | ||||||||||||||||
33
Comparison of First Quarter Results
(amounts expressed in thousands of U.S. dollars, unless otherwise stated)
Revenue, Net
The following table presents revenue by type for the periods indicated:
| Three Months Ended March 31, 2022 | Three Months Ended March 31, 2021 | |||||||||||||||||||||||
| Gross revenue | Intercompany revenue | Revenue to external customers | Gross revenue | Intercompany revenue | Revenue to external customers | |||||||||||||||||||
| Retail cannabis | $ | 57,955 | $ | — | $ | 57,955 | $ | 39,277 | $ | — | $ | 39,277 | ||||||||||||
| Wholesale cannabis | 9,443 | (5,595 | ) | 3,848 | 4,192 | (1,883 | ) | 2,309 | ||||||||||||||||
| Other | 85 | — | 85 | 89 | — | 89 | ||||||||||||||||||
| Eliminations | (5,595 | ) | 5,595 | — | (1,883 | ) | 1,883 | — | ||||||||||||||||
| Consolidated | $ | 61,888 | $ | — | $ | 61,888 | $ | 41,675 | $ | — | $ | 41,675 | ||||||||||||
Revenue, net, for the three months ended March 31, 2022 totaled $61,888, as compared to $41,675 for the three months ended March 31, 2021, an increase of $20,213, or 49%. The increase in retail revenue is due primarily to the Company’s expansion of cannabis operations from build outs and acquisitions. Retail revenue for the three months ended March 31, 2022 was derived from twenty-nine cannabis dispensaries located in Pennsylvania (eighteen), Illinois (four), Massachusetts (two), California (two), Virginia (two) and Nevada (one), whereas, for the three months ended March 31, 2021, Retail revenue was derived from seventeen cannabis dispensaries located in Pennsylvania (eleven), Illinois (four), California (one) and Virginia (one).
The increase in wholesale revenue is primarily attributable to increases in cultivation and manufacturing activity at our grower processor facilities: (i) in Massachusetts due to the acquisition of Nature’s Remedy, which occurred in the third quarter of 2021; (ii) in Virginia due to the commencement of operations at the Dalitso facility in the third quarter of 2021; and (iii) in Pennsylvania due to continued increase sales volume. Wholesale revenue includes inter-segment revenue of $5,595, which is eliminated in consolidation.
Cost of Goods Sold and Gross Profit
Cost of goods sold totaled $42,776 for the three months ended March 31, 2022, as compared to $22,934 for the three months ended March 31, 2021, an increase of $19,842, or 87%. The increase in costs of goods sold is primarily attributable to the increase in amount of products sold.
Gross profit totaled $19,112 for the three months ended March 31, 2022, as compared to $18,741 for three months ended March 31, 2021, an increase of approximately $371, or 2%. As a percentage of revenue, gross profit for the three months ended March 31, 2022 and 2021, was 31% and 45%, respectively. Gross margin decreased primarily due to: (1) approximately $3,500 of labor and overhead costs related to the ramp up of operations in our grower-processor facilities in Virginia and Massachusetts and start-up costs at our new retail dispensaries; and (2) the sell through of inventory acquired in the Nature's Remedy and Apothecarium acquisitions which had a fair value step-up of approximately $3,000. Gross margins were also impacted by the increased promotional activity at our retail locations in Illinois and Massachusetts, and pricing compression in wholesale as we continue to build-out our brands across state markets.
34
Operating Expenses
Operating expenses for the three months ended March 31, 2022 were $37,308, as compared to $21,911 for the three months ended March 31, 2021, an increase of $15,397, or 70%.
| Three Months Ended March 31, | ||||||||||||||||
| 2022 | 2021 | $ Change | % Change | |||||||||||||
| Salaries, wages and employee related expenses (S&W) | $ | 17,336 | $ | 9,882 | 7,454 | 75 | % | |||||||||
| Stock-based compensation expense | 6,964 | 4,013 | 2,951 | 74 | % | |||||||||||
| Rent and related expenses | 3,089 | 1,799 | 1,290 | 72 | % | |||||||||||
| Professional fees and legal expenses | 2,706 | 1,693 | 1,013 | 60 | % | |||||||||||
| Depreciation and amortization expense | 2,256 | 974 | 1,282 | 132 | % | |||||||||||
| Software and technology | 1,530 | 567 | 963 | 170 | % | |||||||||||
| Marketing and selling | 969 | 675 | 294 | 44 | % | |||||||||||
| Travel, entertainment and conferences | 739 | 405 | 334 | 82 | % | |||||||||||
| Insurance | 599 | 755 | (156 | ) | (21 | )% | ||||||||||
| Administration and application fees | 140 | 353 | (213 | ) | (60 | )% | ||||||||||
| Acquisition and deal costs | 141 | 238 | (97 | ) | (41 | )% | ||||||||||
| Other G&A | 839 | 557 | 282 | 51 | % | |||||||||||
| Total operating expenses | $ | 37,308 | $ | 21,911 | $ | 15,397 | 70 | % | ||||||||
The total increase in operating expenses is due to the increase in the size and scope of our general and administrative functions to support our expanded operations resulting from organic growth and acquisitions. The primary increases are from an increase in: salaries, wages, labor and employee related expenses as a result of the increase in the number of employees to support our ongoing growth and resulting from recent acquisitions; share-based compensation primarily due to recent stock options granted to new employees and management; professional fees and legal expenses, primarily due to our transition to accounting principles generally accepted in the U.S. (US GAAP) reporting and costs associated with our registration with the SEC, which we expect to complete by the third quarter of 2022; and depreciation and amortization expense and rent and related expenses due to the additions of property, plant and equipment and finance lease right-of-use assets from acquisitions and investment in infrastructure as we continue to scale.
Other (Expense) Income
Interest Expense, Net
Interest expense, net, was $10,116 for the three months ended March 31, 2022, as compared to $6,835 for the three months ended March 31, 2021, an increase of $3,281, or 48%. The increase in interest expense, net, is due primarily to an increase in interest-bearing borrowings including finance leases and acquisition-related financing.
Fair Value Changes in Derivatives
Fair value changes in derivatives was a net gain of $14,309 for the three months ended March 31, 2022, as compared to a net loss of $9,358 for the three months ended March 31, 2021. Fair value changes in derivatives include the fair value gains or losses relating to the derivative warrants liability. The derivative warrants are required to be remeasured at fair value at each reporting period. The fair value changes in derivatives for the three months ended March 31, 2022 and 2021 were primarily attributable to the movement in the Company’s stock price during the corresponding period.
Other, Net
Other expense, net, was $703 for the three months ended March 31, 2022, as compared to $3,376 for the three months ended March 31, 2021, a decrease of $2,673 or 79%. Other expense, net, for the three months ended March 31, 2021 primarily related to losses on redemptions of 10% Senior Secured Notes of $3,815, partially offset by $1,149 gains on investments in securities.
Income Tax Expense
Total income tax expense was $5,051 for the three months ended March 31, 2022, as compared to $8,312 for the three months ended March 31, 2021, a decrease of $3,261, or 39%. The decrease in income tax expense is primarily due to a reduction in our accrual for uncertain tax positions related to periods no longer open under the statute of limitations.
35
Comparison of Fiscal Year Results
(amounts expressed in thousands of U.S. dollars, unless otherwise stated)
Revenue, Net
The following table presents revenue by type for the years indicated:
| Year Ended December 31, 2021 | Year Ended December 31, 2020 | |||||||||||||||||||||||
Gross revenue | Intercompany revenue | Revenue to external customers | Gross revenue | Intercompany revenue | Revenue to external customers | |||||||||||||||||||
| Retail cannabis | $ | 195,085 | $ | — | $ | 195,085 | $ | 75,499 | $ | — | $ | 75,499 | ||||||||||||
| Wholesale cannabis | 29,969 | (16,177 | ) | 13,792 | 6,639 | (1,901 | ) | 4,738 | ||||||||||||||||
| Other | 415 | — | 415 | 535 | — | 535 | ||||||||||||||||||
| Eliminations | (16,177 | ) | 16,177 | — | (1,901 | ) | 1,901 | — | ||||||||||||||||
| Consolidated revenue | $ | 209,292 | $ | — | $ | 209,292 | $ | 80,772 | $ | — | $ | 80,772 | ||||||||||||
Revenue, net, for the year ended December 31, 2021 totaled $209,292, as compared to $80,772 for the year ended December 31, 2020, an increase of $128,520, or 159%. The increase in revenue is due primarily to increases in retail and wholesale cannabis revenues in both medical and adult-use markets. The increase in retail revenue is primarily attributable to the build-out of retail dispensaries, acquisitions made in 2021, along with the continued growth in states where the Company operates. Retail revenue for the year ended December 31, 2021 was derived from a total of twenty-eight cannabis dispensaries located in Pennsylvania (eighteen), Illinois (four), Massachusetts (two), California (two), and Virginia (two), whereas, in the prior year, retail revenue was derived from a total of fifteen cannabis dispensaries located in Pennsylvania (ten), Illinois (three), California (one) and Virginia (one). The increase in wholesale revenue is primarily attributable to increases in cultivation and manufacturing activity at our grower processor facilities: (i) in Pennsylvania due to the acquisition of PAMS in the third quarter of 2020; (ii) in Massachusetts due to the acquisition of Nature’s Remedy in the third quarter of 2021; and (iii) in Virginia due to the commencement of operations at the Dalitso facility in the third quarter of 2021.
Cost of Goods Sold and Gross Profit
Cost of goods sold totaled $125,898 for the year ended December 31, 2021, as compared to $42,431 for the year ended December 31, 2020, an increase of $83,467, or 197%. The increase in costs of goods sold is primarily attributable to the increase in revenue.
Gross profit totaled $83,394 for the year ended December 31, 2021, as compared to $38,341 for year ended December 31, 2020, an increase of approximately $45,053, or 118%. Gross profit increased primarily due to the increase in revenue. As a percentage of revenue, gross profit for the year ended December 31, 2021 and 2020, was 40% and 47%, respectively. Gross margin decreased primarily due to an increase in new location start-up costs of $4,795, a one-time inventory reserve for certain vape products subject to a recall in Pennsylvania of $2,021, price compression in our largest markets of Pennsylvania and Illinois and at the wholesale level due to increased promotional activity as we continue to build-out our brands across state markets. Start-up costs represent costs incurred to prepare a location for its intended use. The inventory recall reserve relates to the potential impact of the Pennsylvania Department of Health recall and ban of vape products containing certain cannabis concentrates.
36
Operating Expenses
Operating expenses for the year ended December 31, 2021 were $119,159, as compared to $54,895 for the year ended December 31, 2020, an increase of $64,264, or 117%.
| Year Ended December 31, | $ | |||||||||||||||
| 2021 | 2020 | Change | % Change | |||||||||||||
| Salaries, wages and employee related expenses (S&W) | $ | 58,228 | $ | 21,781 | $ | 36,447 | 167 | % | ||||||||
| Stock-based compensation expense | 14,506 | 9,592 | 4,914 | 51 | % | |||||||||||
| Depreciation and amortization expense | 5,805 | 4,154 | 1,651 | 40 | % | |||||||||||
| Rent and related expenses | 7,115 | 3,754 | 3,361 | 90 | % | |||||||||||
| Professional fees and legal expenses | 6,507 | 3,975 | $ | 2,532 | 64 | % | ||||||||||
| Marketing and selling | 3,563 | 2,511 | 1,052 | 42 | % | |||||||||||
| Insurance | 2,703 | 1,988 | 715 | 36 | % | |||||||||||
| Administration and application fees | 1,131 | 1,589 | (458 | ) | (29 | )% | ||||||||||
| Software and technology | 3,313 | 1,049 | 2,264 | 216 | % | |||||||||||
| Travel, entertainment and conferences | 2,352 | 756 | 1,596 | 211 | % | |||||||||||
| Facility rent expense | 2,607 | 1,489 | 1,118 | 75 | % | |||||||||||
| Acquisition and deal costs | 1,624 | 810 | 814 | 100 | % | |||||||||||
| Asset impairment | 4,561 | — | 4,561 | 100 | % | |||||||||||
| Goodwill impairment | 1,783 | — | 1,783 | 100 | % | |||||||||||
| Other G&A | 3,361 | 1,447 | 1,914 | 132 | % | |||||||||||
| Total operating expenses | $ | 119,159 | $ | 54,895 | $ | 64,264 | 117 | % | ||||||||
The total increase in operating expenses is due to the increase in the size and scope of our general and administrative functions to support our expanded operations resulting from organic growth and acquisitions. The primary increases are from an increase in salaries, wages, labor and employee-related expenses as well as an increase in share-based compensation, as a result of the increase in the number of employees. In addition, an increase in depreciation and amortization expense and facilities-related expenses were due to the additions of property, plant and equipment and finance lease right-of-use assets from acquisitions and investments in infrastructure that resulted in more capitalized assets. Also, the Company had an increase in professional fees and legal expenses, primarily due to our transition to US GAAP reporting and costs associated with our registration with the SEC, which we expect to complete by the third quarter of 2022. The Company also recorded a goodwill impairment charge of $1,783 and an asset impairment charge of $4,561 for the year ended December 31, 2021 related to the write-off of goodwill associated with Nevada upon completion of the Company’s annual impairment assessment and the asset impairment charges relate to Jushi Europe SA (Jushi Europe).
Other (Expense) Income, Net
Interest Expense, Net
Interest expense, net, was $30,610 for the year ended December 31, 2021, as compared to $15,333 for the year ended December 31, 2020, an increase of $15,277, or 100%. The increase in interest expense, net, is due primarily to an increase in interest-bearing borrowings including finance leases and acquisition-related financing.
37
Fair Value Changes in Derivatives
Fair value changes in derivatives was a net gain of $105,170 for the year ended December 31, 2021, as compared to a net loss of $173,707 for the year ended December 31, 2020. The total net fair value gain on derivatives for the year ended December 31, 2021 were attributed to fair value gains relating to the derivative warrants of $104,594 and a gain on the Pennsylvania Dispensary Solutions, LLC (PADS) purchase option of $575. At the time of the PAMS acquisition, as part of the agreements with Vireo Health, Inc (Vireo), the seller of PAMS, Jushi received an assignable purchase option (PADS Purchase Option) to acquire 100% of the equity of PADS. The derivative warrants were issued in connection with the debt offerings announced in December 2019 and June 2020 and are required to be remeasured at fair value at each reporting period. The gains on derivative warrants for the year ended December 31, 2021 were due to the decrease in the fair value of the derivative warrants liability primarily as a result of a decrease in the Company’s stock price during the year ended December 31, 2021.
Other, Net
Other income, net, was $8,309 for the year ended December 31, 2021, as compared to $3,702 for the year ended December 31, 2020, an increase of approximately $4,607, or 124%. The increase was primarily attributable to: (i) a $10,248 gain on the legal settlement of a breach of contract case involving San Felasco Nurseries, Inc. (the SFN Litigation); (ii) an increase in net investment gains of $3,689 primarily from net fair value gains on investments during 2021 compared to declines in the fair values of the Company’s holdings in investments caused by the general market decline experienced during the first quarter of 2020 as a result of COVID-19; (iii) a decrease of $10,149 in net gains on business combinations, which related to the 2020 acquisitions of PAMS and TGS Illinois Holdings, LLC (TGSIH) (now known as Beyond Hello IL Holdings, LLC (BHILH); and (iv) an increase in losses on debt redemptions and extinguishments/modifications of $1,962.
Income Tax Expense
Total income tax expense was $29,625 for the year ended December 31, 2021, as compared to $10,623 for the year ended December 31, 2020, an increase of $19,002, or 179%. The increase was comprised of an increase in current tax expense of $20,555 and a decrease in deferred tax expense of $1,553. The increase in current tax expense relates primarily to the increase in taxable gross profit generated from the Company’s increased retail and wholesale revenues. The Company also realized additional taxable income from a legal settlement related to the SFN litigation.
Net Loss Attributable to Non-Controlling Interests
Net loss attributable to non-controlling interests was $2,772 for the year ended December 31, 2021, as compared to $1,908 for the year ended December 31, 2020, an increase of $864. Net loss attributable to non-controlling interests for the year ended December 31, 2021, primarily relates to the non-controlling interests of Jushi Europe, whereas net loss attributable to non-controlling interests for the year ended December 31, 2020 related to the non-controlling interests of Dalitso, Jushi Europe, Agape Total Health Care Inc. (Agape) and other non-material non-controlling interests. The non-controlling interests of Dalitso and Agape were purchased by the Company during the fourth quarter of 2020 and the first quarter of 2021, respectively.
38
Liquidity and Capital Resources
Cash Flows
(amounts expressed in thousands of U.S. dollars, unless otherwise stated)
The major components of our statements of cash flows for the periods presented are as follows:
For
the Three Months Ended | For the Year Ended December 31, | |||||||||||||||
| 2022 | 2021 | 2021 | 2020 | |||||||||||||
| Net cash flows used in operating activities (1) | $ | (2,666 | ) | $ | (3,121 | ) | $ | (14,304 | ) | $ | (12,364 | ) | ||||
| Net cash flows used in investing activities | (33,068 | ) | (8,906 | ) | (113,455 | ) | (45,800 | ) | ||||||||
| Net cash flows provided by financing activities (1) | 16,497 | 88,295 | 137,663 | 105,132 | ||||||||||||
| Effect of currency translation on cash | (9 | ) | (40 | ) | (274 | ) | (47 | ) | ||||||||
| Net change in cash and cash equivalents | $ | (19,246 | ) | $ | 76,228 | $ | 9,630 | $ | 46,921 | |||||||
(1) The Company restated the classification of interest payments on its finance leases in the consolidated statement of cash flows during the year ended December 31, 2021. The correction resulted in an increase in net cash flows used in operating activities from $7,928 (as reported) to $14,304 (as restated) and a corresponding increase in net cash flows provided by financing activities from $131,287 (as reported) to $137,663 (as restated). Refer to Note 25 - Correction of Previously Issued Financial Statements of the audited consolidated financial statements included elsewhere in this prospectus.
Cash Flows Used in Operating Activities
Three Months Ended March 31, 2022 and 2021
Net cash used in operations during the three months ended March 31, 2022 was $2,666, as compared to $3,121 for the three months ended March 31, 2021. The improvement in cash used in operations for the three months ended March 31, 2022 is due primarily to improved management of working capital, partially offset by an increase in net loss after non-cash adjustments.
Year Ended December 31, 2021 and 2020
Net cash used in operations during the year ended December 31, 2021 was $14,304, as compared to $12,364 for the year ended December 31, 2020. The increase in cash used in operations is due primarily to increased operating expenses to support our growth and interest paid on finance leases, partially offset by improved management of working capital.
Cash Flows Used in Investing Activities
Three Months Ended March 31, 2022 and 2021
Net cash used in investing activities totaled $33,068 for the three months ended March 31, 2022, as compared to $8,906 for the three months ended March 31, 2021. The net cash used in investing activities for the three months ended March 31, 2022 was comprised of: $26,476 for the purchases of property, plant and equipment for use in the Company’s operations; and $6,592 paid for the acquisition of Apothecarium, net of cash acquired. The net cash used in investing activities for the three months ended March 31, 2021 was comprised of: $3,592 paid for the acquisition of Grover Beach; $8,566 for the purchases of property, plant and equipment for use in the Company’s operations; partially offset by: $3,252 in proceeds from sale of investments.
Year Ended December 31, 2021 and 2020
Net cash used in investing activities totaled $113,455 for the year ended December 31, 2021, as compared to $45,800 for the year ended December 31, 2020. The net cash used in investing activities for the year ended December 31, 2021 was comprised of: $75,296 for the purchases of and deposits for property, plant and equipment for use in the Company’s operations; $47,308 in payments for the acquisitions of Nature’s Remedy, Ohio Green Grow LLC, OhiGrow, Grover Beach and Organic Solutions of the Desert, LLC, net of cash acquired; partially offset by: $9,149 in proceeds from sales and redemptions of investments.
The net cash used in investing activities for the year ended December 31, 2020 was comprised of: $30,117 in payments for the acquisitions of PAMS, PADS (including the PADS Purchase Option), Agape, GSG SBCA, Inc. (GSG Santa Barbara) and BHILH, net of cash acquired (and excluding payments for non-controlling interests); $22,780 for the purchases of and deposits for property, plant and equipment; $11,500 in payments for investments in securities and an equity investment; partially offset by: $18,597 in proceeds from investments and financial assets.
Cash Flows provided by Financing Activities
Three Months Ended March 31, 2022 and 2021
Net cash provided by financing activities totaled $16,497 for the three months ended March 31, 2022, as compared to $88,295 for the three months ended March 31, 2021. The net cash provided by financing activities for the three months ended March 31, 2022 was comprised of: $13,680 in proceeds from private placement equity offerings in January and February 2022; $3,265 in proceeds from other debt; and $541 in proceeds from the exercise of warrants and stock options; partially offset by: $601 in lease obligation payments; $258 in principal redemption repayments on the 10% Senior Secured Notes; and $130 in payments on other debt.
The net cash provided by financing activities for the three months ended March 31, 2021 was comprised of: $85,660 in proceeds from public equity offerings, net of issuance costs, in January and February 2021; $9,886 in proceeds from the exercise of warrants and stock options; and $1,134 in proceeds from other debt; partially offset by: $8,134 in principal redemption repayments on the 10% Senior Secured Notes; and $251 in lease obligation payments.
Year Ended December 31, 2021 and 2020
Net cash provided by financing activities totaled $137,663 for the year ended December 31, 2021, as compared to $105,132 for the year ended December 31, 2020. The net cash provided by financing activities for the year ended December 31, 2021 was comprised of: $85,660 in proceeds from the issuance of shares for cash, net of issuance costs, in connection with public offerings in January and February 2021; $17,128 in proceeds from the exercise of warrants and stock options; $38,299 in proceeds from the new Acquisition Facility and $7,910 in proceeds from other debt instruments, partially offset by: $8,134 in principal redemption repayments on the 10% Senior Secured Notes; $2,037 in payments on acquisition-related promissory notes and other debt; and $1,163 in lease obligation payments.
The net cash provided by financing activities for the year ended December 31, 2020 was comprised of: $51,861 in proceeds from the issuance of the 10% Senior Secured Notes and related warrants, net of financing costs; $46,587 in proceeds from the exercise of warrants and stock options; $29,243 in proceeds from the issuance of shares for cash, net of issuance costs, in connection with a public offering in October of 2020; $3,529 in proceeds from financing obligations and an unsecured credit line, net of a repayments; proceeds of $1,994 from a contribution for Jushi Europe from the non-controlling partner net of other immaterial transactions; partially offset by: $24,003 in principal payments on acquisition-related promissory notes payable; $2,231 in payments for acquisitions from non-controlling interests for Dalitso and BHILH; and $1,848 in lease obligation payments.
39
Balance Sheet Exposure
As of March 31, 2022, primarily all of our balance sheet is exposed to U.S. cannabis-related activities. We believe our operations are in material compliance with all applicable state and local laws, regulations and licensing requirements in the states in which we operate. However, cannabis remains illegal under U.S. federal law. All of our revenue is derived from U.S. cannabis operations. For information about risks related to U.S. cannabis operations, please refer to “Risk Factors” in this prospectus.
Sources and Uses of Liquidity
Since our inception, we have funded our operations and capital spending through cash flows from sales, and the issuance of debt and equity. Jushi has successfully raised approximately $461 million to date (which includes equity offerings, 10% Senior Secured Notes, warrant/option exercises and draw downs on the Acquisition Facility), of which approximately $47 million was invested by management and insiders. We generate cash from sales and are deploying capital to acquire and develop assets capable of producing additional revenues and earnings over both the immediate and long term to support our business growth and expansion. As of March 31, 2022, we have borrowed $40 million from the $100 million Acquisition Facility to fund the cash portions of acquisitions. We may increase the total commitment of the Acquisition Facility by an aggregate amount of up to $25 million subject to certain conditions, and have the ability under our current base shelf prospectus in Canada to issue C$500 million subscription receipts, debt securities, convertible securities, warrants, subordinate voting shares, and units, or any combination thereof.
Our primary uses of cash are for working capital requirements, acquisitions, capital expenditures and debt service payments. Working capital is used principally for our personnel as well as costs related to the purchase of inventory, cultivation, manufacture and production of our products. Our capital expenditures consist primarily of the construction and improvement of grower-processer facilities and retail dispensaries.
As reflected in our unaudited interim condensed consolidated financial statements included elsewhere in this prospectus, we have incurred losses from operations for the three months ended March 31, 2022, have an accumulated deficit of $262,175 as of March 31, 2022, and have cash and cash equivalents of $75,717 as of March 31, 2022. As discussed in Note 9 – Debt in the unaudited interim condensed consolidated financial statements, the 10% Senior Secured Notes, which mature on January 15, 2023, had an aggregate principal amount outstanding of $74,935. In addition, the Acquisition Facility (see Note 9 – Debt in the unaudited interim condensed consolidated financial statements) required us to maintain certain covenants which we may not have been in compliance with if the court accepted Jushi Europe’s petition for bankruptcy. We were also projected to violate certain financial covenants further within the next twelve months from March 31, 2022. We have been working with various lenders to refinance the 10% Senior Secured Notes at terms most favorable to us. Given the changing conditions in the debt capital markets, refinancing term sheets are still being negotiated. These conditions raised a substantial doubt regarding our ability to continue as a going concern.
In April 2022, we entered into an amendment with the lender of the Acquisition Facility, which included a waiver related to Jushi Europe’s bankruptcy and a change to the terms of the Total Leverage ratio, as defined in the Acquisition Facility agreement, and deferred the commencement date of leverage testing under the Acquisition Facility. We are pursuing strategies to obtain the required additional funding primarily to fund the 10% Senior Secured Notes and future operations. The strategies may include, but are not limited to: (i) ongoing efforts with certain lenders to refinance the 10% Senior Secured Notes; (ii) deferral of certain expenditures, including capital projects, and reallocate funds for debt repayment, if the need arose; (iii) alternative sources of debt and equity financing, including secured borrowings and through a base shelf prospectus, which allows us to offer up to C$500,000 in securities in Canada through the end of 2023. However, there can be no assurance that we will be able to refinance the 10% Senior Secured Notes, generate positive results from operations, or obtain additional liquidity when needed or under acceptable terms, if at all.
40
Description of Indebtedness
The Company had the following instruments as of March 31, 2022:
| Designation of Instrument (1) | Number March
31, | Aggregate Amount | Interest Rate | Conversion Price | Maturity Date | |||||||||||
| 10% Senior Secured Notes | 68,122 | (2) | $ | 74.9 | 10 | % | N/A | 01/15/2023 | ||||||||
| Acquisition Facility | 1 | $ | 40.0 | 9.5 | % | N/A | 10/21/2026 | |||||||||
| Unsecured Promissory Note - Nature's Remedy | 1 | $ | 11.5 | 8 | % | N/A | 09/10/2024 | |||||||||
| Unsecured Promissory Note - Nature's Remedy | 1 | $ | 5.0 | 8 | % | N/A | 09/10/2026 | |||||||||
| Unsecured Promissory Note - Apothecarium | 1 | $ | 5.9 | 0 | % | N/A | 3/16/2027 | |||||||||
| Unsecured Promissory Note - Apothecarium | 1 | $ | 3.9 | 0 | % | N/A | 3/16/2027 | |||||||||
| Unsecured Promissory Note - PAMS | 1 | $ | 3.8 | 8 | % | N/A | 08/11/2024 | |||||||||
| Secured Promissory Note - OSD | 1 | $ | 3.1 | 4 | % | N/A | 04/30/2027 | |||||||||
| Unsecured Credit Line - Jushi Europe (3) | 2 | $ | 1.7 | 5 | % | N/A | 11/11/2024 | |||||||||
| Unsecured Credit Line - Jushi Europe (4) | 2 | $ | 1.7 | 0.5 | % | N/A | 03/07/2022 | |||||||||
| Secured Promissory Note - Arlington Facility | 1 | $ | 4.9 | 5.9 | % | N/A | 01/01/2027 | |||||||||
| Unsecured Convertible Promissory Note - Dalitso (5) | 1 | $ | 2.4 | 1 | % | $2.65 | 11/19/2022 | |||||||||
Notes:
| (1) | Excludes leases and financing obligations for property, plant and equipment. |
| (2) | This number includes 68,093 10% Senior Secured Notes due January 15, 2023, which are issued in denominations of $1,000. This number also includes 29 Warrant Notes which were not exchanged for 10% Senior Secured Notes due January 15, 2023. |
| (3) | The total funds available under this facility are EUR 1.5 million, which has been converted into U.S. Dollars for purposes of this prospectus. |
| (4) | The total funds available under this facility are approximately EUR 1.49 million which has been converted into U.S. Dollars for purposes of this prospectus. |
| (5) | Convertible into 910,000 Subordinate Voting Shares at a fixed conversion price per share of $2.65 after first anniversary. |
Senior Notes
On December 23, 2019, we announced the receipt of initial subscriptions totaling $27.5 million in connection with a debt financing (the Debt Financing). Insiders and certain Founders committed an aggregate of $18.5 million in connection with the Debt Financing. Investors were provided two financing structure options. The first structure allowed subscribers to acquire Warrant Notes maturing on January 15, 2023. The principal amount outstanding under such Warrant Notes bears interest at a rate of 10.0% per annum, payable in cash quarterly. Under the first structure subscribers were also issued Debt Warrants to acquire a number of Subordinate Voting Shares of the Company equal to 75% of the principal amount of the applicable Warrant Note divided by the exercise price of $1.5787. The Debt Warrants have an expiration date of December 23, 2024. The second structure allowed subscribers to acquire Original Issue Discount 10% Senior Secured Notes (OID Notes) maturing on January 15, 2023. The principal amount outstanding under such OID Notes bears interest at a rate of 10.0% per annum, payable in cash quarterly. The combined annual yield on the OID Notes is approximately 17%. Participants electing to receive OID Notes did not receive Debt Warrants.
In addition to the maturity dates, both structures have the same key terms. The Company’s obligations under both the Warrant Notes and the OID Notes are secured by the assets of the Company and certain of its subsidiaries (subject to certain exclusions) and are guaranteed by certain subsidiaries of the Company. Due to insider participation, a special committee of the board of directors, comprised entirely of independent members, was formed to set, review, negotiate and approve of the terms of the Debt Financing. ATB Capital Markets (formerly, AltaCorp Capital Inc.) was engaged by the special committee of the board of directors to provide its opinion as to the fairness, from a financial perspective, of the terms of the Debt Financing.
On January 31, 2020, the Company announced an upsizing of the Debt Financing, and was in receipt of aggregate cash proceeds of $35.7 million in subscriptions (inclusive of the $27.5 million cash proceeds already received by the Company). The Company also announced that $9.6 million of debt and interest expenses assumed by the Company pursuant to the legal settlement of a matter relating to former entity TGS National Holdings, LLC and its subsidiaries and the acquisition of BHILH and its suibsidiaries which were exchanged into Warrant Notes with a slightly modified redemption right (subject to an unrelated contingency).
41
On July 31, 2020, the Company announced a second upsizing to the previously announced Debt Financing and closed on $33.3 million of new subscription receipts, receiving $27.8 million in cash proceeds. Included in the $33.3 million was $12.3 million of subscription receipts contingent upon the closing of the PAMS acquisition. Due to founder and other insider participation, a special committee of the board of directors, comprised entirely of independent members, was formed to set, review, negotiate and approve of the terms of the Debt Financing. ATB Capital Markets (formerly, AltaCorp Capital Inc.) was engaged by the special committee of the board of directors to provide its opinion as to the fairness, from a financial perspective, of the terms of the upsizings to the Debt Financing.
On December 1, 2020, certain participants in the Debt Financing elected to exchange their Warrant Notes or OID Notes for an equal principal amount of 10% Senior Secured Notes due January 15, 2023, which commenced trading on the CSE under the trading symbol “JUSH.DB.U”. Holders of an aggregate principal amount of $76.4 million in Warrant Notes and OID Notes elected to participate in such exchange. Holders of an aggregate principal amount of $7.0 million in Warrant Notes and OID Notes elected to retain their Warrant Notes or OID Notes, as applicable, which were amended and restated in connection with the listing of the 10% Senior Secured Notes due January 15, 2023. On December 31, 2020, an aggregate of approximately $83.3 million of listed and unlisted notes remain outstanding.
Acquisition Facility
On October 20, 2021, the Company entered into definitive documentation in respect of a $100 million Senior Secured Credit Facility (the Acquisition Facility) with Roxbury, LP acting as agent to SunStream Bancorp Inc. (Sunstream), a joint venture sponsored by Sundial Growers Inc. (NASDAQ:SNDL). As of August 4, 2022, Jushi had drawn down $65.0 million from the Acquisition Facility to fund the cash portion of the completed acquisitions of Nature’s Remedy, Apothecarium and NuLeaf. Additionally, the Company will consider borrowing future amounts under the Acquisition Facility for potential strategic expansion opportunities in both its core and developing markets. After being drawn, loans issued under the Acquisition Facility will bear an interest rate of 9.5% per annum, payable quarterly, and will mature five years from the closing date. The Company will be able to make additional draws under the facility for an 18-month period, and will have a two-year interest-only period before partial amortization begins on a quarterly basis. The Company also may increase the total commitment of the Acquisition Facility by an aggregate amount of up to $25 million, subject to certain conditions. The Acquisition Facility is secured by a first lien over certain Company assets and on a pari passu basis with current senior indebtedness on existing assets that are collateralized under the Company’s current senior debt.
For additional information and information about our other debt instruments, refer to Note 11 - Debt in the audited consolidated financial statements and Note 9 - Debt in the unaudited interim condensed consolidated financial statements.
Critical Accounting Policies and Estimates
Critical Accounting Estimates
The preparation of the consolidated financial statements in conformity with US GAAP requires management to make judgments, estimates, and assumptions about the carrying amounts of assets and liabilities that are not readily apparent from other sources. The estimates and underlying assumptions are reviewed on an ongoing basis. The estimates and associated assumptions are based on historical experience and other factors that are considered to be relevant. Actual results may differ from these estimates and revisions to accounting estimates are recognized in the period in which the estimate is revised. Our significant accounting policies are described in Note 2 - Basis of Presentation and Summary of Significant Accounting Policies in the audited consolidated financial statements.
Significant judgments, estimates, and assumptions that have the most significant effect on the amounts recognized in the consolidated financial statements are described below.
42
Estimated Useful Lives and Depreciation of Property and Equipment and Intangible Assets
Depreciation and amortization of property and equipment and intangible assets are dependent upon estimates of useful lives, which are determined through the exercise of judgment. The assessment of any impairment of these assets is dependent upon estimates of recoverable amounts that take into account factors such as economic and market conditions and the useful lives of assets. Our licenses have indefinite lives and are not amortized. Absent such licenses, we cannot continue conducting business in the associated jurisdiction and as such, there is no foreseeable limit to the period over which these assets are expected to generate future cash inflows to us.
Accounting for Acquisitions and Business Combinations
In determining the fair value of all identifiable assets, liabilities and contingent liabilities acquired, the most significant estimates relate to contingent consideration and intangible assets. Management exercises judgement in estimating the probability and timing of when earn-outs are expected to be achieved, which is used as the basis for estimating fair value. For any intangible asset identified, depending on the type of intangible asset and the complexity of determining its fair value, management with the assistance of an independent valuation expert, may estimate the fair value, using appropriate valuation techniques, which are generally based on a forecast of the total expected future net cash flows.
Cannabis licenses are the primary intangible asset acquired in business combinations as they provide us the ability to operate in each market. We utilize the expected future net cash flows of the acquired business in calculating the fair value of these intangible assets. The key assumptions used in these cash flow projections include discount rates and terminal growth rates. Of the key assumptions used, the impact of the estimated fair value of the intangible assets have the greatest sensitivity to the estimated discount rate used in the valuation. Management selected discount rates are primarily dependent upon the markets in which each of the acquisitions operates. The terminal growth rate represents the rate at which these businesses will continue to grow in perpetuity. Other significant assumptions include estimated revenue, gross profit, operating expenses and anticipated capital expenditures which are based upon the business’ historical operations along with management projections.
The evaluations are linked closely to the assumptions made by management regarding the future performance of these assets and any changes in the discount rate applied.
Inventories
The net realizable value of inventories represents the estimated selling price for inventories in the ordinary course of business, less all estimated costs of completion and costs necessary to make the sale. The determination of net realizable value requires significant judgment, including consideration of factors such as shrinkage, the aging of and future demand for inventory, expected selling price and the contractual arrangements with customers. Reserves for excess and obsolete inventory are based upon quantities on hand, projected volumes from demand forecasts and net realizable value. The estimates are judgmental in nature and are made at a point in time, using available information, expected business plans and expected market conditions. As a result, the actual amount received on sale could differ from the estimated value of inventory. Periodic reviews are performed on the inventory balance. The impact of changes in inventory reserves is reflected in cost of goods sold.
Goodwill and Indefinite Lived Intangibles Impairment
Goodwill and indefinite lived intangibles are tested for impairment annually and whenever events or changes in circumstances indicate that the carrying amount of the asset has been impaired. In order to determine if the value of goodwill or indefinite lived intangible assets may have been impaired, we perform a qualitative assessment to determine if it is more-likely-than-not that the reporting unit’s carrying value is less than the fair value, indicating the potential for impairment, and therefore requiring a quantitative assessment. If we determine that a quantitative impairment test is required, for testing of goodwill, we typically use a combination of an income approach, i.e., a discounted cash flow calculation, and a market approach, i.e., using a market multiple method, to determine the fair value of each reporting unit, and then compare the fair value to its carrying amount to determine the amount of impairment, if any. For the testing of indefinite-lived intangibles, we typically utilize an income approach, i.e., the excess earnings approach. When applying these valuation techniques, we rely on a number of factors, including historical results, business plans, forecasts and market data. Changes in the conditions for these judgments and estimates can significantly affect the assessed value of goodwill and indefinite lived intangible assets.
43
Share-based Payment Arrangements
We account for stock based compensation expense in accordance with ASC 718 Compensation – Stock Compensation, which requires the measurement and recognition of stock-based compensation expense based on estimated fair values, for all stock-based payment awards made to employees. We use the Black-Scholes pricing model to determine the fair value of stock options and warrants granted to employees and directors under share-based payment arrangements, where appropriate. In estimating fair value, management is required to make certain assumptions and estimates such as the expected life of the awards, volatility of share price, risk free rates, and future dividend yields at the initial grant date. Changes in assumptions used to estimate fair value could result in materially different results.
Leases
ASC 842 Leases (ASC 842) requires lessees to recognize Right of Use (ROU) assets and lease liabilities on the balance sheet. ROU assets and lease liabilities are recognized at the commencement date based on the present value of lease payments over the lease term, using a discount rate equivalent to our incremental borrowing rate for a term similar to the estimated duration of the lease. ASC 842 requires lessees to discount lease payments using the rate implicit in the lease if that rate is readily available. If that rate cannot be readily determined, the lessee is required to use its incremental borrowing rate. Accordingly, we generally use our incremental borrowing rate when initially recording leases. We determine the incremental borrowing rate as the interest rate we would pay to borrow over a similar term the funds necessary to obtain an asset of a similar value to the right-of-use asset in a similar economic environment. In addition, ASC 842 requires lessees to estimate the lease term. In determining the period which we have the right to use an underlying asset, management considers the non-cancellable period along with all facts and circumstances that create an economic incentive to exercise an extension option, or not to exercise a termination option.
Income Taxes
Current tax assets and/or liabilities comprise those claims from, or obligations to, fiscal authorities relating to the current or prior reporting periods that are unpaid at the reporting date. Current tax is payable on taxable profit, which differs from profit or loss in the financial statements. Calculation of current tax is based on tax rates and tax laws that have been enacted by the end of the reporting period.
Income taxes are accounted for under the asset and liability method, which requires the recognition of deferred tax assets and liabilities for the expected future tax consequences of events that have been included in the financial statements. Under this method, deferred tax assets and liabilities are determined on the basis of the differences between the financial statement and tax basis of assets and liabilities by using enacted tax rates in effect for the year in which the differences are expected to reverse. The effect of a change in tax rates on deferred tax assets and liabilities is recognized in income in the period that includes the enactment date.
Deferred tax assets are recognized to the extent that we believe that these assets are more likely than not to be realized. In making such a determination, all available positive and negative evidence are considered, including future reversals of existing taxable temporary differences, projected future taxable income, tax-planning strategies, and results of recent operations. If it is determined that we would be able to realize deferred tax assets in the future in excess of their net recorded amount, an adjustment to the deferred tax asset valuation allowance is recorded, which would reduce the provision for income taxes
Uncertain tax positions are recorded in accordance with ASC 740 Income Taxes on the basis of a two-step process in which (1) we determine whether it is more likely than not that the tax positions will be sustained on the basis of the technical merits of the position and (2) for those tax positions that meet the more-likely-than-not recognition threshold, we recognize the largest amount of tax benefit that is more than 50 percent likely to be realized upon ultimate settlement with the related tax authority.
As we operate in the legal cannabis industry, we are subject to the limits of Section 280E of the Code for U.S. federal income tax purposes as well as state income tax purposes for all states except for California and Colorado. Under Section 280E of the Code, we are only allowed to deduct expenses directly related to the cost of goods sold. This results in permanent differences between ordinary and necessary business expenses deemed non-allowable under Section 280E of the Code.
44
Recently Adopted Accounting Standards and Recently Issued Accounting Pronouncements
For information about our recently adopted accounting standards and recently issued accounting standards not yet adopted, refer to Note 2 - Basis of Presentation and Summary of Significant Accounting Policies of the audited consolidated financial statements.
Changes in Accountants
On June 2, 2021, we replaced MNP LLP as our independent accountants.
The reports of MNP LLP on our consolidated financial statements for the years ended December 31, 2020 and 2019, which were prepared in accordance with International Financial Reporting Standards, did not contain any adverse opinion or disclaimer of opinion, nor were such reports qualified or modified as to uncertainty, audit scope, or accounting principles. MNP LLP conducted their audits in accordance with Canadian generally accepted auditing standards.
During the years ended December 31, 2020 and 2019 and the subsequent interim period through June 2, 2021, MNP LLP did not have any disagreement with us on any matter of accounting principles or practices, financial statement disclosure or auditing scope or procedure, which disagreement, if not resolved to the satisfaction of MNP LLP would have caused it to make reference to the subject matter of the disagreement in connection with its report on our consolidated financial statements.
During the years ended December 31, 2020 and 2019 and the subsequent interim period through June 2, 2021, there were no “reportable events” as such term is defined in Item 304(a)(1)(v) of Regulation S-K.
We provided a copy of this disclosure to MNP LLP and requested that they furnish us a letter addressed to the SEC stating whether they agree with the above statements. Their letter to the SEC will be attached as an exhibit to the registration statement of which this prospectus is a part.
On June 2, 2021, we engaged Marcum LLP as our independent registered public accounting firm. Marcum LLP conducted the audits of the Company’s financial statements for the fiscal years ended December 31, 2021 and 2020, which were prepared in accordance with US GAAP and appear in this prospectus.
45
QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
We are exposed to market risks in the ordinary course of business. Some potential market risks are discussed below:
Market Risk
Strategic and operational risks arise if we fail to carry out business operations and/or raise sufficient equity and/or debt financing. These strategic opportunities or threats arise from a range of factors that might include changing economic and political circumstances and regulatory approvals and competitor actions. The risk is mitigated by consideration of other potential development opportunities and challenges which management may undertake.
Currency Risk
Our operating results and financial position are reported in U.S. dollars. Some of our financial transactions are denominated in currencies other than the U.S. dollar. The results of operations are subject to currency transaction risks.
We have no hedging agreements in place with respect to foreign exchange rates. We have not entered into any agreements or purchased any instruments to hedge possible currency risks at this time.
Credit Risk
Management does not believe that the Company currently has credit risk related to its operations, as the Company’s revenue is generated primarily through cash transactions. Concentrations of credit risk with respect to our cash and cash equivalents are limited primarily to amounts held with financial institutions. Credit risk related to operations could increase as the Company continues to expand wholesale operations.
Price Risk
Price risk is the risk of variability in the fair value due to movements in equity or market prices. Our investments, primarily mutual funds, are susceptible to price risk arising from uncertainties about their future outlook, future values and the impact of market conditions.
Interest Rate Risk
Interest rate risk is the risk that the fair value or the future cash flows of a financial instrument will fluctuate as a result of changes in market interest rates. Cash equivalents bear interest at market rates. The majority of our financial debts have fixed rates of interest and therefore our exposure is limited.
Inflation Risk
If our costs become subject to significant inflationary pressures, we may not be able to fully offset such higher costs through price increases. Our inability or failure to do so could harm our business, financial condition, and operating results.
Asset Forfeiture risk
Because the cannabis industry remains illegal under U.S. federal law, any property owned by participants in the cannabis industry which are either used in the course of conducting such business, or are the proceeds of such business, could be subject to seizure by law enforcement and subsequent civil asset forfeiture. Even if the owner of the property were never charged with a crime, the property in question could still be seized and subject to an administrative proceeding by which, with minimal due process, it could be subject to forfeiture.
Banking Risk
Notwithstanding that a majority of states have legalized medical marijuana, there has been no change in U.S. federal banking laws related to the deposit and holding of funds derived from activities related to the marijuana industry. Given that U.S. federal law provides that the production and possession of cannabis is illegal, there is a strong argument that banks cannot accept for deposit funds from businesses involved with the marijuana industry. Consequently, businesses involved in the marijuana industry often have difficulty accessing the U.S. banking system and traditional financing sources. The inability to open bank accounts with certain institutions may make it difficult to operate the businesses of Jushi, its subsidiaries and investee companies, and leaves their cash holdings vulnerable. We have banking relationships in all jurisdictions in which we operate.
46
Overview
We are a vertically integrated, multi-state cannabis operator engaged in retail, distribution, cultivation, and processing operations in both medical and adult-use markets. We are focused on building a diverse portfolio of cannabis assets through opportunistic investments and pursuing application opportunities in attractive limited license markets. We have targeted assets in highly populated, limited license medical markets that are on a trajectory toward adult-use legalization, including Pennsylvania and Ohio, markets that are in the process of transitioning to adult-use, namely Virginia, and limited license, fast-growing, large adult-use markets, such as Illinois, Nevada and Massachusetts, and certain municipalities of California.
Business Strategy
Our business strategy is to find market opportunities with favorable relevant local competitive and regulatory landscapes, supply/demand dynamics, and growth potential. We evaluate the economic viability of each opportunity before making capital allocation decisions and may decide to participate in one or more facets of the supply chain based on the dynamics of each individual market. In certain markets, we may seek to apply a capital-light or retail-focused strategy, especially where cultivation may become further commoditized in future years. In limited license medical markets (e.g., Pennsylvania), or markets in the process of transitioning to adult-use (e.g., Virginia), we may seek to expand our cultivation assets despite the high level of capital investment required, given the significant market opportunity. Also, in other markets, we may seek a more balanced capital allocation approach where we may acquire a grower-processor and/or additional retail dispensaries in a market where we currently operate, such as Illinois, Ohio, California, Massachusetts, and Nevada. Lastly, in limited license adult-use cannabis markets that are expanding, we may allocate significant capital to acquire a vertically integrated operator. By establishing a strong platform and retail-brand recognition in markets that have the greatest growth potential, we expect to be well-positioned for future growth in adult-use cannabis once it is further legalized.
Growth Strategy
We remain intensely focused on expanding our retail presence in current markets while pursuing acquisition opportunities across the supply chain in limited license markets that complement our existing portfolio. Our financial capacity allows us to operate from a position of strength and it is expected that such financial capacity will help us emerge as an even stronger player in this industry. We plan to implement our growth strategy by expanding our presence in current markets, increasing our offering of branded product lines, targeting acquisition opportunities across the supply chain, and applying for de novo licenses.
Current Operations
Retail
We operate eighteen medical cannabis dispensaries, under our BEYOND/HELLO™ brand, in the Commonwealth of Pennsylvania.
We operate four adult-use (with two co-located medical) cannabis dispensaries, under our BEYOND/HELLO™ brand, in the State of Illinois. Additionally, our partner was awarded a conditional retail dispensary license in Illinois via the state’s lottery process.
We operate two medical cannabis dispensaries, under our BEYOND/HELLO™ brand, in the Commonwealth of Virginia. We are permitted to open four additional dispensaries in the Commonwealth of Virginia, subject to local zoning and state regulatory approvals.
We operate two adult-use cannabis dispensaries, under our Nature’s Remedy™ brand, in the Commonwealth of Massachusetts (with one co-located medical).
47
We operate four adult-use and medical retail dispensaries in Nevada in the Las Vegas and Lake Tahoe areas.
In Ohio, our 100% owned subsidiary, Campbell Hill Ventures, was awarded a provisional medical marijuana dispensary license in the medical marijuana retail lottery and is awaiting certification of licenses by the Ohio Board of Pharmacy.
We operate three medical and adult-use cannabis dispensaries, under our BEYOND/HELLO™ brand, in the State of California located in the City of Santa Barbara, the City of Palm Springs and the City of Grover Beach. Our Palm Springs dispensary’s operations, which were voluntarily suspended while the store underwent a significant renovation, resumed operations in the third quarter of 2022.
Online Platforms
We operate three age-gated online platforms through www.beyond-hello.com, www.naturesremedyma.com, and www.nuleafnv.com for patients and customers (the Online Platforms). The Online Platforms are not intended to be used for advertising activities but are intended to be used as a virtual tool, allowing patients and customers to understand the cannabis products that we offer and view real-time pricing and product availability at our dispensaries. The Online Platforms do not provide any information or any other functionalities with respect to any third-party dispensaries.
No cannabis purchase and sale transactions occur on the Online Platforms. A patient or customer may reserve products using the Online Platforms, but the patient or customer must be physically present at the point-of-sale to consummate the purchase and sale of products. This requirement allows us and dispensary staff to ensure that the standard operating procedures (including our compliance program(s)) are applied to all patients and customers in connection with the purchase and sale of products.
In jurisdictions where medical cannabis is legal, upon arrival of the patient at the applicable dispensary, or at the point of delivery (where permissible), dispensary staff must verify the patient’s identity and accreditation (such as a state-issued medical cannabis card) and confirm the patient’s allotment to ensure the user is not exceeding the state’s allotment limits. Once the foregoing is verified, the patient may pay for the product(s) to complete the purchase. If the patient does not have valid identification and accreditation, the patient will not be able to purchase medical cannabis at our applicable dispensary, or at the point of delivery (where permissible), irrespective of any reservation(s) made on one of our Online Platforms.
In jurisdictions where recreational cannabis is legal, upon arrival of the customer at the applicable dispensary, or at the point of delivery (where permissible), dispensary staff must verify that the customer is at least 21 years of age by verifying the customer’s government-issued identification. Once the identification is verified, the customer may pay for the product(s) to complete the transaction. If the customer does not have valid identification, the customer will not be able to purchase recreational cannabis at our applicable dispensary, or at the point of delivery (where permissible), irrespective of any reservation(s) made on one of our Online Platforms.
As of May 3, 2022, we no longer sell our “Nira” branded products through our CBD e-commerce platform (www.niracbd.com).
Cultivation & Production
We are currently engaged in cannabis cultivation and production operations in Pennsylvania, at our grower-processor facility operated by PAMS. On November 23, 2020, we announced our intention to launch a phased expansion of the PAMS facility to better serve the growing medical cannabis market in the Commonwealth of Pennsylvania. The first phase of the expansion, which was completed in the second quarter of 2022, expanded the facility from 81,000 sq. ft. to 123,000 sq. ft., increased total canopy from approximately 16,000 sq. ft. to approximately 35,000 sq. ft., and increased annual biomass capacity from about 8,000 lbs. to approximately 22,000 lbs. Phase two of the expansion, which would commence pending favorable regulatory developments in the Commonwealth of Pennsylvania, such as adult-use legislation, is expected to increase the PAMS facility from 123,000 sq. ft. to 210,000 sq. ft., increase total canopy from 35,000 sq. ft. to approximately 107,000 sq. ft., and increase annual biomass capacity from about 22,000 lbs. to approximately 60,000 lbs. In addition to these two contemplated phases of buildout, PAMS continues to assess and develop further expansion opportunities at the PAMS facility to meet the needs of patients and wholesale market demand, now and in the future.
48
We are also engaged in cannabis cultivation and processing in Virginia. Through our subsidiary Dalitso, we operate the Manassas Facility, which allows us to cultivate, process, dispense and deliver medical cannabis to registered patients in Virginia. The Manassas Facility is approximately 93,000 sq. ft.
In May 2021, we began phase one of the expansion of the existing Manassas Facility, which added approximately 63,000 sq. ft. of cultivation, manufacturing and processing capacity and was completed in the second quarter of 2022. At full capacity, the facility will have approximately 19,000 sq. ft. of canopy and an annual biomass production capacity of approximately 12,000 lbs. that should begin to come to market by the third quarter 2022. We are also in the design phase of constructing a second connected on-site building that would also be built in two phases (phase two and phase three), pending regulatory developments. Phase two of the second building is expected to increase the facility from 93,000 sq. ft. to 195,000 sq. ft., increase total canopy from about 19,000 sq. ft. to approximately 54,000 sq. ft., and increase annual biomass production from about 12,000 lbs. to approximately 35,000 lbs. We anticipate commencing phase two of the expansion when there is clear line of sight into the timing of the state’s regulatory developments surrounding the beginning of an adult-use sales program. Phase three would add another approximately 68,000 sq. ft. to the facility, 69,000 sq. ft. of canopy, and 45,000 lbs. of annual biomass production for a total of approximately 263,000 sq. ft., 123,000 sq. ft. of canopy, and 80,000 lbs. of annual biomass capacity. Dalitso’s planned buildout of their facility, enables Dalitso to efficiently produce a consistent supply of medical cannabis products as patient access improves and the medical cannabis program continues to mature and expand.
In September 2021, we closed on our acquisition of Nature’s Remedy. Nature’s Remedy currently operates a 50,000 sq. ft. cultivation and production facility in Lakeville, Massachusetts with high-quality, indoor flower canopy and state-of-the-art extraction and manufacturing capabilities (the Lakeville Facility). The Lakeville Facility recently completed an expansion to approximately 33,000 sq. ft. of canopy during the second half of 2021. As part of the expansion, Nature’s Remedy expects to increase annual biomass production to approximately 21,000 lbs. The Lakeville Facility could potentially accommodate an additional 18,000 to 20,000 sq. ft. of flower canopy through the expansion into approximately 26,000 sq. ft. of adjacent space in the existing building. Nature’s Remedy is also evaluating further expansion opportunities in the existing Lakeville industrial complex and/or land owned by Nature’s Remedy in Grafton, Massachusetts. These expansions are subject to business evaluations and needs, and receipt of applicable regulatory approvals.
Further, in Nevada, through our subsidiary FBS NV, we engage in cultivation and production operations in North Las Vegas, Nevada, and are permitted to purchase and/or sell cannabis and cannabis products to other authorized licensees on a wholesale basis. Pursuant to the hemp handler license, FBS NV is also permitted to handle raw industrial hemp, purchase hemp-derived constituents (such as hemp-derived CBD) from licensed hemp operators, and to infuse or manufacture products containing hemp-derived constituents. FBS NV operates a 7,200 sq. ft. facility featuring a state-of-the-art, indoor, double-stacked cultivation that yields approximately 2,800 lbs. of high-quality dry flower per year. FBS NV plans to create a single production space for a total of approximately 17,400 sq. ft., which is expected to add approximately 5,300 sq. ft. of canopy to the facility’s cultivation capacity.
On April 6, 2022, we expanded our capabilities in Nevada by acquiring NuLeaf, a Nevada-based vertically integrated business operating a 27,000 sq. ft. cultivation facility in Sparks, Nevada, and a 13,000 sq. ft. processing facility in Reno. NuLeaf’s cultivation operations span 27,000 sq. ft. over two floors in a 15,000 sq. ft. building in Sparks, Nevada. The building is equipped with ten single-stacked flower rooms, with a flower canopy encompassing approximately 6,800 sq. ft., and a custom automation system for controlled temperature, humidity, lighting, and CO2 enrichment. NuLeaf also operates a separate 13,000 sq. ft. processing facility in Reno, Nevada. The facility currently has hydrocarbon extraction capabilities and can hold an additional extraction unit within the existing space.
In August 2021, we completed the acquisition of FBS OH, a licensed medical marijuana processor in Ohio. FBS OH operates an 8,000 sq. ft. state-of-the-art processing facility located in Columbus, Ohio. Additionally, we have acquired Ohio Green Grow LLC and OhiGrow located in Toledo, Ohio. OhiGrow holds a Level II cultivation license that allows for an initial 3,000 sq. ft. of cultivation area. OhiGrow currently operates approximately 1,900 sq. ft. of canopy in a free-standing building. There is additional available vacant space on the property, which can be further developed. We intend to apply for the necessary approvals to expand the OhiGrow facility’s cultivation area to the maximum currently permitted under the Level II cultivation license.
49
Product Selection and Offerings
With respect to our cannabis business, senior leaders from the business development, operations, finance, marketing, and sales teams negotiate with potential brand vendors across all product categories including flower, vape pens, oils, extracts, edibles and pre-rolls to make future product selection decisions. Leveraging managements’ experience, we analyze market dynamics, product quality, profit and loss, impact and consumer demand to carry out our long-term strategy in each market. With high-impact retail locations in key markets, we expect to be a desirable partner for nationally scaling brands and/or in-house products.
In Pennsylvania, Virginia, Massachusetts, and Nevada, our dispensaries sell our own in-house brands and a variety of third-party cannabis products, including, cannabis dry flower, vaporizer forms of cannabis, edibles (where permissible), cannabis oil in capsule, tinctures, cannabis in topical products, and other cannabis products. In Illinois and California, our dispensaries sell a variety of third-party cannabis products, including cannabis dry flower, vaporizer forms of cannabis, cannabis oil in capsule, tinctures, cannabis in topical products, cannabis edible products and other cannabis products. In Ohio, we wholesale a series of brands and products in the state, which are currently available for purchase at partner dispensaries across Ohio.
In connection with such cultivation and processing, we intend to utilize intellectual property, including trademarks, trade secrets, extraction techniques, concentrates and other proprietary information related to our cannabis brands within the states such cannabis brands are marketed and sold. We own the domains jushico.com, beyond-hello.com, thebankflower.com, findthelab.com, thetasteology.com, niraplus.com, secheflower.com, niracbd.com, and several related domains. We have received state approval to produce and market these products under such brands where applicable. We will pursue trademark registration of material brands with the U.S. Patent and Trade Office promptly following a change in the U.S. Patent and Trade Office's policy position towards cannabis products, which currently does not accept such registrations, or a change in related laws. We rely on non-disclosure and confidentiality agreements to protect our intellectual property rights. These brands and formulations include:
Premium Flower: The Bank
Jushi relaunched its acquired, award-winning Colorado brand, The Bank, known for its superior plant genetics and next-level cultivation. The Bank offers pre-packaged flower, infused blunts and pre-rolls comprised of three tiered lines: Gold Standard, Cache and Vault each offering varying degrees of quality, availability and price. Currently, The Bank is available at our BEYOND/HELLO™ locations in Pennsylvania and Virginia, our NuLeaf and Nature’s Remedy branded dispensaries in Nevada and Massachusetts, respectively, and other licensed retailers across these markets.
Vapes & Concentrates: The Lab
We relaunched another of our acquired, award-winning Colorado brands, The Lab, renowned for high-quality, precision vape products, and concentrates, including the pioneering of live resin. The Lab offers a wide selection of vape cartridges, disposables and concentrates. The Lab products are available at dispensaries across Pennsylvania, Virginia, Massachusetts, Nevada, and Ohio, including our BEYOND/HELLO™ locations in Pennsylvania and Virginia, our NuLeaf stores in Nevada, and our Nature’s Remedy locations in Massachusetts.
Within The Lab brand family, we launched our first line of solventless live resin extracts, the new top-shelf product line, produced purely from premium flower and extracted simply with ice and water, includes a 0.5g vape extract cartridge available now and 1g jarred concentrates coming soon for purchase exclusively at BEYOND/HELLO™ store locations in Pennsylvania under the name, The Lab™ Solventless Live RSN. It is also expected to launch at our Nature’s Remedy locations in Massachusetts, our NuLeaf stores in Nevada, and our BEYOND/HELLO™ stores in Virginia, pending regulatory approval.
Also, within The Lab brand family, we debuted our first line of concentrates made using hydrocarbon extraction. The Lab™ Live Resin is the second of several single-source concentrate product lines to be launched. Initially, we will exclusively carry The Lab™ Live Resin 500mg full-spectrum 0.5 gram 510 cartridges at BEYOND/HELLO™ retail locations in Pennsylvania. We plan to roll out our hydrocarbon-extracted line at partner dispensaries across the Commonwealth of Pennsylvania in the coming months, as well as in additional states such as Massachusetts, Virginia and Nevada. We also plan to launch a 300mg rechargeable, all-in-one 0.3g vape and a variety of 1 gram jarred cured concentrates in the coming months, pending regulatory approval.
Edibles: Tasteology
Jushi launched Tasteology, an edible brand offering premium, real fruit, cannabis-infused gummies and chewable tablets. Tasteology is the culmination of extensive consumer research into both the taste and effect preferences of people in Jushi’s markets where edibles can be offered. Currently, Tasteology is available at dispensaries in Nevada and Ohio as well as BEYOND/HELLO™ dispensaries in Virginia. Tasteology products are expected to launch in Massachusetts in the third quarter of 2022, pending regulatory approval.
Medicinal: Nira + Medicinals
Nira + Medicinals (Nira+) develops high-quality, THC and CBD-rich medical products aimed at improving the quality of life for all cannabis patients. Nira+ product line includes tinctures, capsules, softgels and topicals. Currently, Nira+ is available at dispensaries across Pennsylvania, including our BEYOND/HELLO™ locations in Pennsylvania, and Nature’s Remedy retail locations and partner dispensaries in Massachusetts. All our CBD products are derived from hemp containing no more than 0.3 percent THC.
50
Fine Grind (Shake), Fine Flower (Popcorn) and Singles (Pre-Rolls): Sèchè
Sèchè is a new category in cannabis that redefines the perception of value products like shake and popcorn. Sèchè offers products like Fine Grind (Shake), Fine Flower (Popcorn) and Singles (Pre-Rolls). Currently, Sèchè is available at dispensaries across Virginia, Nevada, Ohio, Massachusetts, and Pennsylvania, including at the Company’s BEYOND/HELLO™ locations in Virginia and Pennsylvania, our NuLeaf stores in Nevada, as well as Nature’s Remedy stores in Massachusetts.
CBD: Nira
As of May 3, 2022, we no longer sell “Nira” branded CBD products through our online store (www.niracbd.com).
In addition to branded and manufactured finished products, we plan to sell bulk refined cannabinoids and terpenes to vendors for use in their own finished products, as our production capacity increases in certain markets. The full scale and allocation of production utilization will depend upon the scale of our owned and managed retail footprint in addition to the production capacity of our cultivation and production facilities.
Branding and Marketing
We continue the rollout of our flagship retail brand BEYOND/HELLO™ across our key operating markets. After the launch of our online pre-ordering platform, BEYOND/HELLO™ has evolved into a fully integrated digital to brick-and-mortar experience, providing customers real-time access to pricing and product availability. All of our current retail locations operate under the BEYOND/HELLO™ brand except in Massachusetts and Nevada. In Massachusetts our retail locations operate under the Nature’s Remedy brand, and in Nevada, we operate three retail locations under the NuLeaf brand and one under the Apothecarium brand. We are planning for our future retail locations in Virginia, California, and other states to operate under the BEYOND/HELLO™ brand as well.
We operate a state-by-state opt-in loyalty program, “The Hello Club,” that rewards patients and customers with points and other exclusive offers based on their past purchases. We leverage SMS and email lists to promote specific products.
We provide retail partners with approved merchandise, and other display materials to support sales. We create product imagery, video and descriptions which are included across online dispensary menus where our products are sold.
We take advantage of various directory platforms for cannabis businesses to help prospective patients and customers find our respective retail locations. We also run out-of-home marketing campaigns in approved markets and locations for our retail dispensaries.
Sales and Distribution
With respect to cannabis retail locations, we target highly visible locations adjacent or near heavily trafficked roads. For cultivation, production and other forms of industrial activity, we target locations with immediate capabilities as well as future expansion potential. We use an internal team for the selection of real estate, as well as a broad network of real estate brokers. We make the determination to purchase or lease our underlying real estate on a case-by-case basis.
We plan to expand our network of cannabis retail locations in select markets. We have developed key indicators to identify attractive sites based on existing competition, population, real estate, parking, traffic and regulatory market attractiveness. We intend to educate patients and consumers about our product offerings in a welcoming environment through one-on-one interactions with staff.
51
Principal Markets & Competition
We compete against other retail and vertical licensees across the various state markets that we operate in. In certain markets, such as California, many of our competitors are small local dispensaries; however, we expect to compete against both large Multi-State Operators (MSO), as well as Canadian licensed producers, if and when cannabis is federally legal in the U.S. A "Multi-State Operator" is a colloquial term used to describe a company that engages in the cultivation, production and/or sale of cannabis and cannabis products in more than one state in accordance with applicable state and local laws, rules and regulations. In addition, we expect to compete against both third party and direct delivery services. We seek to address our competitive risk in these markets by picking strategic locations, with defensible buffers naturally built in through local regulations and local dispensaries laws.
With respect to cultivation and production, we compete with both MSO's and local operators in the states in which we operate. In Pennsylvania, Virginia, Ohio, Massachusetts, and Nevada, we compete with larger MSO's that may have access to public markets, more experienced management teams, or are further along in terms of reaching scale. We are positioning ourselves to minimize all of the above risks through accretive acquisitions, superior execution, and thoughtful location of retail and manufacturing sites.
Business in Europe
We hold a 51% interest in Jushi Europe. Jushi Europe’s wholly owned Portuguese subsidiary, JPTREH Unipessoal Limitada, a business entity organized under the laws of Portugal (Jushi Portugal), submitted an application to Portugal’s National Authority for Medicines and Health Products (INFARMED) for import, cultivation and export of medical cannabis. Jushi Portugal was granted a pre-license on November 9, 2020. Jushi Portugal acquired 32 acres of land to construct a greenhouse cultivation facility in southern Portugal. The build-out of the greenhouse cultivation facility commenced, but was subsequently halted.
On February 22, 2022, Jushi Europe filed a notice of over-indebtedness with the Swiss courts. Then on March 14, 2022, the auditor for Jushi Europe also filed a notice of over-indebtedness with the Swiss courts. Both notices are pending with the Swiss courts. As a result of the impending bankruptcy of Jushi Europe, we determined that the assets of Jushi Europe were impaired and recognized an impairment loss of $4.6 million for the year ended December 31, 2021, which is included in operating expenses in the audited consolidated statements of operation and comprehensive income (loss).
Neither Jushi Europe nor Jushi Portugal operates nor plans to operate in any emerging markets (as defined by OSC Staff Notice 51-720 – Issuer Guide for Companies Operating in Emerging Markets).
52
Organizational Structure
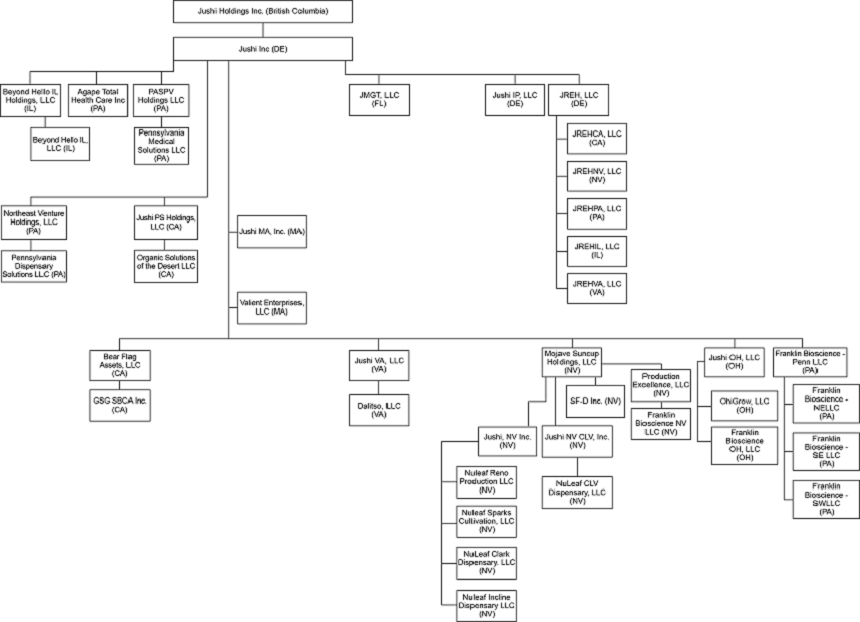
The organizational chart of Jushi and our material subsidiaries, including the percentage of voting securities of each material subsidiary held by us, either directly or indirectly, and their respective jurisdictions of incorporation, continuance, formation or organization as at the date of this prospectus, is set forth below. Unless otherwise noted in the box containing the name of the applicable subsidiary, the parent of such subsidiary owns 100% of the outstanding securities of such subsidiary.
Regulatory Overview
Below is a discussion of the federal and state-level U.S. regulatory regimes in those jurisdictions where we are currently directly involved, through our subsidiaries, in the cannabis industry.
53
Federal Regulation of Cannabis in the U.S.
Under U.S. federal law, marijuana is classified as a Schedule I drug. The CSA has five different tiers or schedules. A Schedule I drug means the Drug Enforcement Agency considers it to have a high potential for abuse, no accepted medical treatment, and lack of accepted safety for the use of it even under medical supervision. Other Schedule I drugs include heroin, LSD and ecstasy. In June 2018, the U.S. Food and Drug Administration (the FDA) approved Epidiolex, a purified form of CBD derived from the marijuana plant and used to treat two rare, intractable forms of epilepsy. We believe marijuana’s categorization as a Schedule I drug is thus not reflective of the medicinal properties of marijuana or the public perception thereof, and numerous studies show cannabis is not able to be abused in the same way as other Schedule I drugs, has medicinal properties, and can be safely administered. In this respect, 37 states, the District of Columbia, Guam, Puerto Rico and the U.S. Virgin Islands have passed laws authorizing comprehensive, publicly available medical marijuana programs, and 19 of those states and the District of Columbia have passed laws legalizing marijuana for adult-use.
In an effort to address incongruities between marijuana prohibition under the CSA and legalization under various state laws, the federal government issued guidance to law enforcement agencies and financial institutions during the Presidency of Barack Obama through Department of Justice (DOJ) memoranda. The most recent such memorandum is a DOJ memorandum issued by Deputy Attorney General James Cole in 2013 (the Cole Memo). The Cole Memo provided guidance to federal enforcement agencies as to how they should prioritize civil enforcement, criminal investigations and prosecutions regarding marijuana in all states. The Cole Memo shielded individuals and businesses participating in state-legal marijuana operations from prosecution under federal drug laws, excepting marijuana-related conduct that fell into one of the following enumerated prosecution priorities:
| 1. | Preventing the distribution of marijuana to minors; |
| 2. | Preventing revenue from the sale of marijuana from going to criminal enterprises, gangs and cartels; |
| 3. | Preventing the diversion of marijuana from states where it is legal under state law in some form to other states; |
| 4. | Preventing the state-authorized marijuana activity from being used as a cover or pretext for the trafficking of other illegal drugs or other illegal activity; |
| 5. | Preventing the violence and the use of firearms in the cultivation and distribution of marijuana; |
| 6. | Preventing the drugged driving and the exacerbation of other adverse public health consequences associated with marijuana use; |
| 7. | Preventing the growing of marijuana on public lands and the attendant public safety and environmental dangers posed by marijuana production on public lands; and |
| 8. | Preventing marijuana possession or use on federal property. |
On January 4, 2018, then U.S. Attorney General Jeff Sessions issued the Sessions Memo, which rescinded the Cole Memo. Rather than provide nationwide guidance respecting marijuana-related crimes in jurisdictions where certain marijuana activity was legal under state law, the Sessions Memo instructs that “[i]n deciding which marijuana activities to prosecute. With the DOJ’s finite resources, prosecutors should follow the well-established principles that govern all federal prosecutions.” Namely, these include the seriousness of the offense, history of criminal activity, deterrent effect of prosecution, the interests of victims, and other principles. Former U.S. Attorney General Jeff Sessions resigned in November 2018 and was replaced by Matthew Whitaker as interim Attorney General. In February 2019, William Barr was sworn in as Attorney General. Mr. Barr resigned as Attorney General on December 23, 2020 and Merrick Garland was sworn in as Attorney General in March 2021. Attorney General Merrick Garland’s public comments to date suggest that the prosecution priorities outlined in the Cole Memo shape the Department of Justice’s prosecutorial priorities under his tenure.
Despite rescission of the Cole Memo, we remain mindful of the common-sense prosecution priorities set forth therein and have not modified policies or procedures intended to support its underlying safety-focused intent. To this end, we and our operating subsidiaries adhere to industry best practices for operations, mandate strict compliance with applicable state and local laws, rules, regulations, ordinances, guidance and like authority, implement procedures designed to ensure operations do not exceed what is authorized under applicable licenses, perform stringent diligence on third-parties with whom we do business, perform background checks on employees, and maintain state-of-the-art seed-to-sale inventory tracking and other security infrastructure. Regular reviews of the foregoing and related operations, premises, documentation and the like are performed to ensure compliance with our safety, security and compliance standards.
54
Due to the CSA categorization of marijuana as a Schedule I drug, U.S. federal law makes it illegal for financial institutions that depend on the Federal Reserve’s money transfer system to take any proceeds from marijuana sales as deposits. Banks and other financial institutions could be prosecuted and possibly convicted of money laundering for providing services to cannabis businesses under the Bank Secrecy Act. Under U.S. federal law, banks or other financial institutions that provide a cannabis business with a checking account, debit or credit card, small business loan, or any other service could be found guilty of money laundering or conspiracy.
While there has been no change in U.S. federal banking laws to account for the trend towards legalizing medical and adult-use marijuana by U.S. states, FinCEN has issued guidance in 2014 to prosecutors handling money laundering and other financial crimes advising them not to focus enforcement efforts on banks and other financial institutions servicing marijuana-related businesses so long as such businesses are legally operating under state law and not engaging in conduct within the scope of a Cole Memo prosecution priority (such as keeping marijuana away from minors and out of the hands of organized crime). The 2014 FinCEN guidance also clarifies how financial institutions can provide services to marijuana-related businesses consistent with their Bank Secrecy Act obligations, including thorough customer due diligence, but makes it clear that they are doing so at their own risk. The customer due diligence steps include:
| 1. | Verifying with the appropriate state authorities whether the business is duly licensed and registered; |
| 2. | Reviewing the license application (and related documentation) submitted by the business for obtaining a state license to operate its marijuana-related business; |
| 3. | Requesting from state licensing and enforcement authorities available information about the business and related parties; |
| 4. | Developing an understanding of the normal and expected activity for the business, including the types of products to be sold and the type of customers to be served (e.g., medical versus adult-use customers); |
| 5. | Ongoing monitoring of publicly available sources for adverse information about the business and related parties; |
| 6. | Ongoing monitoring for suspicious activity, including for any of the red flags described in this guidance; and |
| 7. | Refreshing information obtained as part of customer due diligence on a periodic basis and commensurate with the risk. |
With respect to information regarding state licensure obtained in connection with such customer due diligence, the 2014 FinCEN guidance allows financial institutions to reasonably rely on the accuracy of information provided by state licensing authorities where states make such information available.
Unlike the Cole Memo, 2014 FinCEN guidance remains effective as of the date of this prospectus. During the Trump Administration, Secretary of the Treasury Steven Mnuchin publicly voiced his intent to leave such guidance in force and effect. The current Secretary of the Treasury, Janet Yellen, has not provided any public comment regarding her positions on the 2014 FinCEN guidance, but has previously indicated that she would be in favor of legislation that would provide safe harbor to financial institutions that worked with state-legal marijuana-related businesses. Nonetheless, despite FinCEN’s guidance, most banks and other financial institutions are still unwilling to provide banking or other financial services to marijuana businesses resulting in largely cash-based operations. While the FinCEN guidance decreased some risk for banks and financial institutions that accept marijuana business, it has not increased the industry’s access to banking services because financial institutions are required to perform extensive, continuous customer diligence respecting marijuana customers and are not immune from prosecution based on transacting business with such customers. In fact, some banks that had been servicing marijuana businesses have been closing the marijuana businesses’ accounts and are now refusing to open accounts for new marijuana businesses due to cost, risk, or both.
Although the Cole Memo was rescinded and FinCEN’s guidance has not made financial services widely available to legal marijuana businesses, a key legislative safeguard for the medical cannabis industry remains in place. Specifically, certain temporary federal legislative enactments that protect the medical marijuana industry have also been in effect. For instance, certain marijuana businesses receive a measure of protection from federal prosecution by operation of a temporary appropriations measures that has been enacted into law as an amendment or “rider” to federal spending bills passed by Congress and signed by both Presidents Obama and Trump. First adopted in the Appropriations Act of 2015, Congress has since included in successive budgets a “rider” that prohibits the DOJ from expending any funds to enforce any law that interferes with a state’s implementation of its own medical marijuana laws. The rider is known as the “Rohrbacher-Farr” Amendment after its original lead sponsors (it is also sometimes referred to as the Rohrabacher-Blumenauer Amendment or the Joyce-Leahy Amendment). In 2021, President Biden proposed a budget with the Rohrbacher-Farr amendment included. The amendment was then renewed through a series of stopgap spending bills, and on March 15, 2021, the amendment was renewed through the signing of the fiscal year 2022 omnibus spending bill, effective through September 30, 2022.
55
Though there is no guarantee the Presidency of Joe Biden or a future administration will not change relevant federal policy, as a practical matter, the legal marijuana industry has not seen a material change in federal enforcement activities since rescission of the Cole Memo. However, it is possible existing appropriation rider protection and existing prosecutorial discretion not to enforce federal drug laws against state-legal marijuana business could change at any time.
On July 14, 2021, Senate Majority Leader Chuck Schumer (D-NY) and Senators Cory Booker (D-NJ) and Ron Wyden (D-OR) released a discussion draft of their co-sponsored “Cannabis Administration and Opportunity Act” bill (the CAOA). This proposed legislation would legalize and regulate cannabis in the U.S. at the federal level. The sponsoring offices have invited the public to comment on the discussion draft through September 1, 2021. While the goals set forth in the CAOA are admirable, the discussion draft remains subject to considerable revision, and it remains unclear whether or to what extent all or portions of it may garner enough support to pass.
Finally, revenue from our marijuana operations is subject to Section 280E of the Code. Section 280E of the Code prohibits marijuana businesses from deducting ordinary and necessary business expenses, resulting in a materially higher effective federal income tax rate than businesses in other industries Therefore, businesses in the legal cannabis industry may be less profitable than they would otherwise be in a different industry.
Industrial Hemp
On December 20, 2018, the Agricultural Improvement Act of 2018 (the Farm Bill) became law in the U.S. Under the Farm Bill, industrial and commercial hemp is no longer to be classified as a Schedule I controlled substance in the U.S. Hemp includes the plant cannabis sativa L and any part of that plant, including seeds, derivatives, extracts, cannabinoids and isomers. To qualify under the Farm Bill, hemp must contain no more than 0.3 % of delta-9-THC. The Farm Bill explicitly allows interstate commerce of hemp which will enable the transportation and shipment of hemp across state lines, thus, the Farm Bill fundamentally changed how hemp and hemp-derived products (such as those containing CBD extracted from hemp) are regulated in the U.S.
State Regulatory Environment
The following sections describe the legal and regulatory landscape in states where our subsidiaries currently operate or intend to operate in the near-term future. While we actively work to ensure all of our operations are fully compliant with applicable state and local laws, rules, regulations, licensing requirements, ordinances and other applicable governing authority, the rules and regulations as outlined below are not a comprehensive representation of all the rules that we and our subsidiaries are required to follow in each applicable state. There are significant risks associated with our business and readers are strongly encouraged to carefully review and consider all of the risks set forth and described herein.
Common State Law Requirements
Although each state has its own laws and regulations regarding the operation of cannabis businesses, certain of the laws and regulations are consistent across jurisdictions. For example, to operate legally under state laws, marijuana businesses must typically obtain a license from the state. In some states, local marijuana-specific approvals are also required. In these jurisdictions, local governments may be authorized to prohibit or otherwise impose material restrictions on cannabis operations, including by proscribing rules limiting the type(s) and/or number of license(s) allowed (such authority is in addition to ordinary and customary building, fire and land use regulatory control). In many cases, securing local approval(s) is a prerequisite to state issuance of a full or unconditional license.
License application and renewal processes are unique to each state, and as applicable, each locality. However, generally each state’s application process requires a comprehensive criminal history disclosure of key individuals (such as major shareholders, directors, officers, certain managers and other individuals to the extent they are known at the time of application (Key Individuals)), and as to the applicant entity (and often its affiliates) and such Key Individuals, marijuana licensing and compliance history, financial and personal disclosures, detailed operating plans, facility information (often including drawings and plans), security-related plans, an affirmative obligation to report changes to or deviations from information set forth in the application, and other information designed to ensure only reputable, law-abiding individuals and entities ready, willing and able to operate in compliance with applicable state laws, rules and regulations are awarded marijuana licenses.
56
Applicants for marijuana licenses are commonly required to submit standard operating procedures (SOPs) describing how the proposed business will secure its facility(ies), manage inventory, comply with inventory tracking requirements and other reporting obligations, effectuate safe marijuana transactions, handle waste, train employees, implement quality control measures, and perform other tasks necessary and appropriate to operate in a safe, secure, and compliant manner. SOPs submitted as part of licensing applications are typically reviewed, evaluated and ultimately approved by regulators, and must generally remain in force and effect after issuance of a license. Any material change to SOPs requires prior written regulatory approval in nearly all cases. Finally, marijuana operations are continuously subject to inspection, with or without notice, by cannabis regulators and certain authorized law enforcement agencies.
California
In California, State and local medical and adult-use cannabis business licenses are renewed annually. Each year, licensees are required to submit a renewal application per guidelines published by the Department of Cannabis Control (the DCC), which is the successor regulator to the Bureau of Cannabis Control. While renewals are annual, there is no limit to the number of renewals a licensee may obtain. Assuming requisite renewal fees are paid, renewal applications are submitted in a timely manner, and the establishment has not been cited for material violations, renewal applicants can anticipate approval in the ordinary course of business. However, any unexpected denials, delays or costs associated with a licensing renewal could impede planned operations and may have a material adverse effect on our business, financial condition, results of operations or prospects.
License and Regulations
Adult-use retailer licenses permit the sale of cannabis and cannabis products to any individual age 21 years of age or older who does not possess a physician’s recommendation. Thus, should a subsidiary be awarded a license, it will be authorized to sell cannabis and cannabis products to adults over the age of 21 subject to customer presentation of a valid government-issued photo ID. As with all state-legal marijuana programs, only cannabis grown in California can be sold in California and retail licensees may only sell cannabis products procured from a duly licensed distributor or licensed microbusiness authorized to engage in distribution. All cannabis products are subject to appropriate laboratory testing, packaging, labeling, and tracking requirements. Upon receipt, licensed retailers must confirm cannabis products have not expired, are properly packaged and bear batch numbers which correspond with tracking and laboratory analysis documentation. Cannabis and cannabis products may only be displayed for inspection and sale on the sales floor of the facility, and may only be removed from packaging for customer inspection if placed in a proper container provided by the licensee and not readily accessible without the assistance of licensee staff (who must remain with the customer throughout such inspection). Any cannabis product displayed or inspected in this manner must be destroyed following inspection or when no longer being used for display purposes and may not be sold or consumed. Retailers may only provide free cannabis products under certain, very limited circumstances and may not sell other goods, with the exception of cannabis accessories and branded merchandise.
Medicinal retailer licenses permit the sale of medicinal cannabis and cannabis products for use pursuant to the Compassionate Use Act of 1996 (CUA), found at Section 11362.5 of the California Health and Safety Code, by a medicinal cannabis patient in California who possesses a physician’s recommendation. Only certified physicians may provide medicinal marijuana recommendations. We maintain an open, transparent and collaborative relationship with the DCC and local-level cannabis regulators.
Reporting Requirements
The State of California has selected Franwell Inc.’s METRC solution (METRC) as the State’s track-and-trace (T&T) system used to track commercial cannabis activity and movement along the legal supply chain. While METRC is interoperable with other third-party systems via application programming interface, only licensees have access to METRC itself.
Operating Procedure Requirements
Licensing applicants must submit SOPs describing how the operator will, among other requirements, secure the facility, manage inventory, comply with seed-to-sale requirements, dispense cannabis, and handle waste. Once an SOP is approved by the governing regulating body(ies), licensees must provide their employees with SOP training and seek written approval from governing regulating bodies before materially changing their SOPs.
57
Site-Visits & Inspections
The DCC and its authorized representatives have broad authority, with or without notice, to inspect licensed cannabis operations, including premises, facilities, equipment, books and records (which may be copied, and such copies retained), and cannabis products. Failure to grant DCC representatives full and immediate access to facilities, property, and premises, and to cooperate with inspections and investigations may result in disciplinary action. Laws and regulations enacted by many local jurisdictions grant local cannabis governing bodies and law enforcement agencies similar inspection authority.
Storage and Security
To ensure the safety and security of cannabis facilities and operations, the DCC requires licensees to:
| 1. | Maintain a fully operational security alarm system; |
| 2. | Contract for security guard services; |
| 3. | Maintain a video surveillance system that records continuously 24 hours a day; |
| 4. | Ensure adequate lighting is installed and maintained on and about licensed facilities; |
| 5. | Only transact business during authorized hours of operations; |
| 6. | Store cannabis and cannabis product only in areas identified for such purposes on drawings submitted to and approved by the State of California in connection with licensing; |
| 7. | Store all cannabis and cannabis products in a secured, locked room or a vault; |
| 8. | Report to local law enforcement within 24 hours after being notified or becoming aware of the theft, diversion, or loss of cannabis; and |
| 9. | To the extent applicable based on a licensee’s authorized scope of operations, ensure the safe transport of cannabis and cannabis products between licensed facilities, and maintain a delivery manifest in any vehicle transporting cannabis and cannabis products. Only vehicles registered with the DCC, that meet DCC distribution requirements, are to be used to transport cannabis and cannabis products. |
In addition to DCC storage and security requirements, local jurisdictions may have additional storage and security requirements. Such requirements, to the extent they exist, may vary from one locality to another.
We are in compliance with the laws of the State of California and the related cannabis licensing framework. There are no current incidences of non-compliance, citations or notices of violation which are outstanding which may have an impact on our licenses, business activities or operations in the State of California. Notwithstanding the foregoing, like most businesses, we may from time-to-time experience incidences of non-compliance with applicable rules and regulations in the states in which we operate, including the State of California, and such non-compliance may have an impact on our licenses, business activities or operations in the applicable state. However, we take steps to minimize, disclose and remedy all incidences of non-compliance which may have an impact on our licenses, business activities or operations in all states in which we operate, including the State of California. See “Regulatory Framework – Compliance.”
We, through our subsidiaries, currently hold three (3) medical and adult-use cannabis business licenses to operate dispensaries in Santa Barbara, California, Palm Springs, California and Grover Beach, California. Our dispensaries located in Santa Barbara and Grover Beach are currently operational. Our Palm Springs dispensary’s operations, which were voluntarily suspended while the store underwent a significant renovation, resumed operations in the third quarter of 2022. In addition, one of our subsidiaries entered into a long-term lease agreement for a bespoke, ground-up build in Culver City, California. We also received approval to move forward in the merit-based application process as one of three selected applicants for a storefront retail (and ancillary delivery) permit in Culver City, California, and we estimate the Culver City dispensary will be operational in the first quarter of 2023.
58
Illinois
Illinois Regulatory Landscape
In January 2014, the Compassionate Use of Medical Cannabis Pilot Program Act, which allows individuals diagnosed with certain debilitating or “qualified” medical conditions to access medical marijuana, became effective. There are over 35 qualifying conditions as part of the medical program, including epilepsy, traumatic brain injury, and post-traumatic stress disorder. In January 2019, the Illinois Department of Health launched the Opioid Alternative Pilot Program, that allows individuals who have/could receive a prescription for opioids to access medical marijuana.
In June 2019, Illinois legalized adult-use marijuana pursuant to the Cannabis Regulation and Tax Act (the IL Act). Effective January 1, 2020, Illinois residents 21 years of age and older may possess up to 30 grams of marijuana (non-residents may possess up to 15 grams). The IL Act authorizes the Illinois Department of Financial and Professional Regulation (IDFPR) to issue up to 75 Conditional Adult Use Dispensing Organization licenses before May 1, 2020 and an additional 110 conditional licenses during 2021 (no person may hold a financial interest in more than 10 dispensing organizations); however, conditional licenses have yet to be issued due to procedural delays related to litigation against the State of Illinois to which we are not currently a party to. Existing medical dispensaries were able to apply for an “Early Approval Adult Use Dispensing Organization License” to serve adult users at an existing medical dispensary or at a secondary site. The IDFPR has granted approximately 48 Early Approval Adult Use Dispensing Organization licenses to date. The IDFPR also held an application period for Conditional Adult Use Cannabis Dispensary Licenses from December 10, 2019 through January 2, 2020. Licenses from this round of applications have not yet been awarded, and the anticipated award date has been delayed due to the COVID-19 pandemic and challenges to the application process.
The Illinois Department of Agriculture (the IL Ag. Department) is authorized to make up to 30 cultivation center licenses available between the state’s medical and adult-use programs. As with existing medical dispensaries, existing cultivation centers were able to apply for an “Early Approval Adult Use Cultivation Center License.” The IL Ag. Department has issued approximately 21 Early Approval Adult Use Cultivation Centers to date. No person can hold a financial interest in more than three cultivation centers, and the centers are limited to 210,000 sq. ft. of canopy space. Cultivation centers are also prohibited from discriminating in price when selling to dispensaries, craft growers, or infuser organizations. The IL Ag. Department is also permitted to license up to 40 craft growers and 40 infuser organizations by July 1, 2020 (license awards have been delayed due to the COVID-19 pandemic) and another 60 of each license type by the end of 2021; however, the remaining 60 craft grower licenses have yet to be awarded due to procedural delays related to litigation against the State of Illinois to which we are not currently a party to. The IL Ag. Department closed the application period for craft growers, infusers, and cannabis transporters in March 2020.
The IL Act imposes several operational requirements on adult-use licensees and requires prospective licensees to demonstrate their plans to comply with such requirements. For example, applicants for dispensary licenses must include an employee training plan, a security plan, recordkeeping and inventory plans, a quality control plan, and an operating plan. Applicants for craft growers must similarly submit a facility plan, an employee training plan, a security plan, a record keeping plan, a cultivation plan, a product safety and labeling plan, a business plan, an environmental plan, and more.
Licensees must establish methods for identifying, recording, and reporting diversion, theft, or loss, correcting inventory errors, and complying with product recalls. Licensees also must comply with detailed inventory, storage, and security requirements. Cultivation licenses are subject to similar operational requirements, such as complying with detailed security and storage requirements, and must also establish plans to address energy, water, and waste-management needs. Dispensary licenses will be renewed bi-annually, and cultivation licenses, craft grower licenses, infuser organization licenses, and transporter licenses will be renewed annually.
The IL Ag. Department is authorized to promulgate, and has promulgated, regulations for cultivators, craft growers, infuser organizations, and transporting organizations. The IDFPR is authorized to regulate dispensaries but has not yet issued adult-use regulations. Therefore, currently licensed adult-use retail operations are governed by the IL Act and adult-use retail applications submitted during the application window which closed on January 2, 2020 will be evaluated under and in accordance with the IL Act.
We, through our subsidiaries in the State of Illinois, are in compliance with applicable licensing requirements in the State of Illinois.
59
To the knowledge of management, there have not been any statements or guidance made by federal authorities or prosecutors regarding the risk of enforcement action specific to the State of Illinois. For more information on federal enforcement and the risks associated with the U.S. cannabis regulatory environment generally, see without limitation, “Risk Factors – Risks Related to the Regulatory Environment.”
Illinois Licenses
As of the date of this prospectus, we operate four adult-use (with two co-located medical) cannabis dispensaries in the State of Illinois, located in or around the cities of Sauget, Normal and Bloomington. All four dispensaries are operated under our BEYOND/HELLO™ brand.
On August 19, 2021, Jushi’s partner Northern Cardinal Ventures, LLC was selected to receive a conditional retail dispensary license in Illinois via the state’s lottery process. The dispensary location is designated for the Peoria Bureau of Labor Statistics region in Illinois and will be BEYOND/HELLO’s fifth location in the state. The issuance of a conditional retail dispensary license to Northern Cardinal Ventures, LLC, along with other selected recipients via the state’s lottery process, is presently enjoined pursuant to ongoing litigation against the state, of which neither Northern Cardinal Ventures, LLC nor Jushi is a party to.
All medical and adult-use dispensing organizations licensed by IDFPR hold registration certificates valid for a period of one year and subject to annual renewals after required fees are paid and the organization remains in good standing. Renewals are generally communicated by IDFPR within 90 days of a license's expiration through email and include a renewal form. Provided that the requisite renewal fees are paid, the renewal application is submitted in a timely manner, and there are no material violations noted against the applicable license, Beyond Hello IL, LLC (BHIL) would expect to receive the applicable renewed license in the ordinary course of business. Any unexpected delays or costs associated with the licensing renewal process could impede the ongoing or planned operations and could have a material adverse effect on our business, financial condition, results of operations or prospects.
License and Regulations
Medical marijuana retail dispensary licenses permit BHIL to purchase cannabis and cannabis products from licensed cultivation/processing facilities and to sell cannabis and cannabis products to registered patients. The adult-use dispensing organization license permits BHIL to acquire cannabis from a licensed cultivation center, craft grower, processing organization, or another dispensary and to sell cannabis and cannabis products (and limited other items) to adult-use purchasers, registered medical cannabis patients and registered caregivers.
BHIL must operate in accordance with the representations made in its license application materials, unless otherwise approved by the IDFPR. It must include its name on the packaging of any cannabis product it sells. All medical products must be obtained from an Illinois registered medical cultivation center, while all adult-use products must be obtained from a licensed adult-use cultivation center, craft grower, processing organization, or another dispensary. BHIL must inspect and document (e.g., through the State of Illinois tracking system and in accordance with SOPs) all cannabis and cannabis products it acquires for resale. Any cannabis or cannabis products not properly packaged, labeled or inconsistent with State of Illinois tracking records must be rejected at the time of delivery. At all times, dispensing facilities must remain in compliance with all applicable building, fire, safety and land use laws, rules and regulations, and may not operate a drive through window or offer delivery services. BHIL may only operate during state regulated approved hours (6 a.m. to 10 p.m., daily) and must ensure two or more employees are present during all operating hours.
Each dispensary must submit a list of all third-party vendors to the IDFPR and identify all service professionals that will work at the dispensary by name and set forth a description of the services such person will provide. No service professional may work in the dispensary until his or her name is provided to IDFPR and appears on the facility’s service professional list.
BHIL may not produce or manufacture cannabis or cannabis products and may not permit on-site consumption at its facilities. BHIL may only sell cannabis or cannabis products to consumers who present a valid medical cannabis registration identification card or valid government-issued photo identification (ID) evidencing the customer is 21 years of age or older. BHIL must deal with all suppliers on the same terms and may not enter into an exclusive agreement with any supplier. Further, BHIL may not contract with, pay, or have a profit-sharing arrangement with third party groups involved in assisting individuals with finding a physician or completing the patient or participant application; nor may it pay a referral fee to a third-party group for sending it patients or participants. No more than 40% of its adult-use inventory may originate from a single supplier. Dispensing organizations are subject to inspections, with or without notice. Licensees are required to cooperate with such inspections and must make all records, plans, logs, reports and other operational documents available for inspection and copying upon request.
60
Craft grower licensees are authorized to cultivate cannabis and manufacture cannabis products (including cannabis infused products), and to sell cannabis and cannabis products to licensed adult-use dispensing organizations or for use at licensed manufacturers. Transportation licensees are authorized to transport cannabis and cannabis products between licensed cannabis facilities.
Storage and Security
BHIL dispensaries must store inventory on-site in a secured and restricted-access area and enter information into the State of Illinois’ tracking system as required by Illinois law and IDFPR rules. Any cannabis or cannabis products in an open or defective package, which have expired, or which we otherwise have reason to believe have been opened or tampered with must be segregated in secure storage until promptly and properly disposed of.
Dispensing facilities are also required to implement security measures designed to deter and prevent unauthorized entry into the facility (and restricted-access areas) and theft, loss or diversion of cannabis or cannabis products. In this respect, dispensing facilities must maintain a commercial grade alarm and surveillance system installed by an Illinois licensed private alarm contractor or private alarm contractor agency. BHIL must also implement various security measures, as required by law, rule regulation or SOPs, designed to protect the premises, customers and dispensing organization agents (employees).
Reporting Requirements
The State of Illinois uses BioTrack THC as its inventory tracking system. All dispensing organization licensees are required to use a real-time, web-based inventory tracking/point-of-sale system that is accessible to IDFPR at any time, and at a minimum, tracks date of sale, amount, price, and currency. BHIL uses BioTrack THC for both inventory management and as a point-of-sale system. Licensees are also required to track each sales transaction at the time of the sale, daily beginning and ending inventory, acquisitions (including information about the supplier and the product) and disposal.
Transportation Requirements
Currently, licensed cultivation centers may transport cannabis and cannabis products in accordance with certain guidelines; however, from July 1, 2020 cultivation centers are prohibited from transporting adult-use cannabis without obtaining a separate transporting organization license, provided that such prohibition was and remains suspended pursuant to Executive Order 2020-45. For medical marijuana, dispensing organizations must receive a copy of the shipping manifest prepared by the cultivation center in advance of transport and is required to check the product delivered against such manifest at the time of delivery. All cannabis and cannabis products must be packaged in properly labeled and sealed containers and may not be accepted by a dispensary recipient if packaging is damaged or labels are missing, damaged or tampered with.
We are in compliance with the laws of the State of Illinois and the related cannabis licensing framework. There are no current incidences of non-compliance, citations or notices of violation outstanding which have an impact on our licenses, business activities or operations in the State of Illinois. Notwithstanding the foregoing, like all businesses we may from time-to-time experience incidences of non-compliance with applicable rules and regulations in the states in which we operate, including the State of Illinois, and such non-compliance may have an impact on our licenses, business activities or operations in the applicable state. However, we take steps to minimize, disclose and remedy all incidences of non-compliance which may have an impact on our licenses, business activities or operations in all states in which we operate, including the State of Illinois. See “Regulatory Framework – Compliance.”
61
Massachusetts
Massachusetts Regulatory Landscape
Cannabis for medical use was legalized in Massachusetts by voter approval of the Massachusetts Medical Marijuana Initiative in 2012. The law took effect on January 1, 2013, eliminating criminal and civil penalties for the possession and use of up to a 60-day or ten (10) ounce supply of marijuana for medical use for patients possessing a State-issued registration card. On November 8, 2016, Massachusetts voters approved Question 4 or the Massachusetts Marijuana Legalization, Regulation and Taxation of Marijuana Initiative, which allowed for recreational or “adult-use” cannabis in the Commonwealth of Massachusetts. On July 28, 2017, the Cannabis Control Commission (the CCC) was established under Chapter 55 of the Acts of 2017 to implement and administer laws enabling access to medical and adult-use cannabis. The Commission was appointed on September 12, 2017. On November 16, 2018, the CCC issued the first notices for retail marijuana establishments to commence adult-use operations in Massachusetts. Under the current law, there are no State-wide limits on the total number of licenses issued; however, no individual or entity shall be an owner or a controlling person over more than three licenses in a particular class of license. Similarly, no individual, corporation or other entity shall be an owner or in a position to control the decision making of more than three licenses in a particular class of license. In addition, all marijuana establishments are required to enter into host community agreements with the municipality in which they are located.
Massachusetts Licenses
Through an affiliate we acquired the licenses previously held by Nature’s Remedy, which include one (1) final adult-use cultivation license, one (1) final adult-use product manufacturer license and two (2) final adult-use retailer licenses in the Commonwealth of Massachusetts. Nature’s Remedy also holds one (1) provisional license for adult-use cultivation. Furthermore, Nature’s Remedy held one (1) medical license that permits vertically-integrated operations including the ability to maintain one (1) medical marijuana dispensary in the Commonwealth of Massachusetts. Under the terms of the adult-use marijuana cultivator license, the licensee may cultivate, process and package marijuana, and transfer marijuana products to marijuana establishments, but not to consumers. An adult-use marijuana product manufacturer is an entity authorized to obtain, manufacture, process and package marijuana and marijuana products, to transfer marijuana and marijuana products to marijuana establishments, but not to consumers. An adult-use marijuana retailer is an entity authorized to purchase, repackage, white label, and transport marijuana and marijuana products from marijuana establishments and transfer marijuana and marijuana products to marijuana establishments and to sell to consumers. The medical marijuana treatment center (MTC) licenses are vertically integrated and permit a licensee to cultivate, manufacture, process, package, transport, deliver, sell, and purchase marijuana pursuant to the terms of the medical licenses. Massachusetts does not issue a single vertically integrated adult-use license like the MTC license. License types for adult-use are individual for each function and a licensee may pursue multiple license types. Because marijuana is not federally legal, a licensee can sell only cannabis that is grown and manufactured in the Commonwealth of Massachusetts. An adult-use marijuana retailer provides a retail location which may be accessed by consumers 21 years of age or older or, if the retailer is co-located with an MTC, by individuals who are registered qualifying patients with the Medical Use of Marijuana Program with a registration card. In order for a customer to be dispensed marijuana, they must present a valid government issued photo ID immediately upon entry of the retail facility. If the individual is younger than 21 years old but 18 years of age or older, he or she shall not be admitted unless he or she produces an active medical registration card issued by the CCC. If the individual is younger than 18 years old, he or she shall not be admitted unless he or she produces an active medical registration card and is accompanied by a personal caregiver with an active medical registration card. In addition to the medical registration card, registered qualifying patients 18 years of age and older and personal caregivers must also produce proof of identification. Each recreational customer may be dispensed no more than one ounce of marijuana or five grams of marijuana concentrate per transaction as outlined in 935 CMR 500.140(3)(a)(1). Medical patients may be dispensed up to a 60-day supply of marijuana, or the equivalent amount of marijuana in marijuana infused products, that a registered qualifying patient would reasonably be expected to need over a period of 60 calendar days for his or her personal medical use, which is ten ounces, subject to 935 CMR 501.140(3)(a). Allowable forms of marijuana in Massachusetts include smokable dried flower, dried flower for vaporizing, cannabis derivative products (i.e., vape pens, gel caps, tinctures, etc.) and medical cannabis-infused products, including edibles.
Reporting Requirements
The CCC has selected METRC as the Commonwealth of Masachusetts’ seed to sale tracking system used to track commercial cannabis activity and movement across the distribution chain. The system allows for other third-party system integration via application programming interface.
62
Medical Cannabis Regulations
The Commonwealth of Massachusetts has authorized the cultivation, possession and distribution of marijuana for medical purposes by certain licensed Massachusetts marijuana businesses. The Medical Use of Marijuana Program (the MUMP) registers qualifying patients, personal caregivers, MTCs, and MTC agents. MTCs were formerly known as Registered Marijuana Dispensaries (RMDs). The MUMP was established by Chapter 369 of the Acts of 2012, “An Act for the Humanitarian Medical Use of Marijuana”, following the passage of the Massachusetts Medical Marijuana Initiative, Ballot Question 3, in the 2012 general election. Additional statutory requirements governing the MUMP were enacted by the Legislature in 2017 and codified at G.L. c. 94I, et. seq. (referred to herein as the Massachusetts Medical Act). MTC Certificates of Registration are vertically integrated licenses in that each MTC Certificate of Registration entitles a license holder to one cultivation facility, one processing facility and one dispensary locations. There is a limit of three MTC licenses per person/entity. The CCC regulations, 935 CMR 501.000 et seq. (referred to herein as the Massachusetts Medical Regulations), provide a regulatory framework that requires MTCs to cultivate, process, transport and dispense medical cannabis in a vertically integrated marketplace. Patients with debilitating medical conditions qualify to participate in the program, including conditions such as cancer, glaucoma, positive status for human immunodeficiency virus (HIV), acquired immune deficiency virus (AIDS), hepatitis C, amyotrophic lateral sclerosis, Crohn’s disease, Parkinson’s disease, and multiple sclerosis when such diseases are debilitating, and other debilitating conditions as determined in writing by a qualifying patient’s healthcare provider. The CCC assumed control of the MUMP from the Department of Public Health on December 23, 2018. The CCC approved revised regulations for the MUMP on November 30, 2020, which are now effective.
Medical Cannabis Licensing Requirements
The Massachusetts Medical Regulations delineate the licensing requirements for MTCs in Massachusetts. Licensed entities must demonstrate the following: (i) they are licensed and in good standing with the Secretary of the Commonwealth of Massachusetts, Department of Revenue and Department of Unemployment Assistance; (ii) no executive, member or any entity owned or controlled by such executive or member directly or indirectly controls more than three MTC licenses; (iii) an MTC may not cultivate medical cannabis from more than two locations statewide; (iv) MTC agents must be registered with the CCC; (v) an MTC must have a program to provide reduced cost or free marijuana to patients with documented verifiable financial hardships; (vi) one executive of an MTC must register with the Massachusetts Department of Criminal Justice Information Services on behalf of the entity as an organization user of the Criminal Offender Record Information system; (vii) the MTC applicant has at least $500,000 in its control as evidenced by bank statements, lines of credit or equivalent; and (viii) payment of the required application fee.
In an MTC application, an applicant must also demonstrate or include: (i) the name, address, date of birth and resumes of each executive of the applicant and of the members of the entity; (ii) a plan to obtain liability insurance coverage in compliance with statutes; (iii) a detailed summary of the business plan for the MTC; (iv) an operational plan for the cultivation of marijuana including a detailed summary of policies and procedures; and (v) a detailed summary of the operating policies and procedures for the MTC including security, prevention of diversion, storage of marijuana, transportation of marijuana, inventory procedures, procedures for quality control and testing of product for potential contaminants, procedures for maintaining confidentiality as required by law, personnel policies, dispensing procedures, record keeping procedures, plans for patient education and any plans for patient or personal caregiver home delivery. An MTC applicant must also demonstrate that it has: (i) a successful track record of running a business; (ii) a history of providing healthcare services or services providing marijuana for medical purposes in or outside of Massachusetts; (iii) proof of compliance with the laws of the Commonwealth of Massachusetts; (iv) complied with the laws and orders of the Commonwealth of Massachusetts; and (v) a satisfactory criminal and civil background. Finally, an MTC applicant must specify a cultivation tier for their license, which establishes the minimum and maximum square footage of canopy for their cultivation operation.
Upon the determination by the CCC that an MTC applicant has responded to the application requirements in a satisfactory fashion, the MTC applicant is required to pay the applicable registration fee and shall be issued a Provisional MTC license and, following completion of certain regulatory requirements, a Final MTC license.
After receipt of a Provisional MTC license, the CCC shall review architectural plans for the building of the MTC’s cultivation facility and/or dispensing facilities, and shall either approve, modify or deny the same. Once approved, the MTC provisional license holder shall construct its facilities in conformance with the requirements of the Massachusetts Medical Regulations. Once the CCC completes its inspections and issues approval for an MTC of its facilities, the CCC shall issue a Final MTC License to the MTC applicant. Final MTC Licenses are valid for one year, and shall be renewed by filing the required renewal application no later than sixty days prior to the expiration of the certificate of registration. A licensee may not begin cultivating marijuana until it has been issued a Final MTC License by the CCC.
63
MTC Licenses in Massachusetts are renewed annually. Before expiry, licensees are required to submit a renewal application. While renewals are granted annually, there is no ultimate expiry after which no renewals are permitted. Additionally, in respect of the renewal process, provided that the requisite renewal fees are paid, the renewal application is submitted in a timely manner, and there are no material violations noted against the applicable license, we would expect to receive the applicable renewed licenses in the ordinary course of business.
Massachusetts Medical Cannabis Dispensary Operational Requirements
An MTC shall follow its written and approved operation procedures in the operation of its dispensary locations. Operating procedures shall include (i) security measures in compliance with the Massachusetts Medical Regulations; (ii) employee security policies including personal safety and crime prevention techniques; (iii) hours of operation and after-hours contact information; (iv) a price list for marijuana; (v) storage and waste disposal protocols in compliance with state law; (vi) a description of the various strains of marijuana that will be cultivated and dispensed, and the forms that will be dispensed; (vii) procedures to ensure accurate recordkeeping including inventory protocols; (viii) plans for quality control; (ix) a staffing plan and staffing records; (x) diversion identification and reporting protocols; and (xi) policies and procedures for the handling of cash on MTC premises including storage, collection frequency and transport to financial institutions. The siting of dispensary locations is expressly subject to local/municipal approvals pursuant to state law, and municipalities control the permitting application process that an MTC must comply with. More specifically, an MTC is to comply with all local requirements regarding siting, provided however that if no local requirements exist, an MTC shall not be sited within a radius of 500 feet of a school, daycare center, or any facility in which children commonly congregate. The 500-foot distance under this section is measured in a straight line from the nearest point of the facility in question to the nearest point of the proposed MTC. The Massachusetts Medical Regulations require that MTCs limit their inventory of seeds, plants, and useable marijuana to reflect the projected needs of registered qualifying patients. An MTC may only dispense to a registered qualifying patient or caregiver who has a current valid certification.
Massachusetts Medical Cannabis Security and Storage Requirements
An MTC is to implement sufficient security measures to deter and prevent unauthorized entrance into areas containing marijuana and theft of marijuana at the MTC. These measures must include: (i) allowing only registered qualifying patients, caregivers, dispensary agents, authorized persons, or approved outside contractors access to the MTC facility; (ii) preventing individuals from remaining on the premises of an MTC if they are not engaging in activities that are permitted; (iii) disposing of marijuana or by-products in compliance with law; (iv) establishing limited access areas accessible only to authorized personnel; (v) storing finished marijuana in a secure locked safe or vault; (vi) keeping equipment, safes, vaults or secured areas securely locked; (vii) ensuring that the outside perimeter of the MTC is sufficiently lit to facilitate surveillance; and (viii) ensuring that landscaping or foliage outside of the RMD does not allow a person to conceal themselves. An MTC shall also utilize a security/alarm system that: (i) monitors entry and exit points and windows and doors, (ii) includes a panic/duress alarm, (iii) includes system failure notifications, (iv) includes 24-hour video surveillance of safes, vaults, sales areas, areas where marijuana is cultivated, processed or dispensed, and (v) includes date and time stamping of all records and the ability to produce a clear, color still photo. The video surveillance system shall have the capacity to remain operational during a power outage. The MTC must also maintain a backup alarm system with the capabilities of the primary system, and both systems are to be maintained in good working order and are to be inspected and tested on regular intervals.
Massachusetts Medical Cannabis Transportation Requirements
Marijuana or marijuana-infused products (MIPs) may be transported between licensed MTCs by MTC agents on behalf of an MTC. MTCs or deliver-only retailers may, with CCC approval, transport marijuana or MIPS directly to registered qualifying patients and caregivers as part of a home delivery program. An MTC shall staff transport vehicles with a minimum of two dispensary agents. At least one agent shall remain with the vehicle when the vehicle contains marijuana or MIPs. Prior to leaving the origination location, an MTC must weigh, inventory, and account for, on video, the marijuana to be transported.
64
Marijuana must be packaged in sealed, labeled, and tamper-proof packaging prior to and during transportation. In the case of an emergency stop, a log must be maintained describing the reason for the stop, the duration, the location, and any activities of personnel exiting the vehicle. An MTC shall ensure that delivery times and routes are randomized. Each MTC agent shall carry his or her CCC-issued MUMP ID Card when transporting marijuana or MIPs and shall produce it to CCC representatives or law enforcement officials upon request. Where videotaping is required when weighing, inventorying, and accounting of marijuana before transportation or after receipt, the video must show each product being weighed, the weight, and the manifest. An MTC must document and report any unusual discrepancy in weight or inventory to the CCC and local law enforcement within 24 hours. An MTC shall report to the CCC and local law enforcement any vehicle accidents, diversions, losses, or other reportable incidents that occur during transport, within 24 hours. An MTC shall retain transportation manifests for no less than one year and make them available to the CCC upon request. Any cash received from a qualifying patient or personal caregiver must be transported to an MTC immediately upon completion of the scheduled deliveries. Vehicles used in transportation must be owned, leased or rented by the MTC, properly registered, and contain a GPS system that is monitored by the MTC during transport of marijuana and said vehicle must be inspected and approved by the CCC prior to use.
During transit, an MTC is to ensure that: (i) marijuana or MIPs are transported in a secure, locked storage compartment that is part of the vehicle transporting the marijuana or MIPs; (ii) the storage compartment cannot be easily removed (for example, bolts, fittings, straps or other types of fasteners may not be easily accessible and not capable of being manipulated with commonly available tools); (iii) marijuana or MIPs are not visible from outside the vehicle; and (iv) the product is transported in a vehicle that bears no markings indicating that the vehicle is being used to transport marijuana or MIPs and does not indicate the name of the MTC. Each MTC agent transporting marijuana or MIPs shall have access to a secure form of communication with personnel at the origination location when the vehicle contains marijuana or MIPs.
Massachusetts Adult-Use Cannabis Licensing Requirements
Many of the same application requirements exist for an adult-use marijuana establishment license application as to those for a medical MTC application, and each owner, officer or member must undergo background checks and fingerprinting with the CCC. Applicants must submit the location and identification of each site, and must establish a property interest in the same, and the applicant and the local municipality must have entered into a host agreement authorizing the location of the adult-use marijuana establishment within the municipality, and said agreement must be included in the application. Applicants must include disclosure of any regulatory actions against it by the Commonwealth of Massachusetts, as well as the civil and criminal history of the applicant and its owners, officers, principals or members. The application must include, amongst other information, the proposed timeline for achieving operations, liability insurance, a business plan, and a detailed summary describing the marijuana establishment’s proposed operating policies including security, prevention of diversion, storage, transportation, inventory procedures, quality control, dispensing procedures, personnel policies, record keeping, maintenance of financial records, diversity plans, and employee training protocols.
Massachusetts Adult-Use Cannabis Dispensary Operational Requirements
Marijuana retailers are subject to certain operational requirements in addition to those imposed on marijuana establishments generally. Dispensaries must immediately inspect patrons’ identification to ensure that everyone who enters is at least 21 years of age. Dispensaries may not dispense more than one ounce of marijuana or five grams of marijuana concentrate per transaction. Point-of-sale systems must be approved by the CCC, and retailers must record sales data. Records must be retained and available for auditing by the CCC and the Secretary of the Commonwealth of Massachusetts, Department of Revenue and Department of Unemployment Assistance. Retailers are required to conduct monthly analyses of equipment and sales data to determine that such systems have not been altered or interfered with to manipulate sales data, and to report any such discrepancies to the CCC.
Dispensaries must also make consumer education materials available to patrons in languages designated by the CCC, with analogous materials for visually- and hearing-impaired persons. Such materials must include:
| 1. | A warning that marijuana has not been analyzed or approved by the FDA, that there is limited information on side effects, that there may be health risks associated with using marijuana, and that it should be kept away from children; |
65
| 2. | A warning that when under the influence of marijuana, driving is prohibited and machinery should not be operated; |
| 3. | Information to assist in the selection of marijuana, describing the potential differing effects of various strains of marijuana, as well as various forms and routes of administration; |
| 4. | Materials offered to consumers to enable them to track the strains used and their associated effects; |
| 5. | Information describing proper dosage and titration for different routes of administration, with an emphasis on using the smallest amount possible to achieve the desired effect; |
| 6. | A discussion of tolerance, dependence, and withdrawal; |
| 7. | Facts regarding substance abuse signs and symptoms, as well as referral information for substance abuse treatment programs; |
| 8. | A statement that consumers may not sell marijuana to any other individual; |
| 9. | Information regarding penalties for possession or distribution of marijuana in violation of Massachusetts law; and |
| 10. | Any other information required by the CCC. |
Massachusetts Adult-Use Cannabis Security and Storage Requirements
Each marijuana establishment must implement sufficient safety measures to deter and prevent unauthorized entrance into areas containing marijuana and theft of marijuana at the establishment. Security measures taken by the establishments to protect the premises, employees, consumers and general public shall include, but not be limited to, the following:
| 1. | Positively identifying and limiting access to individuals 21 years of age or older who are seeking access to the marijuana establishment or to whom marijuana products are being transported; |
| 2. | Adopting procedures to prevent loitering and ensure that only individuals engaging in activity expressly or by necessary implication are allowed to remain on the premises; |
| 3. | Proper disposal of marijuana in accordance with applicable regulations; |
| 4. | Securing all entrances to the marijuana establishment to prevent unauthorized access; |
| 5. | Establishing limited access areas which shall be accessible only to specifically authorized personnel limited to include only the minimum number of employees essential for efficient operation; |
| 6. | Storing all finished marijuana products in a secure, locked safe or vault in such a manner as to prevent diversion, theft or loss; |
| 7. | Keeping all safes, vaults, and any other equipment or areas used for the production, cultivation, harvesting, processing or storage, including prior to disposal, of marijuana or marijuana products securely locked and protected from entry, except for the actual time required to remove or replace marijuana; |
| 8. | Keeping all locks and security equipment in good working order; |
| 9. | Prohibiting keys, if any, from being left in the locks or stored or placed in a location accessible to persons other than specifically authorized personnel; |
| 10. | Prohibiting accessibility of security measures, such as combination numbers, passwords or electronic or biometric security systems, to persons other than specifically authorized personnel; |
| 11. | Ensuring that the outside perimeter of the marijuana establishment is sufficiently lit to facilitate surveillance, where applicable; |
| 12. | Ensuring that all marijuana products are kept out of plain sight and are not visible from a public place, outside of the marijuana establishment, without the use of binoculars, optical aids or aircraft; |
| 13. | Developing emergency policies and procedures for securing all product following any instance of diversion, theft or loss of marijuana, and conduct an assessment to determine whether additional safeguards are necessary; |
| 14. | Establishing procedures for safe cash handling and cash transportation to financial institutions to prevent theft, loss and associated risks to the safety of employees, customers and the general public; |
| 15. | Sharing the marijuana establishment’s floor plan or layout of the facility with law enforcement authorities, and in a manner and scope as required by the municipality and identifying when the use of flammable or combustible solvents, chemicals or other materials are in use at the marijuana establishment; |
66
| 16. | Sharing the marijuana establishment’s security plan and procedures with law enforcement authorities, including police and fire services departments, in the municipality where the marijuana establishment is located and periodically updating law enforcement authorities, police and fire services departments, if the plans or procedures are modified in a material way; and |
| 17. | Marijuana must be stored in special limited access areas, and alarm systems must meet certain technical requirements, including the ability to record footage to be retained for at least 90 days. |
Massachusetts Adult-Use Cannabis Transportation Requirements
Marijuana products may only be transported between licensed marijuana establishments by agents of registered marijuana establishments. A licensed marijuana transporter may contract with a marijuana establishment to transport that licensee’s marijuana products to other licensed establishments. All transported marijuana products are linked to the seed-to-sale tracking program. Any marijuana product that is undeliverable or is refused by the destination marijuana establishment shall be transported back to the originating establishment. All vehicles transporting marijuana products shall be staffed with a minimum of two marijuana establishment agents. At least one agent shall remain with the vehicle at all times that the vehicle contains marijuana or marijuana products. Prior to the products leaving a marijuana establishment, the originating marijuana establishment must weigh, inventory, and account for, on video, all marijuana products to be transported. Within eight hours after arrival at the receiving marijuana establishment, the receiving establishment must re-weigh, re-inventory, and account for, on video, all marijuana products transported. Marijuana products must be packaged in sealed, labeled, and tamper or child-resistant packaging prior to and during transportation. In the case of an emergency stop during the transportation of marijuana products, a log must be maintained describing the reason for the stop, the duration, the location, and any activities of personnel exiting the vehicle. A marijuana establishment or a marijuana transporter transporting marijuana products is required to ensure that all transportation times and routes are randomized and remain within Massachusetts.
Vehicles must additionally be equipped with a video system that includes one or more cameras in the storage area of the vehicle and one or more cameras in the driver area of the vehicle. The video cameras must remain operational at all times during the transportation process and have the ability to produce a clear color still photo whether live or recorded, with a date and time stamp embedded and that does not significantly obscure the picture.
Vehicles used for transport must be owned or leased by the marijuana establishment or transporter, and they must be properly registered, inspected, and insured in Massachusetts. Marijuana may not be visible from outside the vehicle, and it must be transported in a secure, locked storage compartment. Each vehicle must have a global positioning system, and any agent transporting marijuana must have access to a secure form of communication with the originating location.
CCC Inspections
The CCC or its agents may inspect an MTC, marijuana establishment and their affiliated vehicles at any time without prior notice. An MTC or marijuana establishment shall immediately upon request make available to the CCC information that may be relevant to a CCC inspection, and the CCC may direct an MTC or marijuana establishment to test marijuana for contaminants. Any violations found will be noted in a deficiency statement that will be provided to the MTC or marijuana establishment, and the MTC or marijuana establishment shall thereafter submit a Plan of Correction to the CCC outlining with particularity each deficiency and the timetable and steps to remediate the same. The CCC shall have the authority to suspend or revoke a certificate of registration in accordance with the applicable regulations.
Nevada
Nevada Regulatory Landscape
Medical marijuana use was legalized in Nevada by a ballot initiative in 2000. In November 2016, voters in Nevada passed an adult-use marijuana measure to allow for the sale of adult-use marijuana in the state. The first dispensaries to sell adult-use marijuana began sales in July 2017. The Nevada Cannabis Compliance Board (NV CCB) is the regulatory agency overseeing the medical and adult-use cannabis programs. Similar to California, cities and counties in Nevada are allowed to determine the number of local marijuana licenses they will issue.
67
To the knowledge of management, there have not been any statements or guidance made by federal authorities or prosecutors regarding the risk of enforcement action specific to the State of Nevada. For more information on federal enforcement and the risks associated with the U.S. cannabis regulatory environment generally, see without limitation, “Risk Factors – Risks Related to the Regulatory Environment.”
Nevada Licenses
FBS NV holds medical and adult-use cannabis cultivation, processing and distribution licenses issued by the NV CCB and a hemp handler license issued by the Nevada Department of Agriculture (NV DOA). Under applicable laws, licenses issued by the NV CCB or the NV DOA permit the applicable entities to cultivate, manufacture/process, package, sell or purchase pursuant to the terms of the license, which is issued by the NV CCB under the provisions of Nevada Revised Statutes section 453A.
In March 2022, we acquired 100% of the equity interest of an entity operating an adult-use and medical retail dispensary under the name The Apothecarium, which is used under license with an affiliate of TerrAscend Corp. in Las Vegas, Nevada. Additionally, in April 2022, we acquired NuLeaf, a Nevada-based vertically integrated operator. NuLeaf operates three adult-use and medical retail dispensaries in Las Vegas, Nevada, and Lake Tahoe, Nevada, in addition to a 27,000 sq. ft. cultivation facility in Sparks, Nevada, as well as a 13,000 sq. ft. processing facility in Reno, Nevada.
All marijuana establishments must register with the NV CCB. If applications contain all required information and after vetting by officers, establishments are issued a medical marijuana establishment registration certificate. In a local governmental jurisdiction that issues business licenses, the issuance by the NV CCB of a medical marijuana establishment registration certificate is considered provisional until the local government has issued a business license for operation and the establishment is in compliance with all applicable local governmental ordinances. Final registration certificates are valid for a period of one year and are subject to annual renewals after required fees are paid and the business remains in good standing. Renewal requests are typically communicated through email from the NV CCB and include a renewal form. The renewal periods serve as an update for the NV CCB on the licensee’s status toward active licensure. It is important to note that provisional licenses do not permit the operation of any commercial or medical cannabis activity. Only after a provisional licensee has gone through necessary state and local inspections, if applicable, and has received a final registration certificate from the NV CCB may an entity engage in cannabis business operation.
Any unexpected delays or costs associated with the licensing renewal process could impede the ongoing or planned operations and could have a material adverse effect on our business, financial condition, results of operations or prospects.
License and Regulations
In the State of Nevada, only cannabis that is grown in the State of Nevada can be sold in the State of Nevada.
Retail dispensary licenses and registration certificates permit a license holder to purchase marijuana from cultivation facilities, marijuana and marijuana products from product manufacturing facilities and marijuana from other retail stores and allows the sale of marijuana and marijuana products to consumers.
Medical cultivation licenses permit a license holder to acquire, possess, cultivate, deliver, transfer, have tested, transport, supply or sell marijuana and related supplies to medical marijuana dispensaries, facilities for the production of edible medical marijuana products and/or medical marijuana-infused products, or other medical marijuana cultivation facilities. One must have a final medical registration certificate in order to apply for adult-use status. Once so licensed, adult-use cultivators are authorized to perform the previously described for the adult-use market.
68
Medical product manufacturing licenses permit a license holder to acquire, possess, manufacture, deliver, transfer, transport, supply, or sell edible marijuana products or marijuana infused products to other medical marijuana production facilities or medical marijuana dispensaries. One must have a final medical registration certificate in order to apply for adult-use status. Once so licensed, adult-use cultivators are authorized to perform the previously described for the adult-use market.
Hemp handler licenses permit a license holder to handle raw industrial hemp, purchase hemp-derived constituents (such as hemp-derived CBD) from licensed hemp operators, and to infuse or manufacture products containing hemp-derived constituents.
Reporting Requirements
The State of Nevada uses METRC as its computerized T&T system used to track commercial cannabis on a seed-to-sale basis. Individual licensees whether, directly or through third-party integration systems, are required to push data to the state to meet all reporting requirements. Our chosen seed-to-sale system will capture the required data points for cultivation, manufacturing and retail as required under state law.
Storage and Security
To ensure the safety and security of cannabis business premises and to maintain adequate controls against diversion, theft, and loss of cannabis and cannabis products, FBS NV is required to do the following:
| 1. | Maintain an enclosed, locked facility; |
| 2. | Have a single secure entrance; |
| 3. | Train employees in security measures and controls, emergency response protocol, confidentiality requirements, safe handling of equipment, procedures for handling products, as well as the differences in strains, methods of consumption, methods of cultivation, methods of fertilization and methods for health monitoring; |
| 4. | Implement and install, at a minimum, the following security equipment and practices to deter and prevent unauthorized entrances: |
| a. | devices that detect unauthorized intrusion (which may include a signal system); |
| b. | exterior lighting designed to facilitate surveillance; |
| c. | electronic monitoring devices, further including (without limitation): |
| a. | at least one call-up monitor that is at least 19 inches in size; |
| b. | a video printer that can immediately produce a clear still photo from any video camera image; |
| c. | video cameras with a recording resolution of at least 704 x 480 that full capture all of the building’s points of ingress and egress as well as all interior limited access areas such that such cameras capture and can identify any activity occurring in or adjacent to the building; |
| d. | a video camera at each point-of-sale location which allows for the identification of any person who holds a valid registry identification card, including, without limitation, a designated primary caregiver, purchasing medical marijuana; |
| e. | a video camera in each grow room that can identify any activity occurring within the grow room in low light conditions; |
| f. | a method for storing video recordings from the video cameras for at least 30 calendar days; |
| g. | a failure notification system that provides an audible and visual notification of any failure in the electronic monitoring system; |
| h. | sufficient battery backup for video cameras and recording equipment to support at least five (5) minutes of recording in the event of a power outage; and |
| i. | a security alarm to alert local law enforcement of unauthorized breach of security; and |
| 1. | Implement security procedures that: |
| a. | restrict access of the establishment to only those persons/employees authorized to be there; |
| b. | deter and prevent theft; |
| c. | provide identification (badge) for those persons/employees authorized to be in the establishment; |
| d. | prevent loitering; |
69
| e. | require and explain electronic monitoring; and |
| f. | require and explain the use of automatic or electronic notifications to alert local law enforcement of any security breaches. |
There are no incidences of non-compliance, citations or notices of violation outstanding which have an impact on our licenses, business activities or operations in the State of Nevada. Notwithstanding the foregoing, as discussed in more detail under the subsection entitled “Compliance” below, like all businesses we may from time-to-time experience incidences of non-compliance with applicable rules and regulations in the states in which we operate, including the State of Nevada, and such non-compliance may have an impact on our licenses, business activities or operations in the applicable state. However, we take steps to minimize, disclose and remedy all incidences of non-compliance which may have an impact on our licenses, business activities or operations in all states in which we operate, including the State of Nevada. We are not aware of any incidence of non-compliance, citations or notices of violation received by FBS NV or the failure of FBS NV to comply with applicable licensing requirements and the regulatory framework enacted by the State of Nevada.
Ohio
Ohio Regulatory Landscape
House Bill 523, effective on September 8, 2016, legalized medical marijuana in Ohio. The Ohio Medical Marijuana Control Program (MMCP) allows people with certain medical conditions, upon the recommendation of an Ohio-licensed physician certified by the State Medical Board of Ohio, to purchase and use medical marijuana. House Bill 523 required that the framework for the MMCP would be in place no later than September 2018. This timeframe allowed for a deliberate process to ensure the safety of the public and to promote access to a safe product. Sales of medical marijuana in Ohio began in January 2019.
The following three state government agencies are responsible for the operation of the MMCP: (i) the Ohio Department of Commerce is responsible for overseeing medical marijuana cultivators, processors and testing laboratories; (ii) the State of Ohio Board of Pharmacy is responsible for overseeing medical marijuana retail dispensaries, the registration of medical marijuana patients and caregivers, the approval of new forms of medical marijuana and coordinating the Medical Marijuana Advisory Committee; and (iii) the State Medical Board of Ohio is responsible for certifying physicians to recommend medical marijuana and may add to the list of qualifying conditions for which medical marijuana can be recommended. Qualifying medical conditions for medical marijuana include: HIV/AIDS, Lou Gehrig’s disease, Alzheimer’s disease, cancer, chronic traumatic encephalopathy, Crohn’s disease, epilepsy or other seizure disorder, fibromyalgia, glaucoma, hepatitis C, inflammatory bowel disease, multiple sclerosis (MS), pain (either chronic, severe, or intractable), Parkinson’s disease, post-traumatic stress disorder, sickle cell anemia, spinal cord disease or injury, Tourette’s syndrome, traumatic brain injury, ulcerative colitis. In order for a patient to be eligible to obtain medical marijuana, a physician must make the diagnosis of one of these conditions. The State of Ohio Board of Pharmacy has published regulations for dispensaries, for the forms and methods for administering medical marijuana, and for patients and caregivers.
Several forms of medical marijuana are legal in Ohio, these include: inhalation of marijuana through a vaporizer (not direct smoking), oils, tinctures, plant material, edibles, patches and any other forms approved by the State of Ohio Board of Pharmacy.
To the knowledge of management, there have not been any statements or guidance made by federal authorities or prosecutors regarding the risk of enforcement action specific to the State of Ohio. For more information on federal enforcement and the risks associated with the U.S. cannabis regulatory environment generally, see without limitation, “Risk Factors – Risks Related to the Regulatory Environment.”
Ohio Licenses
In June 2019, we, through a subsidiary, entered into a management services agreement with FBS OH, a licensed medical marijuana processor in Ohio. In July 2021, the licensed medical marijuana processor received authorization to commence operation at the processing facility. We and the licensed medical marijuana processor have applied for a change of ownership to state regulators for the licensed medical marijuana processor to become a subsidiary us. In August 2021, we closed on the acquisition of FBS OH.
70
In July 2021, we acquired OhiGrow, a licensed marijuana cultivator, inclusive of an approximately 10,000 sq. ft. cultivation facility and 1.35 acres of land.
License and Regulations
To be considered for approval of a processing license, the applicant must complete all mandated requirements. To obtain a Certificate of Operation for a processing facility, the prospective licensee must be capable of operating in accordance with Chapter 3796 of the Revised Code, the Medical Marijuana Control Program. Certificates of Operation carry one-year terms. Following issuance of a Certificate of Operation, the provisionally licensed processor will be authorized to manufacture and produce medical cannabis products.
Reporting
Ohio uses the METRC system as its seed-to-sale tracking system. Licensees are required to use METRC to push data to the State of Ohio to meet all of the reporting requirements. When the provisionally licensed processor is operational, it intends to implement its tracking system to comply with the State of Ohio’s tracking and reporting requirements.
Storage and Security
All licensees must have a security system that remains operational at all times and that uses commercial grade equipment to prevent and detect diversion, theft or loss of medical cannabis, including:
| • | A perimeter alarm; |
| • | Motion detectors; and |
| • | Duress and panic alarms. |
Video cameras must be installed at the processing facility and directed at all approved safes, approved vaults, cannabis sales areas, and any other area where plant material, medical cannabis extract, or medical cannabis products are being processed, stored or handled. Video surveillance must take place 24 hours a day, 7 days a week. Recordings from all video cameras must be readily available for immediate review by regulating and law enforcement with jurisdiction upon request and must be retained for at least 45 days.
There are no current incidences of non-compliance, citations or notices of violation outstanding which have an impact on our business activities or operations in the State of Ohio. Notwithstanding the foregoing, like all businesses we may from time-to-time experience incidences of non-compliance with applicable rules and regulations in the states in which we operate, including the State of Ohio, and such non-compliance may have an impact on our business activities or operations in the state. However, we take steps to minimize, disclose and remedy all incidences of non-compliance which may have an impact on our licenses, business activities or operations in all states in which we operate, including the State of Ohio. We are not aware of any incidence of non-compliance, citations or notices of violation received by the provisionally licensed medical marijuana processor that will be operated by us pursuant to the applicable management services agreement or the failure of the provisionally licensed medical marijuana processor to comply with applicable licensing requirements and the regulatory framework enacted by the State of Ohio.
71
Pennsylvania
Pennsylvania Regulatory Landscape
The Pennsylvania Medical Marijuana Act (the PAMMA) was signed into law on April 17, 2016 and originally provided access to Pennsylvania residents with one of 17 qualifying conditions, including epilepsy, chronic pain, and post-traumatic stress disorder. Retail sales began in February 2018. The Commonwealth of Pennsylvania, which consists of nearly 13 million residents and qualifies as the fifth largest population in the U.S., operates as a high-barrier market with very limited market participation. The PAMMA authorizes only a maximum of 25 grower/processing permits and 50 dispensary permits. As part of “Phase 1” of the Commonwealth of Pennsylvania’s permitting process in 2017, the Pennsylvania Department of Health (the PA DOH) which administers the Commonwealth of Pennsylvania’s Medical Marijuana Program, originally awarded only 12 grower/processing permits and 27 dispensary permits. Subsequently, in 2018, PA DOH conducted “Phase 2” of the permitting process, during which it awarded the remaining 13 grower/processing permits and 23 dispensary permits authorized under the PAMMA. In July of 2019, the PA DOH expanded the list of qualifying medical conditions to include anxiety disorders and Tourette syndrome, increasing the number of qualifying conditions to 23.
We (through our Pennsylvania subsidiaries) are in compliance with applicable licensing requirements and the regulatory framework enacted by the Commonwealth of Pennsylvania.
To the knowledge of management, there have not been any statements or guidance made by federal authorities or prosecutors regarding the risk of enforcement action specific to the Commonwealth of Pennsylvania. For more information on federal enforcement and the risks associated with the U.S. cannabis regulatory environment generally, see without limitation, “Risk Factors – Risks Related to the Regulatory Environment.”
Pennsylvania Licenses
We, through certain subsidiaries, hold six dispensary permits, allowing for up to 18 medical marijuana retail dispensary locations in applicable regions within the Commonwealth of Pennsylvania. As of the date of this prospectus, we operate 18 medical cannabis dispensaries in the Commonwealth of Pennsylvania, located in or around the cities of Ardmore, Bethlehem, Bristol, Colwyn, Easton, Hazleton, Irwin, Johnstown, Philadelphia (three locations), Reading, Pittsburgh, Pottsville, Scranton (two locations), Stroudsburg, and West Chester. All the dispensaries operate under our BEYOND/HELLO™ brand.
All dispensaries must register with the PA DOH. Registration certificates are valid for a period of one year and are subject to annual renewals after required fees are paid and the business remains in good standing. Renewal requests are typically communicated through email and include a renewal form. Provided that the requisite renewal fees are paid, the renewal application is submitted in a timely manner, and there are no material violations noted against the applicable license, we would expect our Pennsylvania subsidiaries to receive the applicable renewed license in the ordinary course of business. However, any unexpected delays or costs associated with the licensing renewal process could impede the ongoing or planned operations and could have a material adverse effect on our business, financial condition, results of operations or prospects.
License and Regulations
Each retail dispensary license permits the holder to purchase marijuana and marijuana products from grower/processing facilities and allows the sale of marijuana and marijuana products to registered patients.
Site-Visits & Inspections
All licensed dispensary locations must be inspected and approved by the PA DOH before commencing live operations. Thereafter, dispensaries are subject to PA DOH inspection, whether with or without notice.
Reporting Requirements
The Commonwealth of Pennsylvania uses MJ Freeway as a T&T system for seed-to-sale reporting. Individual permittees are required to use MJ Freeway to push data to the Commonwealth of Pennsylvania to meet all reporting requirements. Our subsidiaries use MJ Freeway as our in-house computerized seed-to-sale software, which integrates with the Commonwealth’s MJ Freeway program and captures the required data points for cultivation, manufacturing and retail as required in the Pennsylvania medical marijuana laws and regulations.
72
Storage and Security
All dispensaries are required to have a locked limited access area for the storage of medical marijuana that is expired, damaged, deteriorated, mislabeled, contaminated, recalled or whose containers or packages have been opened or breached until such product is returned to the grower/processor. Our subsidiary dispensaries maintain security systems with professional monitoring, 24-hours a day and seven days a week, and fixed cameras on the interior and exterior of the facilities in a manner consistent with Pennsylvania law. Data for surveillance systems is stored for a period of 4 years in a readily available format for investigative purposes.
We are in compliance with the laws of the Commonwealth of Pennsylvania and the related cannabis licensing framework. There are no current incidences of non-compliance, citations or notices of violation outstanding which have an impact on our licenses, business activities or operations in the Commonwealth of Pennsylvania. Notwithstanding the foregoing, like all businesses we may from time-to-time experience incidences of non-compliance with applicable rules and regulations in the states in which we operate, including the Commonwealth of Pennsylvania, and such non-compliance may have an impact on our licenses, business activities or operations in the applicable state. However, we take steps to minimize, disclose and remedy all incidences of non-compliance which may have an impact on our licenses, business activities or operations in all states in which we operate, including the Commonwealth of Pennsylvania. See “Regulatory Framework – Compliance.”
Virginia
Virginia Regulatory Landscape
Virginia legalized medical marijuana for the treatment of glaucoma and cancer as part of sweeping changes to it’s drug laws in 1979. In 2015, the Commonwealth of Virginia passed legislation that provided an affirmative defense for the possession of CBD or THC-A oil pursuant to a valid written certification for patient use of the oils from a physician to alleviate intractable epilepsy but made no provision for a patient to acquire these substances.
In 2017, Virginia commenced a program that allows registered patients to access and use cannabis oil. The enabling legislation also authorized the Commonwealth of Virginia to issue five (5) pharmaceutical processor licenses that allow the holder thereof to cultivate, manufacture and dispense medical cannabis from a single location. Pharmaceutical processor licenses are issued by the Virginia Board of Pharmacy (the VA BOP) on a regional (restricted) basis such that only one licensee is permitted to operate in each of five (5) defined Health Service Areas across the Commonwealth of Virginia. In 2018, the Commonwealth of Virginia expanded the program to allow eligible practitioners to recommend medical cannabis to patients suffering from any diseases or conditions. Additionally, the law required information about dispensed oils to be reported in the Prescription Monitoring Program (the PMP) and mandated that practitioners check the PMP prior to issuing patient certifications. In March 2020, the Commonwealth of Virginia further expanded the medical marijuana program by authorizing licensees to add 5 off-site dispensing locations within their Health Service Area, replacing definitions of CBD oil and THC-A oil with a single definition of “cannabis oil,” and removing certain restrictions applicable to oil potency. The March 2020 legislation became effective on July 1, 2020, and a subset of the regulations implementing the March 2020 legislation became effective on September 30, 2020 with the remaining provisions taking effect on February 8, 2021.
We, through Dalitso, are in compliance with applicable licensing requirements and the regulatory framework enacted by the Commonwealth of Virginia.
To the knowledge of management, there have not been any statements or guidance made by federal authorities or prosecutors regarding the risk of enforcement action specific to the Commonwealth of Virginia. For more information on federal enforcement and the risks associated with the U.S. cannabis regulatory environment generally, see without limitation, “Risk Factors – Risks Related to the Regulatory Environment.”
73
Virginia Licenses
Dalitso currently holds a conditional approval from the VA BOP to cultivate, manufacture, and dispense medical cannabis in Health Service Area II, which covers Loudoun, Fairfax, Arlington and Prince William Counties. As of the date of this prospectus, we operate two medical cannabis dispensaries and engage in cultivation and manufacturing operations in the Commonwealth of Virginia at our facility located in Prince William County. The dispensaries operate under our BEYOND/HELLO™ brand. We are permitted to open four additional dispensaries in Health Service Area II of the Commonwealth of Virginia.
License and Regulations
Pharmaceutical processors are required to designate a “Pharmacist in Charge” to manage its operation, and to have a supervising pharmacist on duty during all hours of operation. Numerous tasks that involve handling cannabis oil must be performed by a pharmacist or a pharmacy technician acting under a pharmacist’s supervision. Those tasks include, for example, labeling oils, removing oils from inventory, measuring oils for dispensing, and selling oils. Pharmacists and pharmacy technicians must have current licenses, and the ratio of pharmacists to pharmacy technicians cannot exceed 6-to-1 (prior to recent legislative changes, the ratio was 4-to-1). The VA BOP has also imposed certain educational requirements cultivation and manufacturing processes, as well as significant employee training, both upon hire and on a regular, continuous basis thereafter.
A pharmaceutical processor must operate for a minimum of 35 hours per week. Access to the facility is limited to employees performing their job duties (who must display ID badges) and patients (and their parents or guardians). Pharmacists are required to counsel registered patients (and parents/legal guardians as applicable) about medical cannabis products, including (but not limited to) proper use and storage.
As a general matter, the VA BOP prohibits use of pesticides in cultivation (with some exception) and mandates that extraction methods meet industry standards. All medical cannabis products must be branded, tested, and registered with the VA BOP before they can be dispensed. Medical cannabis products must be packaged in child-resistant containers (with limited exceptions), properly labeled, and tested (at the batch level) by qualified independent laboratories. In the course of dispensing operations, a pharmacist or pharmacy technician must check patient identification and certification before dispensing any medical cannabis product(s) and detailed records about all dispensing transactions (along with other records) must be maintained for a period of not less than three years, and the licensee must implement a stringent quality assurance program designed to prevent dispensing errors. Expired, damaged or otherwise waste cannabis plant material and products must be stored in a secure manner until properly destroyed.
Storage and Security
Pharmaceutical processors are subject to a number of inventory and security requirements under Virginia law and VA BOP regulations. For example, they must: conduct an initial comprehensive inventory; establish ongoing inventory controls and procedures; conduct weekly inventory reviews; and prepare an annual inventory report (inventory records must be made available to the VA BOP and its agents for inspection and copying). All parts of the cannabis plant and medical cannabis products (whether finished or in process) must be stored in a locked and secured vault or safe with appropriate access limitations and the pharmaceutical processor must maintain a sophisticated security system that satisfies VA BOP-mandated criteria. Cannabis and cannabis products must be stored in a generally clean, sanitary, and secure area, and storage areas and related procedures are subject to a number of VA BOP requirements. Pharmaceutical processors must install and maintain a video surveillance system that captures all areas where cannabis and cannabis products (whether finished or in process) are handled or stored. Surveillance recordings must be stored for 30 days and made available for the VA BOP’s immediate review upon request. All security breaches or other events must be promptly reported to the VA BOP.
Site-Visits & Inspections
At all times, pharmaceutical processing facilities are subject to inspection by the VA BOP and certain other authorized agencies, and pharmacists and pharmacy technicians on-site must be prepared to present their current license or registration to the VA BOP or its agencies during inspections.
74
Reporting Requirements
Pharmaceutical processors are required to maintain an electronic tracking system comprised of an electronic radio-frequency identification seed-to-sale system capable of tracking cannabis from either the seed or immature plant stage until the cannabis oils are sold to a registered patient, parent, or legal guardian or until the cannabis, including the seeds, parts of plants, and extracts, are destroyed. The electronic tracking system shall include, at a minimum, a central inventory management system and standard and ad hoc reporting functions as required by the VA BOP (and must otherwise satisfy recordkeeping laws, rules and regulations).
We are in compliance with the laws of the Commonwealth of Virginia and the related cannabis licensing framework. There are no current incidences of non-compliance, citations or notices of violation outstanding which have an impact on our licenses, business activities or operations in the Commonwealth of Virginia. Notwithstanding the foregoing, like all businesses we may from time-to-time experience incidences of non-compliance with applicable rules and regulations in the states in which we operate, including the Commonwealth of Virginia, and such non-compliance may have an impact on our licenses, business activities or operations in the applicable state. However, we take steps to minimize, disclose and remedy all incidences of non-compliance which may have an impact on our licenses, business activities or operations in all states in which we operate, including the Commonwealth of Virginia. See “Regulatory Framework – Compliance.”
Compliance
With the oversight and under the direction of the Vice President of Compliance, our legal department oversees, maintains, and implements a compliance program in conjunction with our operations in each jurisdiction. In addition to our legal and compliance departments, we have local regulatory/compliance counsel engaged in every jurisdiction (state and local) in which we operate. Together with on-site management in each jurisdiction, our legal and compliance departments are responsible for ensuring operations and employees strictly comply with applicable laws, regulations, and licensing conditions and ensure that operations do not endanger the health, safety or welfare of the community. We designate a duly qualified and experienced manager at each location who is responsible to coordinate with operational units within each facility (to extent applicable) to ensure that the operation and all employees are following and complying with our written security procedures and all regulatory compliance standards.
In conjunction with our human resources and operations departments, the compliance and quality departments help oversee and implement training for all employees, including on the following topics:
| • | compliance with state and local laws; |
| • | cultivation/manufacturing/dispensing/transport procedures (as applicable); |
| • | security and safety policies and procedures; |
| • | inventory control, T&T, seed-to-sale, and point of sale systems training (as applicable); and |
| • | quality control. |
Our compliance program emphasizes security and inventory control to ensure strict monitoring of cannabis (including living plants and harvested plant material) and cannabis product inventory. Only authorized, properly trained employees are allowed to access our inventory management systems.
Our compliance department and legal team, comprised of in-house and local outside counsel, monitors all compliance notifications from the regulators and inspectors in each market, timely resolving any issues identified. The team maintains records of all compliance notifications received from the state regulators or inspectors and how and when the issue was resolved. We have created comprehensive standard operating procedures that include detailed descriptions and instructions for receiving shipments of inventory, inventory tracking, recordkeeping and record retention practices related to inventory, as well as procedures for performing inventory reconciliation and ensuring the accuracy of inventory tracking and recordkeeping. We maintain accurate records of our inventory at all licensed facilities. Adherence to our standard operating procedures is mandatory and ensures that our operations are compliant with the rules set forth by the applicable state and local laws, regulations, ordinances, licenses and other requirements. Training on these standard operating procedures is mandatory by all employees and defined by function and role.
75
We have developed and continue to refine a robust compliance program designed to ensure operational and regulatory requirements continue to be satisfied and have worked closely with experts and outside counsel to develop compliance procedures intended to assist us in monitoring compliance with U.S. state law on an ongoing basis. We will continue to work closely with outside counsel and other compliance experts to further develop, enhance and improve our compliance and risk management and mitigation processes and procedures in furtherance of continued compliance with the complex regulatory frameworks of the states where we operate. The internal compliance program currently in place includes continued monitoring by managers and executives of us and our subsidiaries to ensure that all operations conform to and comply with required laws, rules, regulations and SOPs. We further require our operating subsidiaries to report and disclose all instances of non-compliance, regulatory, administrative, or legal proceedings that may be initiated against them.
Notwithstanding the foregoing, from time to time, as with all businesses and all rules, it is anticipated that we, through our subsidiaries and establishments to which we provide operational support, may experience incidences of non-compliance with applicable rules and regulations, which may include minor matters such as:
| • | staying open slightly too late due to an excess of customers at stated closing time; |
| • | minor inventory discrepancies with regulatory reporting software; |
| • | missing fields in regulatory reports; |
| • | missing fields entries in a visitor log; |
| • | cleaning schedules not available on display; |
| • | educational materials and/or interpreter services not available in a sufficient number of languages; |
| • | updated staffing plan not immediately available on site; |
| • | improper illumination of external signage; |
| • | marijuana infused product utensils improperly stored; |
| • | partial obstruction of camera views; and/or |
| • | supplemental use of onsite surveillance room (i.e., storage). |
In addition, either on an inspection basis or in response to complaints, such as from neighbors, customers or former employees, State or local regulators may, among other things, issue investigatory- or demand-type letters, give warnings to or cite businesses which we operate or for which we provide operational support for violations, including those listed above. Such regulatory actions could lead to a requirement or directive to submit and thereafter comply with (for example) a plan of correction. Depending on the jurisdiction, it is also possible regulators may assess penalties and/or amendments, suspensions or revocations of licenses or otherwise take action that may impact our licenses, business activities, operational support activities or operations.
To minimize opportunities for non-compliance and among other measures, we have implemented regular compliance reviews to ensure our subsidiaries and establishments to which we provide operational support are operating in conformance with applicable State and local cannabis rules and regulations. In the event non-compliance is discovered, during a compliance review or otherwise, we will promptly remedy the same, including by self-reporting to applicable State and local cannabis regulators as and when required by law and will make all requisite and appropriate public disclosures of non-compliance, citations, notices of violation and the like which may have an impact on its licenses, business activities, operational support activities or operations.
76
State License Renewal Requirements
For each of our provisional and operational licenses, the states impose strict license renewal requirements that vary state by state. We generally must complete the renewal application process within a prescribed period of time prior to the expiration date and pay an application fee. The state licensing body can deny or revoke licenses and renewals for a variety of reasons, including but not limited to (a) submission of materially inaccurate, incomplete or fraudulent information, (b) failure of the company or any of its directors or officers to comply, or have a history of non-compliance, with any applicable law or regulation, including laws relating to minimum age of customers, safety and non-diversion of cannabis or cannabis products, taxes, child support, workers compensation and insurance coverage, or otherwise remain in good standing (c) failure to submit or implement a plan of correction for any identified violation, (d) attempting to assign registration to another entity without state approval, (e) insufficient financial resources, (f) committing, permitting, aiding or abetting of any illegal practices in the operation of a facility, (g) failure to cooperate or give information to relevant law enforcement related to any matter arising out of conduct at a licensed facility and (h) lack of responsible operations, as evidenced by negligence, disorderly or unsanitary facilities or permitting a person to use a registration card belonging to another person. Certain jurisdictions also require licensees to attend a public hearing or forum in connection with their license renewal application.
Human Capital Resources
As of June 30, 2022, we had 1,538 employees. We are committed to hiring talented individuals and maximizing individual potential, while fostering growth and career advancement. Our goal is to use the highest standards in attracting the best talent, offering competitive compensation, as well as implementing best practices in evaluating, recruiting and onboarding our human capital.
Our employees are split across the Company as follows:
| Corporate: | 158 |
| Retail: | 874 |
| Manufacturing: | 506 |
| Total: | 1,538 |
As of June 30, 2022, approximately 100 employees who work in our Scranton, Pennsylvania grower processor facility and approximately 19 employees who work in Scranton, Stroudsburg and Bethlehem, Pennsylvania dispensaries are covered by a collective bargaining agreement with United Food and Commercial Workers Union, Local 1776KS. We did not experience any union work stoppages in 2021 and we consider our relationship with our employees to be good.
Description of Property
Our corporate headquarters are located in Boca Raton, Florida. In addition, we have retail, cultivation or manufacturing locations in the following states: Pennsylvania, Illinois, Virginia, California, Ohio, Nevada and Massachusetts. Most of our locations are leased from third parties. We believe that our facilities and expansion plans are adequate for our current and anticipated needs. Details relating to the properties in each state are within the Business section above under “Current Operations.”
Legal Proceedings
There are no actual or to our knowledge contemplated legal proceedings that we or our subsidiaries are a party to or to any of our subsidiaries’ property is the subject matter.
Available Information
We maintain a website at http://www.jushico.com. Upon the date the registration statement of which this prospectus forms a part is declared effective, we will become subject to the informational and periodic reporting requirements of the Exchange Act, and will thereafter file periodic reports and other information with the SEC. When filed, such period reports, such as our Annual Report on Form 10-K (which will include our audited financial statements), Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and amendments to reports filed or furnished pursuant to Sections 13(a) and 15(d) of the Exchange Act, will be available on our website, free of charge, as soon as reasonably practicable after we electronically file such reports with, or furnish those reports to, the SEC. You may also read and copy these reports, proxy statements and other information on the SEC’s website at www.sec.gov. Additional information relating to the Company is also available under the Company’s profile under SEDAR at www.sedar.com.
77
MARKET PRICE AND DIVIDENDS ON COMMON EQUITY AND RELATED STOCKHOLDER MATTERS
Our Subordinate Voting Shares began trading on the Canadian Securities Exchange under the symbol “JUSH” on December 9, 2019 and began trading on the OTC Markets under the symbol “JUSHF” on September 23, 2019. Any over-the-counter market quotations from the OTC Markets reflect inter-dealer prices, without retail mark-up, mark-down or commission and may not necessarily represent actual transactions.
Holders of Subordinate Voting Shares
As of June 30, 2022, we had approximately 317 shareholders of record of 194,653,132 issued and outstanding Subordinate Voting Shares, and no Multiple Voting Shares or Super Voting Shares issued and outstanding. In calculating the number of shareholders, we consider clearing agencies and security position listings as one shareholder for each agency or listing.
Dividends
We have not declared distributions on Subordinate Voting Shares, Multiple Voting Shares or Super Voting Shares in the past. We currently intend to reinvest all future earnings to finance the development and growth of our business. As a result, we do not intend to pay dividends on Subordinate Voting Shares, Multiple Voting Shares, Super Voting Shares or Preferred Shares in the foreseeable future. Any future determination to pay distributions will be at the discretion of the board of directors and will depend on the financial condition, business environment, operating results, capital requirements, any contractual restrictions on the payment of distributions and any other factors that the board of directors deems relevant.
2019 Equity Incentive Plan
The following table sets forth securities authorized for issuance under our 2019 Equity Incentive Plan, as amended (referred to herein as the 2019 Equity Incentive Plan or the Plan), as of December 31, 2021:
| Plan Category | Number of securities to be issued upon exercise of outstanding options, warrant and rights | Weighted- average exercise price of outstanding options, warrants and rights | Number of securities remaining available for future issuance under equity compensation plans | |||||||||
| Equity compensation plans approved by security holders(1)(2) | 20,429,120 | $ | 3.20 | 7,904,819 | ||||||||
| Equity compensation plans not approved by security holders | — | — | — | |||||||||
| Total | 20,429,120 | $ | 3.20 | 7,904,819 | ||||||||
(1) The maximum number of Subordinate Voting Shares issuable under the 2019 Equity Incentive Plan of the Company as of December 31, 2021 was 31,060,251, representing 17% of the number of the issued and outstanding Subordinate Voting Shares, which we refer to in this prospectus as the Outstanding Share Number.
(2) Excludes 2,859,151 Subordinate Voting Shares issued related to unvested restricted stock awards.
As of December 31, 2021, the following Awards were issued under the 2019 Equity Incentive Plan: (i) a total of 20,429,120 Options, representing approximately 11% of the then Outstanding Share Number; and (ii) a total of 2,859,151 unvested RSAs (as defined below), representing less than 2% of the then Outstanding Share Number. As of December 31, 2021, an aggregate of 7,904,819 Subordinate Voting Shares remained available for issuance under the 2019 Equity Incentive Plan, representing approximately 4% of the then Outstanding Share Number.
78
Executive Officers and Directors
Our executive officers and directors, their positions and their ages as of June 30, 2022 are set forth below:
| Name | Age | Position(s) | ||
| Executive Officers | ||||
| James Cacioppo | 60 | Chairman of the Board of Directors and Chief Executive Officer | ||
| Louis Jon Barack | 45 | President, Corporate Secretary and Interim Chief Financial Officer | ||
| Leonardo Garcia-Berg | 58 | Chief Operations Officer | ||
| Directors | ||||
| Peter Adderton | 55 | Director | ||
| Benjamin Cross | 67 | Director | ||
| Marina Hahn | 64 | Director | ||
| Stephen Monroe | 62 | Director |
Executive Officers
James Cacioppo has served as our Chief Executive Officer and Chairman since he co-founded us in 2018. Prior to founding Jushi, from 1993 to 2018, Mr. Cacioppo spent over two decades managing the business and allocating capital in senior management positions at several large hedge funds. Since January 2006, Mr. Cacioppo was Co-Founder and Managing Partner of One East Partners ($2.3 billion (peak assets under management)). Previously, from 2000 to 2006, Mr. Cacioppo served as President and Co-Portfolio Manager of Sandell Asset Management ($5.0 billion (peak assets under management)) and from 1995 to 2000, he served as Head of Distressed Debt for Halcyon Management, a global investment firm with over $9 billion in assets. Mr. Cacioppo earned his BA from Colgate University and his MBA from Harvard University. We believe Mr. Cacioppo is qualified to serve on our board of directors due to his managerial, start-up, financial and investing experience.
Louis Jon Barack co-founded us in 2018 and has served as our Corporate Secretary since such time, as our President since December of 2019 and as our Interim Chief Financial Officer since July of 2022. Mr. Barack brings extensive financial and cannabis industry investing experience (both public and private) to his role as President, Corporate Secretary and Interim Chief Financial Officer. Mr. Barack spent over ten years in investments at various hedge funds, including working from 2013 to 2018 at One East Capital Advisors where he focused on cannabis investments. Mr. Barack earned his BA from Princeton University and his JD/MBA from Northwestern University.
Leonardo Garcia-Berg has served as our Chief Operations Officer since 2021. Before joining Jushi, Mr. Garcia-Berg served in numerous roles at Anheuser-Busch InBEV (AB InBEV) starting from January 2010 to October 2020. As the Global Director of Value Creation, Global Procurement Officer for AB InBev, Mr. Garcia-Berg led strategies focused on improving manufacturing, logistics, sourcing and operations across the company’s breweries worldwide. In addition to his positions at AB InBEV, he also served as an international consultant for McKinsey & Company, focusing on operational strategies, procurement, organizational transformations, along with supply chain and end-to-end process optimization solutions. Mr. Garcia-Berg received his Bachelor of Science degree in electrical engineering from Instituto Tecnológico de Buenos Aires in Argentina, and earned his MBA from The Wharton School of the University of Pennsylvania.
79
Directors
Peter Adderton has served as a member of our board of directors since June 2019. Starting from 2000, Mr. Adderton is a member of the board of directors and Founder of Boost Mobile, a wireless telecommunications brand based in Australia. Under his leadership, Boost Mobile USA was purchased by Nextel/Sprint and remains a wholly owned subsidiary of Nextel/Sprint. Prior to founding Boost Mobile, Peter founded Amp’d Mobile, a wireless company and Mandalay Digital, now Digital Turbine, a mobile solutions provider. At Mandalay Digital, Peter was the Company’s CEO and Director leading it to become a Nasdaq listed company. Peter graduated from Sydney Technical College. We believe Mr. Adderton is qualified to serve on our board of directors due to his operational and marketing expertise.
Benjamin Cross has served as a member of our board of directors since June 2019. Mr. Cross spent 20 years at Morgan Stanley in both their London and New York offices in the Commodities Division until his retirement in 2015 as a Managing Director at the firm. Prior to joining Morgan Stanley, Mr. Cross worked at Merrill Lynch and the commodities exchange. Mr. Cross earned his BS from Cornell University. Presently, Mr. Cross is a Board Advisor to Ursa Space, a geospatial intelligence firm with an emphasis in measuring global oil inventories and is a commodities reporter for TheStreet.com, which he has been a reporter since July 2015. We believe Mr. Cross is qualified to serve on our board of directors due to his extensive financial markets experience and commodities knowledge.
Marina Hahn has served as a member of our board of directors since May 2021. Since February 2020, Ms. Hahn serves as a consultant at Rotkaeppchen-Mumm, a German market leader in sparkling wines and spirits. Prior to serving as a consultant at Rotkaeppchen-Mumm, Ms. Hahn co-founded ZX Ventures, a growth arm of AB InBEV from April 2018 to January 2020. Prior to ZX Ventures, Ms. Hahn served as President of the Consumer Division at Flex Pharma, from August 2014 to November 2016, an innovative biotech formed as a result of a scientific breakthrough for athletes who suffer from muscle cramps. Ms. Hahn was a founder of SVEDKA Vodka (acquired by Constellation Brands, Inc.), an irreverent lifestyle brand where she originated the iconic spokesbot, SVEDKA_grl. Ms. Hahn is a graduate of Wellesley College. We believe Ms. Hahn is qualified to serve on our board of directors due to her consumer brand experience and prior board experience.
Stephen Monroe has served as a member of our board of directors since June 2019. Since 2016, Mr. Monroe has served as President and Managing Partner of Liquid Capital Alternative Funding, an asset-based lender. Prior to joining Liquid Capital Alternative Funding, Mr. Monroe served as National Sales Manager for Short Duration Products at JP Morgan Chase & Co.; and previously in a variety of senior management positions covering cash and short duration products at Barclays and the Royal Bank of Scotland. Mr. Monroe earned his BA from Williams College. We believe Mr. Monroe is qualified to serve on our board of directors due to his vast experience in financial markets and risk management.
Board Composition
Our Articles of Incorporation, as amended to date, which we refer to as our Articles, provide that so long as we are a public company, the minimum number of directors is three (3) and the number of directors is set by ordinary resolution. Each director holds office until the close of the next annual general meeting of shareholders, or until his or her successor is duly elected or appointed, unless his or her office is earlier vacated. The board of directors currently consists of five (5) directors, although the number of directors set by ordinary resolution is currently six (6) and the board of directors may fill this vacancy at its discretion. Our business and affairs are managed by or under the direction of the board of directors. Pursuant to our Mandate of the board of directors, the board of directors may establish one or more committees of the board of directors, however designated, and delegate to any such committee the full power of the board of directors, to the fullest extent permitted by law.
80
We are not currently subject to listing requirements of any national securities exchange that has requirements that a majority of the board of directors be “independent.” All but one of the five directors are considered to be independent under the CSA Guidelines and in accordance with National Instrument 52-110—Audit Committees (NI 52-110). A director is considered independent for the purposes of NI 52-110 if he or she has no direct or indirect “material relationship” with the issuer, where “material relationship” is defined as a relationship that could, in the view of the issuer’s board of directors, be reasonably expected to interfere with the exercise of a member’s independent judgement. Our independent directors are Peter Adderton, Benjamin Cross, Marina Hahn and Steve Monroe. Mr. Cacioppo is not independent, given that he is our Chief Executive Officer.
The board of directors holds regularly scheduled meetings and at such meetings our independent directors meet in executive session. The board of directors has not appointed a lead independent director; instead, the presiding director for each executive session is rotated among the chairs of our committees.
The board of directors held four meetings during the year ended December 31, 2021. In 2021, each person serving as a director attended at least 50% of the total number of meetings of our board of directors and the majority of any committee on which he or she served.
Board Committees
At present, the board of directors has three standing committees, the Audit Committee, the Nominating and Corporate Governance Committee, and the Compensation Committee. The charters for our committees set forth the scope of the responsibilities of that committee.
Audit Committee.
The board of directors established an audit committee (the Audit Committee:). The Audit Committee is currently comprised of three members: Stephen Monroe (Chair), Peter Adderton and Benjamin Cross. Each of the members of the Audit Committee meets the independence requirements pursuant to NI 52-110 and each is financially literate within the meaning of NI 52-110.
The Audit Committee operates pursuant to a written charter. The principal duties and responsibilities of the Audit Committee are to assist the board of directors to:
| • | Conduct such reviews and discussions with management and the external auditors relating to the audit and financial reporting as are deemed appropriate by the Audit Committee. |
| • | Assess the integrity of internal controls and financial reporting procedures of the Company and ensure implementation of such controls and procedures. |
| • | Review the interim and annual financial statements and management’s discussion and analysis of the Company’s financial position and operating results and in the case of the annual financial statements and related management’s discussion and analysis, report thereon to the board of directors for approval of same. |
| • | Select and monitor the independence and performance of the Company’s external auditors, including attending private meetings with the external auditors and reviewing and approving all renewals or dismissals of the external auditors and their remuneration. |
| • | Provide oversight of all disclosure relating to, and information derived from, financial statements and management’s discussion and analysis. |
In fulfilling its responsibilities, the Audit Committee meets regularly with our auditor and key management members.
The Audit Committee has access to all of our books, records, facilities and personnel and may request any information as it may deem appropriate. It also has the authority to retain and compensate special legal, accounting, financial and other consultants or advisors to advise the Audit Committee. The Audit Committee is responsible for the pre-approval of all non-audit services to be provided by our auditors.
81
Compensation Committee
The board of directors has established a compensation committee (the Compensation Committee). The Compensation Committee is currently comprised of three members: James Cacioppo (Chair), Benjamin Cross and Stephen Monroe. All of the members of the Compensation Committee other than Mr. Cacioppo are independent for purposes of NI 52-110. Mr. Cacioppo recuses himself on compensation committee-related matters relating to himself.
The Compensation Committee operates pursuant to a written charter. The principal duties and responsibilities of the Compensation Committee are to assist the board of directors to:
| • | Review and approve annually the corporate goals and objectives applicable to the compensation of the CEO; |
| • | Evaluate, at least annually, the CEO’s performance in light of the goals and objectives set for the CEO; |
| • | Determine and make recommendations to the board of directors with respect to the CEO’s compensation level (both cash and equity-based). In determining the long-term incentive component of the CEO’s compensation, the Committee may consider our performance, shareholder returns, the value of similar incentive awards given to CEOs at comparable companies and the awards given to the Company’s CEO in past years. |
| • | Make recommendations to the board of directors regarding the compensation of non-CEO senior executive officers and the directors. |
| • | Review and make recommendations to the board of directors regarding incentive compensation plans and equity-based plans, and where appropriate or required, recommend for approval by the shareholders of the Company. |
| • | Review and discuss with management our executive compensation disclosure to be included in our management information circular and any other disclosure with respect to executive compensation to be included in any other public disclosure documents of Jushi. |
| • | Review and make recommendations to the board of directors regarding any employment agreements and any severance arrangements or plans, including any benefits to be provided in connection with a change in control, for the CEO and other executive officers. |
The Compensation Committee also has the authority to retain and compensate a compensation consultant, special legal, and other consultants or advisors.
Nominating and Corporate Governance Committee
The board of directors has established a nominating and corporate governance committee (the Nominating and Corporate Governance Committee). The Nominating and Corporate Governance Committee is currently comprised of three members: James Cacioppo (Chair), Benjamin Cross and Steve Monroe. All of the members of the Nominating and Corporate Governance Committee other than Mr. Cacioppo are independent for purposes of NI 52-110.
The Nominating and Corporate Governance Committee operates pursuant to a written charter. The principal duties and responsibilities of the Nominating and Corporate Governance Committee are to assist the board of directors to:
| • | Identify individuals qualified to become members of the board of directors. |
| • | Recommend director nominees for each annual meeting of the Company’s shareholders and director nominees to fill any vacancies that may occur between meetings of shareholders. |
| • | Be aware of the best practices in corporate governance and develop and recommend to the board of directors a set of corporate governance standards to govern the board of directors, its committees, the Company and its employees in the conduct of the business and affairs of the Company. |
| • | Consider the diversity of the board of directors. |
| • | Develop and oversee the annual board of directors and committees of the board of directors evaluation process. |
82
Board Oversight of Enterprise Risk
One of the key functions of our board of directors is informed oversight of our risk management process. The board of directors does not have a standing risk management committee and instead administers this oversight function directly through the board of directors as a whole, as well as through various standing committees of our board of directors that address risks inherent in their respective areas of oversight. In particular, our board of directors is responsible for monitoring and assessing strategic risk exposure and the Audit Committee will have the responsibility to consider and discuss our major financial risk exposures and the steps our management has taken to monitor and control these exposures, including guidelines and policies to govern the process by which risk assessment and management is undertaken. The Audit Committee also monitors compliance with legal and regulatory requirements.
Board Leadership
The board of directors has no policy regarding the need to separate or combine the offices of Chairman of the board of directors and Chief Executive Officer and instead the board of directors remains free to make this determination from time to time in a manner that seems most appropriate for Jushi. The positions of Chairman of the board of directors and Chief Executive Officer are currently held by James Cacioppo. The board of directors believes the Chief Executive Officer is in the best position to direct the independent directors’ attention on the issues of greatest importance to Jushi and its shareholders. As a result, we do not currently have a lead independent director. Our overall corporate governance policies and practices combined with the strength of our independent directors and our internal controls minimize any potential conflicts that may result from combining the roles of Chairman and Chief Executive Officer.
Corporate Governance Principles and Code of Ethics
The board of directors is committed to sound corporate governance principles and practices. In order to clearly set forth our commitment to conduct our operations in accordance with our high standards of business ethics and applicable laws and regulations, the board of directors also adopted a Code of Business Conduct and Ethics, which we refer to as our Code of Ethics, which is applicable to all directors, officers and employees. A copy of the Code of Ethics is available on our corporate website at https://ir.jushico.com/.
2021 Director Compensation
Starting in the fourth quarter of 2021, each non-employee director is paid an annual retainer of $65,000 per annum in cash or equity (stock options or restricted stock) at the Company’s discretion. Previous to that, the annual retainer was $50,000. Additionally, each non-employee director historically has been provided an annual grant of restricted stock or stock options each year. Directors are also reimbursed for any out-of-pocket travel expenses incurred in order to attend meetings of the board of directors, meetings of committees of the board of directors or meetings of our shareholders.
The following table sets forth information regarding compensation awarded to, earned by or paid to our non-employee directors in connection with their service for the year ended December 31, 2021. We do not pay any compensation to our Chief Executive Officer, who is also the Chair of the board of directors, in connection with his service on our board of directors. See “Executive Compensation” for a discussion of the compensation of Mr. Cacioppo.
| Name | Fees earned or paid in cash ($)(1) | Stock Awards ($)(2) | Option awards ($)(2) | Total ($) | ||||||||||||
| Peter Adderton | $ | — | $ | 48,601 | (3)(4) | $ | 138,360 | (5) | $ | 186,961 | ||||||
| Benjamin Cross | $ | — | $ | 48,601 | (3)(4) | $ | 138,360 | (5) | $ | 186,961 | ||||||
| Marina Hahn | $ | 33,858 | $ | 98,962 | (3) | $ | 46,120 | (5) | $ | 178,940 | ||||||
| Stephen Monroe | $ | — | $ | — | $ | 185,765 | (6) | $ | 185,765 | |||||||
| (1) | Represents amount earned or paid in cash for service as a director during fiscal year 2021. |
| (2) | Represents the grant date fair value of restricted stock and/or option awards granted in fiscal year 2021 in accordance with ASC 718, Compensation—Stock Compensation. Such grant date fair values do not take into account any estimated forfeitures related to service-based vesting. The assumptions used in calculating the grant date fair value of the awards reported in these columns are set forth in Note 15 - Share-Based Compensation and Other Benefits of our audited consolidated financial statements included elsewhere in this prospectus. As of December 31, 2021, the following number of option shares and shares of restricted stock were outstanding for each of our non-employee directors in the aggregate: Mr. Adderton, 60,000 option shares and 109,080 shares of restricted stock; Mr. Cross, 60,000 option shares and 102,513 shares of restricted stock; Ms. Hahn 20,000 option shares and 17,301 shares of restricted stock; and Mr. Monroe, 73,952 option shares and 150,484 shares of restricted stock. |
| (3) | Vested 25% on the grant date and 25% each quarter thereafter. |
| (4) | Represents restricted stock granted in lieu of payment of the cash retainer. |
| (5) | Vested fully on July 1, 2022, with an exercise price of $3.91 per share. |
| (6) | Represents (i) $47,405 of options that vested 25% on the grant date and 25% each quarter thereafter, with an exercise price of $5.71 per share, and such options were granted in lieu of payment of the cash retainer, and (ii) $138,360 of options that that vest fully on July 1, 2022, with an exercise price of $3.91 per share |
Involvement in Certain Legal Proceedings
On April 21, 2021, the Company announced it had applied to the Ontario Securities Commission (the OSC), as principal regulator of the Company, for the imposition of a management cease trade order (the MCTO) under National Policy 12-203 – Management Cease Trade Orders because, due to the Company’s auditor not being able to complete its annual audit procedures in a timely manner, the Company would not be able to file its audited annual financial statements for the year ended December 31, 2020, the related management’s discussion and analysis, related Chief Executive Officer and Chief Financial Officer certificates and annual information form for the year ended December 31, 2020 (the Required Filings) before the required deadline of April 30, 2021. On May 3, 2021, the OSC issued the MCTO. The MCTO restricted the trading of securities of the Corporation by the Chief Executive Officer and Chief Financial Officer of the Corporation until the Required Filings were made. The Required Filings were made on June 9, 2021 and the MCTO was automatically revoked. All of the Company’s current directors and officers, except Leonardo Garcia-Berg and Marina Hahn, were in place on the date when the MCTO was issued on May 3, 2021. Mr. Garcia-Berg was hired by the Company on May 24, 2021 while the MCTO was in place but not appointed as an Executive Officer until June 30, 2021 after the MCTO was revoked.
83
On February 22, 2022, Jushi Europe filed a notice of over-indebtedness with the Swiss courts, and its auditor followed with a notice of over-indebtedness on March 11, 2022. As of the date of this prospectus, both notices are pending with the Swiss courts. Mr. Cacioppo is an officer/director of Jushi Europe. See “Business-Current Operations-Business in Europe” for more information.
84
This section discusses the material components of the executive compensation program for our executive officers who are named in the “Summary Compensation Table” below. As an emerging growth company, we have opted to comply with the executive compensation disclosure rules applicable to “smaller reporting companies” as such term is defined in the rules promulgated under the Securities Act, which require compensation disclosure for our principal executive officer and our two other most highly compensated executive officers. Such “named executive officers” as of December 31, 2021 and their positions were:
| • | James Cacioppo, Chief Executive Officer and Chairman |
| • | Louis Jon Barack, President, Corporate Secretary and Interim Chief Financial Officer |
| • | Leonardo Garcia-Berg, Chief Operations Officer |
This discussion may contain forward-looking statements that are based on our current plans, considerations, expectations and determinations regarding future compensation programs. Actual compensation programs that we adopt following the completion of this offering may differ materially from the currently planned programs summarized in this discussion.
Compensation Philosophy
The board of directors has not adopted any formal policies or procedures to determine the compensation of our directors or executive officers. The compensation of the directors and executive officers is determined by the board of directors, based on the recommendations of the Compensation Committee. Recommendations of the Compensation Committee are made giving consideration to the objectives discussed below and, if applicable, considering applicable industry data.
In April 2021, we hired executive compensation consultants to perform market research on the appropriate compensation for our named executive officers (NEOs) and other individuals in the company. Based on the results of the research, compensation packages were proposed to the Compensation Committee, who approved the current compensation. While our Chief Executive Officer is a member and typically attends meetings of our Compensation Committee, the Compensation Committee meets outside of the presence of our Chief Executive Officer when discussing and approving his compensation. In July 2022, the board of directors approved an employment agreement entered into by us and with Mr. Cacioppo.
The compensation of our NEOs in 2021 was comprised of the following major elements: (a) base salary; (b) an annual, cash bonus; and (c) stock options under the 2019 Equity Incentive Plan. Base salaries, annual bonuses and long-term equity incentive compensation awards were determined on an individual basis, taking into consideration the past, current and potential contribution to our success, the NEO’s experience and expertise, the position and responsibilities of the NEO, and competitive industry pay practices for other high growth, premium brand companies of similar size and revenue growth potential.
The board of directors and our Compensation Committee considered risks associated with executive compensation and does not believe that our executive compensation policies and practices encourage its executive officers to take inappropriate or excessive risks. In addition to cash compensation, NEOs are compensated in part through the granting of options and restricted stock awards which is compensation that is both “at risk” and associated with long-term value creation. The value of such compensation is dependent upon shareholder return over the applicable vesting period which reduces the incentive for executives to take inappropriate or excessive risks as their long-term compensation is at risk.
The Compensation Committee endeavors to ensure that the philosophy and operation of our compensation program reinforces its culture and values, creates a balance between risk and reward, attracts, motivates, and retains executive officers over the long-term and aligns their interests with those of the shareholders.
85
Summary Compensation Table
The following table sets forth information concerning the compensation of our NEOs for the year ended December 31, 2021.
| Name and Principal Position | Year | Salary ($) |
Bonus ($)(2) |
Option awards ($)(3) |
All other compensation ($)(4) | Total ($) |
||||||||||||||||||
James Cacioppo Chief Executive Officer |
2021 | 487,132 | 1,141,333 | 7,125,000 | 12,247 | 8,765,712 | ||||||||||||||||||
Louis Jon Barack President, Corporate Secretary and Interim Chief Financial Officer |
2021 | 337,498 | 436,667 | 2,375,000 | 13,205 | 3,162,370 | ||||||||||||||||||
Leonardo Garcia-Berg (1) Chief Operations Officer |
2021 | 200,088 | 101,680 | 2,319,060 | 7,472 | 2,628,300 | ||||||||||||||||||
| (1) | Mr. Garcia-Berg was hired by the Company on May 24, 2021 and appointed Chief Operations Officer of the Company on June 30, 2021. |
| (2) | Mr. Cacioppo and Mr. Barack each received an annual discretionary cash bonus for work performed January 1, 2021 through December 31, 2021. Mr. Garcia-Berg received a prorated annual discretionary cash bonus for work performed from his hire date through December 31, 2021. |
| (3) | The reported amounts reflect the grant date fair value of stock option awards calculated in accordance with Financial Accounting Standards Board (FASB) ASC 718. For additional information, see Note 15 - Share-Based Compensation and Other Benefits of our audited consolidated financial statements. Such grant date fair values do not take into account any estimated forfeitures. The assumptions used in calculating the grant date fair value of the stock options reported in this table are set forth in the section titled “Management’s Discussion and Analysis of Financial Condition and Results of Operations—Critical Accounting Policies and Estimates—Share-Based Compensation Arrangements.” |
| (4) | Represents our matching contributions to our 401(k) plan and health and disability benefits. |
Outstanding Equity Awards at 2021 Fiscal Year End
The following table provides information regarding outstanding stock options and/or restricted stock awards held by our NEOs as of December 31, 2021:
| Name | Grant Date | Number of Securities Underlying Unexercised Options (#) Exercisable |
Number of Securities Underlying Unexercised Options (#) Unexercisable |
Option Exercise Price ($) |
Option Expiration Date |
Number of share or units of stock that have not vested (#) |
Market
Value of share or units of stock that have not vested ($) |
||||||||||
| James Cacioppo | 4/17/2019 | 2,385,000 | — | 1 | 2.00 | 4/17/2029 | — | — | |||||||||
| 9/22/2020 | — | — | — | — | 466,392 | 2 | $ | 1,026,062 | |||||||||
| 10/27/2021 | 973,333 | 1,946,667 | 3 | 3.91 | 10/27/2031 | — | — | ||||||||||
| 10/27/2021 | 26,667 | 53,333 | 4 | 3.91 | 10/27/2031 | — | — | ||||||||||
| Louis Jon Barack | 4/17/2019 | 528,667 | 264,333 | 5 | 2.00 | — | — | — | |||||||||
| 9/22/2020 | — | — | — | — | 164,609 | 6 | $ | 362,140 | |||||||||
| 10/27/2021 | — | 80,000 | 7 | 3.91 | 10/27/2031 | — | — | ||||||||||
| 10/27/2021 | — | 920,000 | 8 | 3.91 | 10/27/2031 | — | — | ||||||||||
| Leonardo Garcia-Berg | 6/08/2021 | — | 500,000 | 9 | 6.10 | 6/08/2031 | — | — | |||||||||
| 11/03/2021 | — | 100,000 | 10 | 3.70 | 11/03/2031 | — | — | ||||||||||
| (1) | Granted 2,385,000 options which vest ratably over three years beginning on the first anniversary of the date of grant. |
| (2) | Granted 1,399,177 shares of restricted stock which vest ratably over three years beginning on May 1, 2020. |
| (3) | Granted 2,920,000 options which vest ratably over three years beginning on May 1, 2021. |
| (4) | Granted 80,000 options which vest ratably over three years beginning on May 1, 2021. |
| (5) | Granted 793,000 options which vest ratably over three years beginning on the first anniversary of the date of grant. |
| (6) | Granted 493,827 shares of restricted stock which vest ratably over three years beginning on May 1, 2020. |
| (7) | Granted 80,000 options which vest ratably over three years beginning on May 1, 2021. |
| (8) | Granted 920,000 options which vest ratably over three years beginning on May 1, 2021. |
| (9) | Granted 500,000 options which vest ratably over five years beginning on May 24, 2021. On July 28, 2022 the Company’s board of directors approved and Mr. Garcia-Berg accepted the cancellation of such options in exchange for a future grant of the same number of options with the same terms and conditions but with an exercise price reflecting the market price of the Company’s subordinate voting shares on the CSE on the date such replacement options are issued. |
| (10) | Granted 100,000 options which vest ratably over five years beginning on the first anniversary of the date of grant. |
86
Employment Agreements
Cacioppo Employment Agreement
On July 3, 2022, we entered into an employment agreement with Mr. Cacioppo with retroactive effect to January 1, 2022 for the position of Chief Executive Officer and Chairman of the board of directors (the Cacioppo Employment Agreement). The Cacioppo Employment Agreement provides for Mr. Cacioppo’s at-will employment for an initial period of two years and one day following its effective date, with automatic renewal for one successive two-year period, unless written notice is provided at least 60 days prior to the end of the initial period. Mr. Cacioppo’s current annual base salary is $750,000 (which will automatically be increased by $100,000 on each anniversary of the effective date) and he is eligible to receive an annual bonus with a target amount of 100% of his annual base salary. The actual amount of any bonus earned by Mr. Cacioppo will be determined by the board of directors, but in no event will such bonus be less than 100% of Mr. Cacioppo’s base salary. Mr. Cacioppo is also eligible to participate in the employee benefit plans available to our employees, subject to the terms of such plans, including our 2019 Equity Incentive Plan. Beginning on or before July 30, 2022, and each January 1 thereafter through January 1, 2026, Mr. Cacioppo will be eligible to receive an annual equity compensation award (an LTI Award) in the form of a stock option to purchase 3,000,000 shares. The Cacioppo Employment Agreement provides that the first two LTI Awards will be vested as to one-third of the option shares on the grant date, and the remaining option shares will vest in two equal installments on January 1 of each year following the option grant date, subject to Mr. Cacioppo’s continued service through the applicable vesting date. The second two LTI Awards will be vested as to 50% of the option shares on the grant date, and the remaining option shares will vest on the first anniversary of the option grant date, subject to Mr. Cacioppo’s continued service through such date. The fifth LTI Award will be fully vested on the January 1, 2026 option grant date. Each LTI Award will remain exercisable following Mr. Cacioppo’s termination date until the earlier of (i) the expiration of the option term and (ii) the fifth anniversary of Mr. Cacioppo’s termination date.
The Cacioppo Employment Agreement provides that in the event of a termination of Mr. Cacioppo’s employment by the Company without “cause” (as defined in the Cacioppo Employment Agreement), by our election not to renew the initial term of his employment agreement, or by Mr. Cacioppo for “good reason” (as defined in the Cacioppo Employment Agreement), then subject to Mr. Cacioppo’s execution and non-revocation of a general release of claims, Mr. Cacioppo will be eligible to receive: (i) a one-time lump sum payment in an amount equal to $5,000,000, (ii) grant of the next LTI Award that would otherwise have been granted had Mr. Cacioppo remained employed by the Company through the next scheduled LTI Award date (or economically equivalent compensation in an alternative form if the grant of such awards would violate applicable law, or in the event there is an insufficient number of shares available under the company’s equity plan to make the LTI Award), and (iii) full vesting of all of Mr. Cacioppo’s outstanding equity-based awards.
Upon a termination of Mr. Cacioppo’s employment due to death, subject to his estate’s execution of a general release of claims in favor of the Company, his estate will be entitled to (i) a lump sum payment of a pro rata portion of his annual bonus in respect of the fiscal year in which such termination occurs, (ii) a lump sum cash payment in an amount equal to eighteen times the monthly COBRA premiums that Mr. Cacioppo would be required to pay to continue group health coverage at the level in effect on the date of his termination, and (iii) full vesting on the date of death of all outstanding equity-based award, subject to compliance with applicable law (clause (i) and (ii), the Separation Payments). Upon a termination of Mr. Cacioppo’s employment due to disability, Mr. Cacioppo will be eligible to receive the Separation Payments subject to his execution of a general release of claims in favor of the Company.
In the event of a change in control (as defined in the Cacioppo Employment Agreement), Mr. Cacioppo will be eligible to receive (i) a one-time lump sum payment in an amount equal to $5,000,000, (ii) grant of) the next LTI Award that would otherwise have been granted had Mr. Cacioppo remained employed by the Company through the next scheduled LTI Award date, and (iii) full vesting of all of Mr. Cacioppo’s outstanding equity-based awards. If any payment or benefit Mr. Cacioppo receives in connection with a change in control would constitute a “parachute payment” within the meaning of Section 280G of the Code subject to the excise tax under Section 4999, Mr. Cacioppo will receive a full tax gross-up payment.
The Cacioppo Employment Agreement contains various restrictive covenants, including a noncompetition provision during the term of his employment and for 18 months thereafter and a non-solicitation provision during the term of his employment and for 2 years thereafter.
Barack Employment Agreement
We entered into an employment agreement with Mr. Barack effective May 1, 2019, which was modified by a letter agreement dated June 9, 2020. The employment agreement as modified provides for, among other things, an initial base salary of $250,000, which may increase from time to time and has increased annually, most recently in 2021 to $400,000, and a discretionary performance-based bonus that may be paid by us in cash or equity. Mr. Barack is also eligible, subject to approval by our board of directors or the Compensation Committee, for equity grants under the Plan. The employment agreement includes a standard six (6)-month noncompetition, two (2)-year non-solicitation and nondisclosure covenants. In the event Mr. Barack’s employment is terminated without cause (whether or not in connection with a change in control), he is entitled to receive a lump sum cash severance payment equivalent to six (6) months of his base salary, subject to his execution of a general releases of claims. Upon a change of control, he is automatically terminated and is entitled to a cash severance equivalent to six (6) months of his base salary and vesting acceleration of any outstanding equity grants.
Garcia-Berg Employment Agreement
We entered into an employment agreement with Mr. Garcia-Berg effective May 24, 2021. The employment agreement provides for, among other things, an initial base salary of $330,000, which may increase from time to time, and a discretionary performance-based bonus targeted at 50% of his base salary that may be paid by us in cash or equity. Mr. Garcia-Berg is also eligible, subject to approval by our board of directors or the Compensation Committee, for equity grants under the Plan. The employment agreement includes a standard six (6)-month noncompetition, two (2)-year non-solicitation and nondisclosure covenants. In the event Mr. Garcia-Berg’s employment is terminated by the Company without cause either prior to a change in control or after the one year period following a change in control, then subject to his execution of a general release of claims in favor of the Company he is entitled to a lump sum cash payment based on the following schedule: (i) if less than 12 months of employment, 24 months of his base salary, (ii) if 12-17 months of employment, 18 months of his base salary, (iii) if 18-23 months of employment, 12 months of his base salary, or (iv) if greater than 24 months of employment, 6 months of his base salary. In addition, if Mr. Garcia-Berg’s employment is terminated by the Company without cause either upon a change of control or within the one year period following a change in control, then subject to his execution of a general release of claims in favor of the Company, he is entitled to vesting acceleration of his initial grant of stock options, and a lump sum cash payment equal to of 24 months of his base salary if such action occurs within the first 24 months of his employment or 12 months of base salary if such termination occurs thereafter.
Kremer Employment Agreement
We entered into an employment agreement with Mr. Kremer, effective October 5, 2021. Mr. Kremer previously served as our Chief Financial Officer from October 18, 2021 through July 12, 2022. The employment agreement provides for, among other things, an initial base salary of $400,000, which may increase from time to time, and a discretionary performance-based bonus targeted at 50% of his base salary that may be paid by us in cash or equity. However, the bonus he is eligible in his first year only has a feature where he is guaranteed 50% of such bonus and the remaining 50% is based on several financial-related filings and requirements being timely met. Mr. Kremer is also eligible, subject to approval by our board of directors or the Compensation Committee, for equity grants under the Plan. The employment agreement includes a standard six (6)-month noncompetition, two (2)-year non-solicitation and nondisclosure covenants. Prior to a change in control, in the event Mr. Kremer’s employment is terminated without cause or Mr. Cacioppo leaves in the first 12 months of Mr. Kremer’s employment and Mr. Kremer leaves for good reason, then he is entitled to a cash severance of 12 months of base salary to be paid over a 12-month period. Upon or following a change in control, in the event Mr. Kremer’s employment is terminated without cause or he leaves his employment for good reason, he is entitled to a cash severance of 12 months of base salary to be paid over a 12-month period and vesting acceleration of his initial grant of options under the Plan.
On July 12, 2022, Mr. Kremer resigned without good reason, and all unvested equity awards granted to him pursuant to his employment agreement were forfeited, and no severance payments are due.
87
Compensation Elements
The executive compensation program during the fiscal year ended December 31, 2021 consisted of three (3) key elements: (i) base salaries; (ii) cash bonuses; and (iii) equity-based compensation.
Base Salaries
Base salaries are intended to provide an appropriate level of fixed compensation that will assist in employee retention and recruitment. Base salaries are determined on an individual basis, taking into consideration the past, current and potential contribution to our success, the position and responsibilities of the NEOs and competitive industry pay practices for other high growth, similarly sized companies in the industry.
Incentive Compensation
Bonuses are awarded based on qualitative and quantitative performance standards and reward performance of each NEO individually. The determination of an NEO’s performance may vary from year to year depending on economic conditions and conditions in the industry in which we operate and may be based on measures such as acquisition activity, equity raises, revenue growth, EBITDA growth, and talent recruitment and retention, metrics the compensation committee and management believe to provide proper incentives for achieving long-term shareholder value for us at this time. Except as set forth above under the heading “Cacioppo Employment Agreement”, the compensation committee and the board of directors, when required, retain full discretion over performance evaluation and the amount of any bonuses to be paid to NEOs.
Equity-Based Compensation
The long-term component of compensation for executive officers, including the NEOs, is currently based on a combination of restricted stock and/or stock options issued under the 2019 Equity Incentive Plan. This component of compensation is intended to reinforce management’s long-term commitment to our performance. The board of directors believes that incentive compensation in the form of stock option grants and/or restricted stock awards which vest over time, typically three to five years, is beneficial and necessary to attract and retain both senior executives and managerial talent at other levels. Furthermore, the board of directors believes stock option grants are an effective long-term incentive vehicle because they are directly tied to share price over a longer period, up to 10 years, and motivate executives to deliver sustained long-term performance and increase shareholder value, and have a time horizon that aligns with long-term corporate goals. And restricted stock includes a tax component at issuance or vesting, whichever the grantee decides, which incentivizes the restricted stock grantee to deliver mid-term performance and increase shareholder value that aligns with mid-term corporate goals.
Restrictions on Hedging
Our Insider Trading and Reporting Policy prohibits our officers (including the NEOs), directors and employees from buying or selling financial instruments that are designed to hedge or offset a decrease in market value of our equity securities granted as compensation or held, directly or indirectly, by such individuals.
88
Equity Incentive Plan Summary
On or about April 29, 2019, the shareholders of our predecessor first adopted the 2019 Equity Incentive Plan, which amendments were most recently approved by our shareholders at our annual general meeting held on June 29, 2022.
The Plan permits the grant of: (i) nonqualified stock options (NQSOs) and incentive stock options (ISOs and collectively with NQSOs, Options); (ii) restricted stock awards (RSAs); (iii) restricted stock units (RSUs); (iv) stock appreciation rights (SARs); and (v) performance compensation awards (collectively with Options, RSAs, RSUs, and SARs, Awards).
As of December 31, 2021, the following Awards were outstanding under the Plan: (i) a total of 20,429,120 Options, representing approximately 11% of the then Outstanding Share Number; and (ii) a total of 2,859,151 unvested RSAs, representing less than 2% of the then Outstanding Share Number. As of December 31, 2021, an aggregate of 7,904,819 Subordinate Voting Shares remained available for issuance under the Plan, representing approximately 4% of the then Outstanding Share Number. There is an additional 1,314,147 Subordinate Voting Shares available for certain new hires.
89
The number of shares that are available for issuance under the Plan cannot exceed 15% of the number of outstanding Subordinate Voting Shares, including the number of Subordinate Voting Shares underlying the Multiple Voting Shares and Super Voting Shares on an as-converted basis (the Fully Converted Number of Shares), plus an additional 2% of the Fully Converted Number of Shares that may be issued as inducement to employees and officers not previously employed by the Company and who were not previously an insider of the Company. Any Subordinate Voting Shares subject to an Award under the Plan that are forfeited, cancelled, expire unexercised, are settled in cash, or are used or withheld to satisfy tax withholding obligations of a Participant shall again be available for Awards under the Plan. The maximum number of Subordinate Voting Shares that may be issued pursuant to the exercise of Incentive Stock Options will be equal to the share reserve as of May 31, 2022.
In the event of a capitalization adjustment (as defined in the Plan), the board of directors will appropriately and proportionately adjust (i) the class(es) and maximum number of securities that may be issued under the Plan, (ii) the class(es) and maximum number of securities that may be issued pursuant to the exercise of incentive stock options, and (iii) the class(es) and number of securities and price per share subject to outstanding Awards. The board of directors will make such adjustments, and its determination will be final, binding and conclusive.
Purpose
The purpose of the Plan is to: (i) promote and retain employees, directors and consultants capable of assuring our future success; (ii) motivate management to achieve long-range goals; and (iii) to provide compensation and opportunities for ownership and alignment of interests with shareholders.
The Plan permits the grant of (i) Options; (ii) RSAs; (iii) RSUs; (iv) SARs; and (v) other awards, as more fully described below. Pursuant to the Plan, the board of directors administers the Plan, but may delegate some or all of the administration of the Plan to a committee or committees thereof. The Plan is currently administered by the board of directors, and the board of directors has delegated to the Compensation Committee the ability to grant Awards with board of directors review for Directors and Officers.
Eligibility
Any of our employees, officers, directors, and consultants are eligible to participate (each a Participant) in the Plan if selected by the board of directors or the Compensation Committee. The basis of participation of an eligible recipient of an Award under the Plan, and the type and amount of any Award that an individual will be entitled to receive under the Plan, will be determined by board of directors and/or Compensation Committee.
90
Awards
Options
The board of directors or Compensation Committee is authorized to grant Options under the Plan to purchase Subordinate Voting Shares that are either ISOs, meaning they are intended to satisfy the requirements of Section 422 of the Code, or NQSOs, which are not intended to satisfy the requirements of Section 422 of the Code; provided, however, that eligibility to receive an Award of ISOs is limited to our or our subsidiaries employees. Only employees are eligible to receive ISOs. Unless the board of directors or Compensation Committee determines otherwise in the case of an Option substituted for another Option in connection with a corporate transaction, the exercise price of an Option will not be less than the fair market value (as determined under the Plan) of the Subordinate Voting Shares at the time of grant. Options will be subject to such terms, including the exercise price and the conditions and timing of exercise, as may be determined by the board of directors or the Compensation Committee and specified in the applicable award agreement. The maximum term of an Option will be ten years from the date of grant (or five years in the case of an ISO granted to a 10% shareholder). Payment in respect of the exercise of an Option may be made in cash or by check, or by such other method as the board of directors or Compensation Committee may determine to be appropriate. The board of directors or Compensation Committee may, in its discretion, accelerate the vesting and exercisability of Options. Unless otherwise provided in the applicable award agreement or as may be determined by the board of directors or Compensation Committee, upon a Participant’s termination of service with us the unvested portion of an Option will be forfeited.
Restricted Stock
A restricted stock award is a grant of Subordinate Voting Shares, which are subject to forfeiture restrictions during a restriction period. The board of directors or Compensation Committee will determine the price, if any, to be paid by the Participant for each Subordinate Voting Share subject to a restricted stock award. If any payment is required, it may be paid in cash, by check, or by such other method as the board of directors or Compensation Committee may determine to be appropriate. The board of directors or Compensation Committee may condition the expiration of the restriction period, if any, upon: (i) the Participant’s continued service over a period of time with us or our affiliates; (ii) the achievement by the Participant, us or our affiliates of any other performance goals set by the board of directors or the Compensation Committee; or (iii) any combination of the above conditions as specified in the applicable award agreement. If the specified conditions are not attained, the Participant will forfeit the portion of the restricted stock award with respect to which of those conditions are not attained, and the underlying Subordinate Voting Shares will be forfeited or repurchased. At the end of the restriction period, if the conditions (if any) have been satisfied, the restrictions imposed will lapse with respect to the applicable number of Subordinate Voting Shares. During the restriction period, unless otherwise provided in the applicable award agreement, a Participant will have the right to vote the shares underlying the restricted stock and dividends will be paid as determined by the board of directors or Compensation Committee. The board of directors or Compensation Committee may, in its discretion, accelerate the vesting and delivery of shares of restricted stock. Unless otherwise provided in the applicable award agreement or as may be determined by the board of directors or Compensation Committee, upon a Participant’s termination of service with us or our affiliates, the unvested portion of a restricted stock award will be forfeited or repurchased.
Restricted Stock Units
RSUs are granted in reference to a specified number of Subordinate Voting Shares and entitle the holder to receive, on achievement of specific performance goals established by the board of directors or Compensation Committee, after a period of continued service with us or our affiliates or any combination of the above as set forth in the applicable award agreement, one Subordinate Voting Share for each such Subordinate Voting Share covered by the RSU; provided, that the board of directors or Compensation Committee may elect to pay cash, or part cash and part Subordinate Voting Shares in lieu of delivering only Subordinate Voting Shares. The board of directors or Compensation Committee may, in its discretion, accelerate the vesting of RSUs. Unless otherwise provided in the applicable award agreement or as may be determined by the board of directors or Compensation Committee, upon a Participant’s termination of service with us or our affiliates, the unvested portion of the RSUs will be forfeited. RSU holders will not have any shareholder rights, including voting or dividend rights, with respect to their RSUs until Subordinate Voting Shares are issued in settlement of such RSUs; provided that the board of directors or Compensation Committee may provide for dividend equivalents, subject to applicable terms and conditions. The board of directors or Compensation Committee may, in its discretion, accelerate the vesting of RSUs. Unless otherwise provided in the applicable award agreement or as may be determined by the board of directors or Compensation Committee, upon a Participant’s termination of service with us or our affiliates, the unvested portion of an RSU award will be forfeited. As of June 30, 2022, no RSUs have been awarded.
91
Stock Appreciation Rights
A SAR entitles the Participant to receive, upon exercise of the SAR, a payment in an amount equal to the increase in the fair market value of a specified number of Subordinate Voting Shares from the date of the grant of the SAR and the date of exercise payable in Subordinate Voting Shares. Any grant may specify a vesting period or periods before the SAR may become exercisable and permissible dates or periods on or during which the SAR shall be exercisable. No SAR may be exercised more than ten years from the grant date. Upon a Participant’s termination of service with us or our affiliates, the same general conditions applicable to Options as described above would be applicable to the SAR. The board of directors or Compensation Committee may, in its discretion, accelerate the vesting of SARs. Unless otherwise provided in the applicable award agreement or as may be determined by the board of directors or Compensation Committee, upon a Participant’s termination of service with us or our affiliates, the unvested portion of an SAR will be forfeited. As of June 30, 2022, no SARs have been awarded.
Substitute Awards
If we or an affiliate acquires another company by merger, consolidation, stock purchase or asset purchase (an Acquired Entity), the board of directors may authorize the grant of Substitute Awards to current and former employees, directors and consultants of the Acquired Entity in substitution for stock and stock-based awards (Acquired Entity Awards) held by the current and former employees, directors or consultants of the Acquired Entity to in order to preserve the economic value of the Acquired Entity Awards, subject to applicable Canadian securities laws. The number of Subordinate Voting Shares and the exercise price or purchase price (if applicable) underlying the Substitute Awards will be adjusted as the board of directors determines necessary to achieve preservation of economic value.
General
The board of directors or Compensation Committee may impose restrictions on the grant, exercise or payment of an Award as it determines appropriate. Generally, Awards granted under the Plan shall be nontransferable except by will or by the laws of descent and distribution. In general, no Participant shall have any rights as a shareholder with respect to Subordinate Voting Shares covered by Options, SARs, or RSUs, unless and until such Awards are settled in Subordinate Voting Shares. No Option (or, if applicable, SARs) shall be exercisable, no Subordinate Voting Shares shall be issued, no certificates for Subordinate Voting Shares shall be delivered and no payment shall be made under the Plan except in compliance with all applicable laws. The board of directors may amend, alter, suspend, discontinue or terminate the Plan and the board of directors may amend any outstanding Award at any time; provided that (i) such amendment, alteration, suspension, discontinuation, or termination shall be subject to the approval of the shareholders if such approval is necessary to comply with any tax or regulatory requirement applicable to the Plan (including, without limitation, as necessary to comply with any rules or requirements of an applicable securities exchange), and (ii) no such amendment or termination may adversely affect Awards then outstanding without the Award holder’s permission. In the event of a Change in Control, as defined in the Plan, the board of directors may, in its sole discretion, cause any (or a combination) of the following to be effective upon the consummation of the Change in Control (or effective immediately prior to the consummation of the Change in Control, provided that the consummation of the change in control subsequently occurs): (i) arrange for the surviving corporation or acquiring corporation to assume or continue the Awards or to substitute a similar award for the Award (including, but not limited to, an award to acquire the same consideration paid to stockholders of the Company pursuant to the Change in Control), (ii) arrange for the assignment of any reacquisition or repurchase rights held by the Company in respect of Subordinate Voting Shares issued pursuant to the Award to the surviving corporation, (iii) accelerate the vesting, in whole or in part, of any Award to a date prior to the effective time of such Change in Control as the board of directors determines, with such Award terminating if not exercised immediately prior to the effective time of the Change in Control; (iv) arrange for the lapse, in whole or in part, of any reacquisition or repurchase rights held by the Company with respect to any Award; (v) cancel or arrange for the cancellation of any Award, to the extent not vested or not exercised prior to the effective time of the Change in Control, in exchange for such cash consideration, if any, as the board of directors, in its discretion, may consider appropriate; and (vi) make a payment, in such form as may be determined by the board of directors equal to the excess, if any, of (A) the value of the property the participant would have received on exercise of the Award immediately prior to the effective time of the Change in Control, over (B) any exercise price payable by such holder in connection with such exercise.
92
Tax Withholding.
We may take such action as it deems appropriate to ensure that all applicable federal, state, provincial, local and/or foreign payroll, withholding income or other taxes, which are the sole and absolute responsibility of a Participant, are withheld or collected from such Participant.
93
CERTAIN RELATIONSHIPS AND RELATED-PERSON TRANSACTIONS
In addition to the executive officer and director compensation arrangements discussed above under “Management—Director Compensation” and “Executive Compensation,” below we describe transactions since January 1, 2019 to which we have been or will be a participant, in which the amount involved in the transaction exceeds or will exceed $120,000 and in which any of our directors, executive officers, or beneficial holders of more than five percent of any class of our capital stock, or any immediate family member of, or person sharing the household with, any of these individuals, had or will have a direct or indirect material interest.
10% Senior Secured Notes
During the years ended December 31, 2019 and 2020, we issued 10% Senior Secured Notes with warrants, some of which were issued to related parties. 10% Senior Secured Notes and related warrants issued to directors, executive officers, and a significant shareholder are as follows:
| Number of | Principal Outstanding as of December 31, | Interest
Paid during the Years Ended December 31, | ||||||||||||||||||||
| Warrants | (in millions) | (in millions) | ||||||||||||||||||||
| Name | Relationship | Issued(2) | 2021 | 2020 | 2021 | 2020 | ||||||||||||||||
| James Cacioppo and controlled entities (1) | CEO | 6,900,000 | $ | — | $ | 11.5 | $ | — | $ | 1.1 | ||||||||||||
| Denis Arsenault(4) | Shareholder | 6,000,000 | $ | — | $ | 10.0 | $ | — | $ | 0.9 | ||||||||||||
| Joseph Max Cohen | Former Executive and Former Board of Directors’ Member | 450,000 | $ | 0.56 | $ | 0.75 | $ | 0.06 | $ | 0.02 | ||||||||||||
| Erich Mauff | Former Executive and Former Board of Directors’ Member | 300,000 | $ | — | $ | 0.5 | $ | — | $ | 0.04 | ||||||||||||
| Kimberly Bambach and controlled entity (3) | Former Executive | 60,000 | $ | 0.10 | $ | 0.10 | $ | 0.01 | $ | — | ||||||||||||
| Benjamin Cross | Board of Directors’ Member | 120,000 | $ | 0.15 | $ | 0.20 | $ | 0.02 | $ | 0.02 | ||||||||||||
(1) Entities controlled by James Cacioppo include One East Partners LP, OEP Opportunities L.P., JAC Serpentine, LLC, Serpentine Capital Management II LLC.
(2) The warrants have an exercise price of $1.25 and expire on December 23, 2024. During the year ended December 31, 2021, 300,000 warrants were exercised by Mr. Mauff.
(3) The entity controlled by Kimberly Bambach is KABL Holdings LLC.
(4) Beneficially owns more than 5% of our Subordinate Voting Shares.
Pursuant to the terms of the 10% Senior Secured Notes, in connection with the January 2021 and February 2021 equity offerings, in January 2021, we redeemed a total of $8.1 million in 10% Senior Secured Notes. Of the total amount redeemed in January 2021, $3.0 million was paid, at par value, to related parties above. In February 2021 certain of these related parties sold their remaining principal amounts of publicly traded 10% Senior Secured Notes totaling $19.2 million.
Former Loans
On October 30, 2020, we entered into a promissory note with James Cacioppo, pursuant to which Mr. Cacioppo borrowed $1,212,275.05 at a rate of 5% per annum, compounded annually. On October 27, 2021, as part of Mr. Cacioppo’s bonuses earned in 2020 and 2021, the full principal amount plus interest of $60,096.32 was paid by offset from such bonuses. The loan is no longer outstanding.
94
On April 17, 2019 and October 30, 2020, we entered into a promissory note with Louis Jon Barack, pursuant to which Mr. Barack borrowed $800,000 at a rate of 2.89% per annum, compounded annually, and a promissory note in which Mr. Barack borrowed $322,200 at a rate of 5% per annum, compounded annually, respectively. On October 27, 2021, as part of Mr. Barack’s bonuses earned in 2020 and 2021, the full principal amount of the October 30, 2020 note plus interest of $15,972.48 was paid by offset from such bonuses. On December 20, 2021, we repurchased 163,954 subordinate voting shares from Mr. Barack to fully satisfy the full principal amount of the April 17, 2019 note plus interest of $63,352.62. The loans are no longer outstanding.
On October 30, 2020, we entered into a promissory note with our former Co-President and Director, Erich Mauff, pursuant to which Mr. Mauff borrowed $719,810.20 at a rate of 5% per annum, compounded annually. Mr. Mauff, a founder of the Company, resigned as Co-President and Director on June 30, 2021. He serves as a consultant for the Company. On July 31, 2021, as part of Mr. Mauff’s severance, we forgave the full principal amount plus interest of $26,827.60. The loan is no longer outstanding.
On April 17, 2019 and October 30, 2020, we entered into a promissory note with Kimberly Bambach, pursuant to which Ms. Bambach borrowed $1,012,500 at a rate of 2.89% per annum, compounded annually, and a promissory note in which Ms. Bambach borrowed $194,522.98 at a rate of 5% per annum, compounded annually, respectively. On December 1, 2021, as part of Ms. Bambach's severance, we repurchased 307,803 subordinate voting shares from Ms. Bambach to fully satisfy the full principal amount of both notes plus interest of $89,124.96. The loans are no longer outstanding.
Management Services Agreements
During the years ended December 31, 2021 and 2020, we paid $0.04 million and $0.2 million to entities controlled by our CEO for shared costs of administrative services, the provision of financial and research-related advice, and sourcing and assisting in mergers, acquisitions and capital transactions.
Review, Approval or Ratification of Transactions with Related Parties
We do not have any formal written policies and procedures for the review, approval, or ratification of transactions with related persons or conflicted transactions. However, in practice, when such transactions arise, they are referred to the board of directors for consideration and approval, and we rely on the board of directors to review related party transactions on an ongoing basis to prevent conflicts of interest. We intend to adopt a related party transaction policy in the future. In addition, as we are listed on the Canadian Securities Exchange and are a reporting issuer under Canadian securities laws, we are subject to Multilateral Instrument 61-101 – Protection of Minority Securityholders in Special Transactions (MI 61-101) which includes requirements in connection with, among other things, “related party transactions” being transactions by which an issuer directly or indirectly engages in the following with a related party: acquires, sells, leases or transfers an asset, acquires the related party, acquires or issues treasury securities, amends the terms of a security if the security is owned by the related party or assumes or becomes subject to a liability or takes certain other actions with respect to debt. For the purposes of MI 61-101, the term “related party” includes directors, senior officers and holders of more than 10% of the voting rights attached to all outstanding voting securities of the issuer or holders of a sufficient number of any securities of the issuer to materially affect control of the issuer. MI 61-101 requires, subject to certain exceptions, the preparation of a formal valuation relating to certain aspects of the transaction and more detailed disclosure in the proxy material sent to security holders in connection with a related party transaction including related to the valuation. MI 61-101 also requires, subject to certain exceptions, that an issuer not engage in a related party transaction unless the shareholders of the issuer, other than the related parties, approve the transaction by a simple majority of the votes cast.
95
The following table provides information regarding the beneficial ownership of our Subordinate Voting Shares, as of August 4, 2022, by:
| • | each person or entity, or group of affiliated persons or entities, known by us to beneficially own more than 5.0% of our Subordinate Voting Shares; |
| • | each of our directors; |
| • | each of our NEOs; and |
| • | all of our executive officers and directors as a group. |
Beneficial ownership is determined in accordance with the rules of the Securities and Exchange Commission and generally includes voting or investment power with respect to securities. In computing the number of shares beneficially owned by a person and the percentage ownership of that person, any Preferred Shares, Super Voting Shares, Multiple Voting Shares and Subordinate Voting Shares that a person has the right to acquire within 60 days of August 4, 2022 through the exercise of stock options, warrants or other rights are deemed outstanding, but are not deemed outstanding for computing the percentage ownership of any other person. To our knowledge, except as set forth in the footnotes to this table and subject to applicable community property laws, each person named in the table has sole voting and investment power with respect to the shares set forth opposite such person’s name. Each shareholder’s percentage ownership is based on 195,849,084 Subordinate Voting Shares and no Multiple Voting Shares, Super Voting Shares or Preferred Shares that were issued and outstanding as of August 4, 2022. Except as otherwise indicated, the address of each of the persons in this table is c/o Jushi Holdings Inc., 301 Yamato Road, Suite 3250, Boca Raton, Florida 33431.
96
Subordinate Voting Shares(1) |
||||||||
Name, Position and Address of |
Number Beneficially Owned |
% of Subordinate Voting Shares Beneficially Owned |
||||||
| James Cacioppo - Chief Executive Officer and Chairman Florida, U.S. | 36,313,465 | (2) | 16.5 | % | ||||
| Louis Jon Barack – President, Corporate Secretary and Interim Chief Financial Officer Florida, U.S. | 5,448,107 | (3) | 2.7 | % | ||||
| Leonardo Garcia-Berg - Chief Operations Officer Connecticut, U.S. | 100,000 | * | ||||||
| Benjamin Cross - Director Connecticut, U.S. | 419,080 | (4) | * | |||||
| Stephen Monroe - Director New York, U.S. | 227,636 | (5) | * | |||||
| Peter Adderton - Director California, U.S. | 169,080 | (6) | * | |||||
| Marina Hahn - Director New York, U.S. | 37,301 | (7) | * | |||||
| Denis Arsenault – Shareholder Hergiswil, Switzerland | 20,542,863 | 9.8 | % | |||||
| All directors and executive officers as a group(8) | 42,714,669 | 19.66 | % | |||||
* Indicates percentage of less than 1.0%
(1) The total number of issued and outstanding Subordinate Voting Shares for purposes of calculating the percentage of Subordinate Voting Shares beneficially owned by each individual includes all restricted stock granted to such individual regardless of whether such restricted stock is vested or unvested.
(2) Includes 4,385,000 Subordinate Voting Shares subject to stock options that are or become exercisable within 60 days of August 4, 2022 and 20,010,000 Subordinate Voting Shares underlying warrants exercisable within 60 days of August 4, 2022.
(3) Includes 1,126,334 Subordinate Voting Shares subject to stock options that are or become exercisable within 60 days of August 4, 2022 and 1,500,000 Subordinate Voting Shares underlying warrants exercisable within 60 days of August 4, 2022.
(4) Includes 60,000 Subordinate Voting Shares subject to stock options that are or become exercisable within 60 days of August 4, 2022 and 120,000 Subordinate Voting Shares underlying warrants exercisable within 60 days of August 4, 2022.
(5) Includes 73,952 Subordinate Voting Shares subject to stock options that are or become exercisable within 60 days of August 4, 2022.
(6) Includes 60,000 Subordinate Voting Shares subject to stock options that are or become exercisable within 60 days of August 4, 2022.
(7) Includes 20,000 Subordinate Voting Shares subject to stock options that are or become exercisable within 60 days of August 4, 2022.
(8) Excludes Denis Arsenault.
97
We are authorized to issue an unlimited number of Subordinate Voting Shares, an unlimited number of Multiple Voting Shares, an unlimited number of Super Voting Shares and an unlimited number of Preferred Shares. The outstanding capital stock as of August 4, 2022 consists of 195,849,084 Subordinate Voting Shares; (ii) 0 Multiple Voting Shares; (iii) 0 Super Voting Shares and (iv) 0 Preferred Shares. In addition, as of August 4, 2022, there were outstanding warrants to purchase an aggregate 65,456,557 Subordinate Voting Shares and outstanding options to purchase an aggregate of 29,448,120 Subordinate Voting Shares. The following summary description of our capital shares is based on the provisions of our Articles. This information is qualified entirely by reference to the applicable provisions of our Articles. For information on how to obtain copies of our Notice of Articles and Articles, which are exhibits to the registration statement of which this prospectus is a part, see “Where You Can Find More Information.”
Subordinate Voting Shares
Voting Rights. Holders of the Subordinate Voting Shares are entitled to notice of and to attend any meeting of our shareholders, except a meeting of which only holders of another particular class or series of shares shall have the right to vote. At each such meeting holders of Subordinate Voting Shares shall be entitled to one vote in respect of each Subordinate Voting Share held. Voting rights of the Subordinate Voting Shares are non-cumulative.
Alteration to Rights of Subordinate Voting Shares. As long as any Subordinate Voting Shares remain outstanding, we may not, without the consent of the holders of the Subordinate Voting Shares by separate special resolution, prejudice or interfere with any special right attached to the Subordinate Voting Shares. A special resolution means either (a) a resolution approved by two-thirds of the votes cast on the resolution at a properly called meeting of the shareholders, or (b) a resolution approved in writing by all of the shareholders holding shares that carry the right to vote on the matter at a shareholders meeting.
Dividends. Holders of Subordinate Voting Shares are entitled to receive as and when declared by the directors, dividends in cash or our property. No dividend will be declared or paid on the Subordinate Voting Shares unless we simultaneously declare or pay, as applicable, equivalent dividends (on an as-converted to Subordinate Voting Share basis) on the Multiple Voting Shares and Super Voting Shares.
Liquidation, Dissolution or Winding-Up. In the event of our liquidation, dissolution or winding-up, whether voluntary or involuntary, or in the event of any other distribution of our assets among our shareholders for the purpose of winding up our affairs, the holders of Subordinate Voting Shares are, subject to the prior rights of the holders of any shares ranking in priority to the Subordinate Voting Shares, entitled to participate ratably along with all other holders of Multiple Voting Shares (on an as-converted to Subordinate Voting Share basis), Subordinate Voting Shares and Super Voting Shares (on an as-converted to Subordinate Voting Share basis).
Rights to Subscribe; Pre-Emptive Rights. Holders of Subordinate Voting Shares are not entitled to a right of first refusal to subscribe for, purchase or receive any part of any issue of Subordinate Voting Shares, or bonds, debentures or other securities now or in the future.
Subdivision or Consolidation. No subdivision or consolidation of the Subordinate Voting Shares, Multiple Voting Shares or Super Voting Shares shall occur unless, simultaneously, the Subordinate Voting Shares, Multiple Voting Shares and Super Voting Shares are subdivided or consolidated in the same manner or such other adjustment is made so as to maintain and preserve the relative rights of the holders of the shares of each of the said classes.
Super Voting Shares
Voting Rights. Holders of Super Voting Shares are entitled to notice of and to attend at any meeting of the shareholders, except a meeting of which only holders of another particular class or series of shares shall have the right to vote. At each such meeting, holders of Super Voting Shares are entitled to ten (10) votes in respect of each Subordinate Voting Share into which such Super Voting Share could ultimately then be converted (1000 votes per Super Voting Share based on the current Conversion Ratio (as defined in the Articles)). Voting of the Super Voting Shares are non-cumulative.
98
Alteration to Rights of Super Voting Shares. As long as any Super Voting Shares remain outstanding, we may not, without the consent of the holders of the Super Voting Shares by separate special resolution, prejudice or interfere with any special right attached to the Super Voting Shares. Consent of the holders of a majority of the outstanding Super Voting Shares is required for any action that authorizes or creates shares of any class having preferences superior to or on a parity with the Super Voting Shares. In connection with the exercise of the voting rights in respect of any proposed alteration of rights, each holder of Super Voting Shares has one vote in respect of each Super Voting Share held. A special resolution means either (a) a resolution approved by two-thirds of the votes cast on the resolution at a properly called meeting of the shareholders, or (b) a resolution approved in writing by all of the shareholders holding shares that carry the right to vote on the matter at a shareholders meeting.
Dividends. Holders of Super Voting Shares have the right to receive dividends, out of any cash or other assets legally available therefor, pari passu (on an as converted to Subordinated Voting Share basis) as to dividends and any declaration or payment of any dividend on the Subordinate Voting Shares.
Liquidation, Dissolution or Winding-Up. In the event of our liquidation, dissolution or winding-up, whether voluntary or involuntary, or in the event of any other distribution of our assets among our shareholders for the purpose of winding up our affairs, holders of Super Voting Shares are, subject to the prior rights of the holders of any shares ranking in priority to the Super Voting Shares, entitled to participate ratably along with all other holders of Super Voting Shares (on an as-converted to Subordinate Voting Share basis), Subordinate Voting Shares and Multiple Voting Shares (on an as-converted to Subordinate Voting Share basis).
Rights to Subscribe; Pre-Emptive Rights. Holders of Super Voting Shares are not entitled to a right of first refusal to subscribe for, purchase or receive any part of any issue of Subordinate Voting Shares, or bonds, debentures or other securities now or in the future.
Conversion. Holders of Super Voting Shares Holders have the following conversion rights:
| (i) | Right to Convert. The Super Voting Shares each have a restricted right to convert into 100 Subordinate Voting Shares, subject to adjustments for certain customary corporate changes. The ability to convert the Super Voting Shares is subject to a restriction that the aggregate number of Subordinate Voting Shares and Super Voting Shares held of record, directly or indirectly, by residents of the U.S. (as determined in accordance with Rules 3b-4 and 12g3-2(a) under the Securities Exchange Act of 1934, as amended, may not exceed forty five percent (45%) of the aggregate number of Subordinate Voting Shares, Multiple Voting Shares and Super Voting Shares issued and outstanding after giving effect to such conversions and to a restriction on beneficial ownership of Subordinate Voting Shares exceeding certain levels. In addition, each Super Voting Share shall automatically be converted in certain circumstances described below. |
| (ii) | Automatic Conversion. A Super Voting Share will automatically be converted (without further action by the holder thereof) into one Subordinate Voting Share upon the transfer by the holder thereof to anyone other than another Founder, an immediate family member of a Founder or a transfer for purposes of estate or tax planning to a company or person that is wholly beneficially owned by a Founder or immediate family members of a Founder or which a Founder or immediate family members of a Founder are the sole beneficiaries thereof, which we refer to as a Transfer Conversion. |
99
| (iii) | Anti-Dilution Provision. The Super Voting Shares are subject to standard anti-dilution adjustments in the event the Company declares a distribution to holders of Subordinate Voting Shares, effects a recapitalization of the Subordinate Voting Shares, issues Subordinate Voting Shares as a dividend or other distribution on outstanding Subordinate Voting Shares, or subdivides or consolidates the outstanding Subordinate Voting Shares. In the event such an anti-dilution adjustment occurs, it shall be effected by adjusting the Conversion Ratio applicable to the Super Voting Shares at such time. As a result, holders of Super Voting Shares shall be entitled to (i) a proportionate share of any distribution as though they were holders of the number of Subordinate Voting Shares into which their Super Voting Shares are convertible as of the record date fixed for determination of the holders of Subordinate Voting Shares entitled to receive such distribution and (ii) receive, upon conversion of Super Voting Shares, the number of Subordinate Voting Shares or other securities or property of the Company or otherwise, to which a holder of Subordinate Voting Shares deliverable upon conversion would have been entitled in connection with a recapitalization or stock split. |
| (iv) | No Fractional Shares and Certificate as to Adjustments. No fractional Multiple Voting Shares shall be issued upon the conversion of any share or shares of Super Voting Shares and the number of Multiple Voting Shares to be issued shall be rounded up to the nearest whole Multiple Voting Share. |
Multiple Voting Shares
Voting Rights. Holders of Multiple Voting Shares are entitled to notice of and to attend at any meeting of our shareholders, except a meeting of which only holders of another particular class or series of shares have the right to vote. At each such meeting, holders of Multiple Voting Shares are entitled to one vote in respect of each Subordinate Voting Share into which such Multiple Voting Share could ultimately then be converted (10 votes per Multiple Voting Share based on the current Conversion Ratio). Voting of the Multiple Voting Shares are non-cumulative.
Alteration to Rights of Multiple Voting Shares. As long as any Multiple Voting Shares remain outstanding, we may not, without the consent of the holders of the Multiple Voting Shares and Super Voting Shares by separate special resolution, prejudice or interfere with any special right attached to the Multiple Voting Shares. In connection with the exercise of the voting rights relating to any proposed alteration of rights, each holder of Multiple Voting Shares has one vote in respect of each Multiple Voting Share held. A special resolution means either (a) a resolution approved by two-thirds of the votes cast on the resolution at a properly called meeting of the shareholders, or (b) a resolution approved in writing by all of the shareholders holding shares that carry the right to vote on the matter at a shareholders meeting.
Dividends. Holders of Multiple Voting Shares have the right to receive dividends, out of any cash or other assets legally available therefor, pari passu (on an as converted basis, assuming conversion of all Multiple Voting Shares into Subordinate Voting Shares at the Conversion Ratio) as to dividends and any declaration or payment of any dividend on the Subordinate Voting Shares. No dividend may be declared or paid on the Multiple Voting Shares unless we simultaneously declare or pay, as applicable, equivalent dividends (on an as-converted to Subordinate Voting Share basis) on the Subordinate Voting Shares and Super Voting Shares.
Liquidation, Dissolution or Winding-Up. In the event of the liquidation, dissolution or winding-up of Jushi whether voluntary or involuntary, or in the event of any other distribution of our assets among our shareholders for the purpose of winding up our affairs, holders of Multiple Voting Shares, subject to the prior rights of the holders of any shares ranking in priority to the Multiple Voting Shares, are entitled to participate ratably along with all other holders of Multiple Voting Shares (on an as-converted to Subordinate Voting Share basis), Subordinate Voting Shares and Super Voting Shares (on an as-converted to Subordinate Voting Share basis).
Rights to Subscribe; Pre-Emptive Rights. Holders of Multiple Voting Shares are not entitled to a right of first refusal to subscribe for, purchase or receive any part of any issue of Subordinate Voting Shares, or bonds, debentures or other securities now or in the future.
100
Conversion. Subject to the Conversion Restrictions described below, holders of Multiple Voting Shares Holders have the following conversion rights:
| (i) | Right to Convert. Each Multiple Voting Share is convertible, at the option of the holder thereof, at any time after the date of issuance of such share, into such number of fully paid and non-assessable Subordinate Voting Shares as is determined by multiplying the number of Multiple Voting Shares by the Conversion Ratio applicable to such share in effect on the date the Multiple Voting Share is surrendered for conversion. The initial “Conversion Ratio” for Multiple Voting Shares is one (1) Subordinate Voting Shares for each Multiple Voting Share, subject to adjustment as described below. |
| (ii) | Conversion Limitations. The Company shall not effect any conversion of Multiple Voting Shares and a holder of Multiple Voting Shares shall not have the right to convert any portion of its Multiple Voting Shares to the extent that after giving effect to such issuance after conversion, such holder (together with such holder’s Affiliates (each, an Affiliate as defined in Rule 12b-2 under the Exchange Act, and any other persons acting as a group together with the such holder or any holder’s Affiliates), would beneficially own in excess of 9.99% of the number of the Subordinate Voting Shares outstanding immediately after giving effect to the issuance of Subordinate Voting Shares issuable upon conversion of the Multiple Voting Shares subject to applicable conversion (the Beneficial Ownership Limitation). A holder of Multiple Voting Shares, upon notice to the Company, may increase or decrease the Beneficial Ownership Limitation, provided that the Beneficial Ownership Limitation in no event exceeds 19.99% of the number of the Subordinate Voting Shares outstanding immediately after giving effect to the issuance of Subordinate Voting Shares upon conversion of Multiple Voting Shares subject to the applicable conversion. Any increase in the Beneficial Ownership Limitation will not be effective until the 61st day after such notice is delivered to the Company. |
| (iii) | Mandatory Conversion. We may require each holder of Multiple Voting Shares to convert all, and not less than all, the Multiple Voting Shares at the applicable Conversion Ratio if at any time all the following conditions are satisfied (or otherwise waived by special resolution of holders of Multiple Voting Shares): |
| (A) | the Subordinate Voting Shares issuable upon conversion of all the Multiple Voting Shares are registered for resale and may be sold by the holder thereof pursuant to an effective registration statement and/or prospectus covering the Subordinate Voting Shares under the Securities Act; |
| (B) | the Company is subject to the reporting requirements of Section 13 or 15(d) of the Exchange Act; and |
| (C) | the Subordinate Voting Shares are listed or quoted (and are not suspended from trading) on a recognized North American stock exchange or by way of reverse takeover transaction on the Toronto Stock Exchange, the TSX Venture Exchange, the Canadian Securities Exchange or Aequitas NEO Exchange (or any other stock exchange recognized as such by the Ontario Securities Commission). |
| (iv) | Anti-Dilution. The Multiple Voting Shares are subject to standard anti-dilution adjustments in the event the Company declares a distribution to holders of Subordinate Voting Shares, effects a recapitalization of the Subordinate Voting Shares, issues Subordinate Voting Shares as a dividend or other distribution on outstanding Subordinate Voting Shares, or subdivides or consolidates the outstanding Subordinate Voting Shares. In the event such an anti-dilution adjustment occurs, it shall be effected by adjusting the Conversion Ratio applicable to the Multiple Voting Shares at such time. As a result, holders of Multiple Voting Shares shall be entitled to (i) a proportionate share of any distribution as though they were holders of the number of Subordinate Voting Shares into which their Multiple Voting Shares are convertible as of the record date fixed for determination of the holders of Subordinate Voting Shares entitled to receive such distribution and (ii) receive, upon conversion of Multiple Voting Shares, the number of Subordinate Voting Shares or other securities or property of the Company or otherwise, to which a holder of Subordinate Voting Shares deliverable upon conversion would have been entitled in connection with a recapitalization or stock split. |
101
| (v) | No Fractional Shares and Certificate as to Adjustments. No fractional Subordinate Voting Shares shall be issued upon the conversion of any share or shares of Multiple Voting Shares and the number of Subordinate Voting Shares to be issued shall be rounded up to the nearest whole Subordinate Voting Share. |
Preferred Shares
The voting rights, rights to dividends and rights on the liquidation, voting, conversion, exchange or reclassification, redemption, retraction or purchase for cancellation rights and other rights of the holders of the Preferred Shares will determined by our board of directors, if, as and when such Preferred Shares are issued. Some or all of such terms could be in priority or preference to, and could adversely affect the voting power or other rights of the holders of the Subordinate Voting Shares, Super Voting Shares and Multiple Voting Shares.
Note Warrants
The Note Warrants were issued in connection with the 10% Senior Secured Notes debt financing. Subscribers of the 10% Senior Secured Notes debt financing were issued Note Warrants to acquire Subordinate Voting Shares of the Company equal to 75% of the principal amount of the applicable 10% Senior Secured Note divided by an exercise price of $1.25. The Note Warrants have an expiration date of December 23, 2024. See Note 13 - Derivative Liabilities to our consolidated financial statements for the years ended December 31, 2021 and 2020 included elsewhere in this prospectus.
Provisions of British Columbia Law Governing Business Combinations
All provinces of Canada have adopted National Instrument 62-104 entitled “Take-Over Bids and Issuer Bids” and related forms to harmonize and consolidate take-over bid and issuer bid regimes nationally (NI 62-104). The Canadian Securities Administrators, or CSA, have also issued National Policy 62-203 entitled “Take-Over Bids and Issuer Bids,” or the National Policy, which contains regulatory guidance on the interpretation and application of NI 62-104 and on the conduct of parties involved in a bid. The National Policy and NI 62-104 are collectively referred to as the “Bid Regime.” The National Policy does not have the force of law, but is an indication by the CSA of what the intentions and desires of the regulators are in the areas covered by their policies. Unlike some regimes where the take-over bid rules are primarily policy-driven, in Canada the regulatory framework for take-over bids is primarily rules-based, which rules are supported by policy.
A “take-over bid” or “bid” is an offer to acquire outstanding voting or equity securities of a class made to any person who is in one of the provinces of Canada or to any securityholder of an offeree issuer whose last address as shown on the books of a target is in such province, where the securities subject to the offer to acquire, together with the securities “beneficially owned” by the offeror, or any other person acting jointly or in concert with the offeror, constitute in the aggregate 20% or more of the outstanding securities of that class of securities at the date of the offer to acquire. For the purposes of the Bid Regime, a security is deemed to be “beneficially owned” by an offeror as of a specific date if the offeror is the beneficial owner of a security convertible into the security within 60 days following that date, or has a right or obligation permitting or requiring the offeror, whether or not on conditions, to acquire beneficial ownership of the security within 60 days by a single transaction or a series of linked transactions. Offerors are also subject to early warning requirements, where an offeror who acquires “beneficial ownership of”, or control or direction over, voting or equity securities of any class of a reporting issuer or securities convertible into, voting or equity securities of any class of a target that, together with the offeror’s securities, would constitute 10% or more of the outstanding securities of that class must promptly publicly issue and file a news release containing certain prescribed information, and, within two business days, file an early warning report containing substantially the same information as is contained in the news release.
In addition, where an offeror is required to file an early warning report or a further report as described and the offeror acquires or disposes of beneficial ownership of, or the power to exercise control or direction over, an additional 2% or more of the outstanding securities of the class, or disposes of beneficial ownership of outstanding securities of the class below 10%, the offeror must issue an additional press release and file a new early warning report. Any material change in a previously filed early warning report also triggers the issuance and filing of a new press release and early warning report. During the period commencing on the occurrence of an event in respect of which an early warning report is required and terminating on the expiry of one business day from the date that the early warning report is filed, the offeror may not acquire or offer to acquire beneficial ownership of any securities of the class in respect of which the early warning report was required to be filed or any securities convertible into securities of that class. This requirement does not apply to an offeror that has beneficial ownership of, or control or direction over, securities that comprise 20% of more of the outstanding securities of the class.
Related party transactions, issuer bids and insider bids are subject to additional regulation that may differ depending on the particular jurisdiction of Canada in which it occurs.
102
Take-Over Bid Protection
There are currently no Super Voting Shares or Multiple Voting Shares issued and outstanding. Under applicable Canadian law, an offer to purchase Super Voting Shares would not necessarily require that an offer be made to purchase Subordinate Voting Shares or Multiple Voting Shares. In accordance with the rules applicable to most issuers with dual class share structures in Canada, in the event of a take-over bid, the holders of Subordinate Voting Shares or of Multiple Voting Shares must be entitled to participate on an equal footing with holders of Super Voting Shares.
As a result, if in the future we decide to issue Super Voting Shares, as a condition to the issuance of such shares, we would enter into a coattail agreement with the holders of the Super Voting Shares and Odyssey Trust Company or some other trustee, which we refer to as the “Coattail Agreement”. Such Coattail Agreement would contain provisions customary for dual class, listed corporations designed to prevent transactions that would otherwise deprive the holders of Subordinate Voting Shares or of Multiple Voting Shares of rights under applicable provincial take-over bid legislation to which they would have been entitled if the Super Voting Shares had been Subordinate Voting Shares or Multiple Voting Shares.
Among other terms and conditions that would be contained in the Coattail agreement and would be subject to negotiation and approval of our board of directors, the holders of the Super Voting Shares and the trustee, the undertakings in the Coattail Agreement would not apply to prevent a sale by any holder of Super Voting Shares if concurrently an offer is made to purchase Subordinate Voting Shares and Multiple Voting Shares that:
(a) offers a price per Subordinate Voting Share or Multiple Voting Share (on an as converted to Subordinate Share basis) at least as high as the highest price per share paid pursuant to the take-over bid for the Super Voting Shares (on an as converted Subordinate Voting Share basis);
(b) provides that the percentage of outstanding Subordinate Voting Shares or Multiple Voting Shares be taken up (exclusive of shares owned immediately prior to the offer by the offeror or persons acting jointly or in concert with the offeror) is at least as high as the percentage of Super Voting Shares to be sold (exclusive of Super Voting Shares owned immediately prior to the offer by the offeror and persons acting jointly or in concert with the offeror);
(c) has no condition attached other than the right not to take up and pay for the Subordinate Voting Shares or Multiple Voting Shares tendered if no shares are purchased pursuant to the offer for Super Voting Shares; and
(d) is in all other material respects identical to the offer for Super Voting Shares.
Other Important Provisions in our Articles
The following is a summary of certain important provisions of our Articles. Please note that this is only a summary, is not intended to be exhaustive and is qualified in its entirety by reference to our Articles. For further information, please refer to the full version of our Articles which have been filed as an exhibit to the registration statement of which this prospectus forms a part.
Objects and Purposes of the Company
Our Articles do not contain and are not required to contain a description of our objects and purposes. There is no restriction contained in our Articles on the business that we may carry on.
General Borrowing Power
Pursuant to section 8 our Articles, we may, if authorized by our board of directors: (i) borrow money in the manner and amount, on the security, from the sources, and on the terms and conditions that our directors consider appropriate; (ii) issue bonds, debentures and other debt obligations either outright or as security for any liability or obligation of us or any other person and at such discounts or premiums and on such other terms as our directors consider appropriate; (iii) guarantee the repayment of money by any other person or the performance of any other obligation by any other person; and (iv) mortgage, charge, whether by way of a specific or floating charge, grant a security interest in, or give other security on, the whole or any part of the present and future assets and undertaking of us.
103
Share Rights
See the discussion in the section of this prospectus entitled “Description of Capital Stock” for a summary of our authorized capital and the rights attached to our super voting shares, multiple voting shares and subordinate voting shares.
Alteration of Authorized Share Structure
Pursuant to Section 9 of our Articles, and subject to the Business Corporation Act (British Columbia), our board of directors may, by resolution: (a) create one or more classes or series of shares or, if none of the shares of a class or series of shares are allotted or issued, eliminate that class or series of shares; (b) increase, reduce or eliminate the maximum number of shares that our company is authorized to issue out of any class or series of shares or establish a maximum number of shares that our company is authorized to issue out of any class or series of shares for which no maximum is established: (c) alter the identifying name of any of our shares; (d) subdivide or consolidate all or any of our unissued, or fully paid issued, shares; (e) if our company is authorized to issue shares of a class of shares with par value: (A) decrease the par value of those shares; or (B) if none of the shares of that class of shares are allotted or issued, increase the par value of those shares; (f) change all or any of our unissued, or fully paid issued, shares with par value into shares without par value or any of its unissued shares without par value into shares with par value: or (g) otherwise alter our shares or authorized share structure when required or permitted to do so by the Business Corporations Act (British Columbia). Our board of directors may, by resolution, authorize an alternation of our Notice of Articles in order to change our company’s name or adopt or change any translation of that name.
Quorum
Under our Articles, the quorum for the transaction of business at a meeting of our board of directors may be set by the directors and, if not so set, is deemed to be set at two directors or, if the number of directors is set at one, is deemed to be set at one director. Under our Articles, the quorum for the transaction of business at a meeting of our shareholders is two persons who are, or who represent by proxy, shareholders entitled to vote at the meeting, who hold in the aggregate, at least 5% of our issued shares entitled to vote at such meeting.
Impediments to Change of Control
Our Articles do not contain any change of control limitations with respect to a merger, acquisition or corporate restructuring that involves us.
Forum for Adjudication of Certain Disputes
Pursuant to section 28 of our Articles, unless our company approves or consents in writing to the selection of an alternative forum, the courts of the province of British Columbia and appellate courts therefrom shall be the sole and exclusive forum for: (a) any derivative action or proceeding brought on behalf of our company, (b) any action asserting a claim of breach of a fiduciary duty owed by any director or officer of our company to our company, (c) any action asserting a claim arising pursuant to any provision of the Business Corporations Act (British Columbia) or the Notice of Articles or Articles of our company (as either may be amended from time to time); or (d) any action asserting a claim otherwise related to the relationships among our company, its affiliates and their respective shareholders, directors and/or officers, but this does not include claims related to the business carried on by our company or such affiliates; provided however it is uncertain whether such provision would apply to actions arising under U.S. federal securities laws, and if it does, whether a British Columbia Court would enforce such provision, since in accordance with Section 27 of the Exchange Act, United States federal courts shall have jurisdiction over all suits and any action brought to enforce any duty or liability created by the Exchange Act or the rules and regulations thereunder and that in accordance with Section 22 of the Securities Act, United States federal and state courts shall have concurrent jurisdiction over all suits brought to enforce any duty or liability created by the Securities Act or the rules and regulations thereunder. Our shareholders will not be deemed, by operation of the choice of forum provision, to have waived our obligation to comply with all applicable United States federal securities laws and the rules and regulations thereunder.
Compliance Provisions
Section 29 of our Articles contains certain provisions, which we refer to as the Compliance Provisions, which include a combination of certain remedies such as an automatic suspension of voting and/or dividend rights, a discretionary right to force a share transfer to a third party and/or a discretionary redemption right in favor of our company.
104
The purpose of the Compliance Provisions are to provide our company with a means of protecting itself from having a shareholder, or as determined by our board of directors, a group of shareholders acting jointly or in concert, with an ownership interest of, whether of record or beneficially (or having the power to exercise control or direction over) (Owning or Controlling), five percent (5%) or more of the issued and outstanding shares of our company, or such other number as is determined by our board of directors from time to time, and: (i) who a governmental authority granting licenses to, or otherwise governing the operations of, our company or its subsidiaries has determined to be unsuitable to own any of our company’s shares; (ii) whose ownership of any of shares may reasonably result in the loss, suspension or revocation (or similar action) with respect to any licenses or permits relating to our company’s or its subsidiaries’ conduct of business (being the conduct of any activities relating to the cultivation, manufacturing and dispensing of cannabis and cannabis-derived products in the U.S., which include the owning and operating of cannabis licenses) or in our company being unable to obtain any new licenses or permits in the normal course, all as determined by our board of directors; or (iii) who have not been determined by the applicable regulatory authority to be an acceptable person or otherwise have not received the requisite consent of such regulatory authority to own shares, in each case within a reasonable time period acceptable to our board of directors or prior to acquiring any shares, as applicable.
Advance Notice Policy
We have adopted an advance notice policy (the Advance Notice Policy) which provides that shareholders seeking to nominate candidates for election as directors other than pursuant to a proposal or requisition of shareholders made in accordance with the provisions of the Business Corporations Act (British Columbia), must provide timely written notice to our Corporate Secretary. To be timely, a shareholder’s notice must be received (i) in the case of an annual meeting of shareholders, not later than close of business on the 30th day prior to the date of the annual meeting of shareholders; provided, however, that in the event that the annual meeting of shareholders is to be held on a date that is less than 50 days after the date on which the first public announcement of the date of the annual meeting was made, notice by the shareholder must be received not later than the close of business on the 10th day following the date of such public announcement; and (ii) in the case of a special meeting (which is not also an annual meeting) of shareholders called for any purpose which includes the election of directors to the board of directors, not later than the close of business on the 15th day following the day on which the first public announcement of the date of the special meeting was made. The Advance Notice Policy also prescribes the proper written form for a shareholder’s notice.
Ownership and Exchange Controls
Limitations on the ability to acquire and hold our shares may be imposed by the Competition Act (Canada). This legislation establishes a pre-merger notification regime for certain types of merger transactions that exceed certain statutory shareholding and financial thresholds. Transactions that are subject to notification cannot be closed until the required materials are filed and the applicable statutory waiting period has expired or been waived by the Commissioner of Competition, or the Commissioner. Further, the Competition Act (Canada) permits the Commissioner to review any acquisition of control over or of a significant interest in us, whether or not it is subject to mandatory notification. This legislation grants the Commissioner jurisdiction, for up to one year, to challenge this type of acquisition before the Canadian Competition Tribunal if it would, or would be likely to, substantially prevent or lessen competition in any market in Canada.
105
CERTAIN CANADIAN FEDERAL INCOME TAX
CONSIDERATIONS FOR UNITED STATES RESIDENTS
The following is, at the date hereof, a summary of certain Canadian federal income tax considerations generally applicable to a holder of Subordinate Voting Shares who, at all relevant times, (A) for the purposes of the Income Tax Act (Canada) (the Canadian Tax Act), (i) is not resident, or deemed to be resident, in Canada, (ii) deals at “arm’s length” with us, (iii) holds all Subordinate Voting Shares as capital property, (iv) does not use or hold any of the Subordinate Voting Shares in the course of carrying on, or otherwise in connection with, a business carried on or deemed to be carried on in Canada, (v) is not an insurer who carries on an insurance business in Canada and elsewhere, and (vi) is not an “authorized foreign bank” (as defined in the Canadian Tax Act), and (B) for the purposes of the Canada-U.S. Tax Convention (1980) (the Tax Treaty), (i) is a resident of the U.S., (ii) has never been a resident of Canada, and (iii) who qualifies for the full benefits of the Tax Treaty. Holders of Subordinate Voting Shares who meet all of the above criteria are referred to herein as “U.S. Holders”, and this summary only addresses such U.S. Holders.
106
This summary is based on the current provisions of the Canadian Tax Act in force as of the date hereof, the regulations thereunder in force at the date hereof (the Regulations), the current provisions of the Tax Treaty, in force as of the date hereof, and an understanding of the administrative policies and assessing practices of the Canada Revenue Agency published in writing prior to the date hereof. This summary takes into account all specific proposals to amend the Canadian Tax Act and Regulations publicly announced by or on behalf of the Minister of Finance (Canada) prior to the date hereof, which we refer to as the Proposed Amendments, and assumes that such Proposed Amendments will be enacted in the form proposed. However, such Proposed Amendments might not be enacted in the form proposed, or at all. This summary does not otherwise take into account or anticipate any changes in law or administrative or assessing practices, whether by legislative, governmental or judicial decision or action, nor does it take into account tax laws of any province or territory of Canada or of any other jurisdiction outside Canada, which may differ significantly from those discussed in this summary.
For the purposes of the Canadian Tax Act, all amounts relating to the acquisition, holding or disposition of Subordinate Voting Shares generally must be converted into Canadian dollars, including dividends, adjusted cost base and proceeds of disposition, using the single daily exchange rate as quoted by the Bank of Canada for the relevant day, or such other rate of exchange that is acceptable to the Canada Revenue Agency.
THIS SUMMARY IS OF A GENERAL NATURE ONLY AND IS NOT INTENDED TO BE, NOR SHOULD IT BE CONSTRUED TO BE, LEGAL OR TAX ADVICE TO ANY PARTICULAR U.S. HOLDER, AND NO REPRESENTATION WITH RESPECT TO THE CANADIAN FEDERAL INCOME TAX CONSEQUENCES TO ANY PARTICULAR U.S. HOLDER OR PROSPECTIVE U.S. HOLDER IS MADE. THIS SUMMARY IS NOT EXHAUSTIVE OF ALL CANADIAN FEDERAL INCOME TAX CONSIDERATIONS. ACCORDINGLY, ALL PROSPECTIVE HOLDERS (INCLUDING U.S. HOLDERS AS DEFINED ABOVE) SHOULD CONSULT WITH THEIR OWN TAX ADVISORS FOR ADVICE WITH RESPECT TO THEIR OWN PARTICULAR CIRCUMSTANCES.
Withholding Tax on Dividends
Amounts paid or credited or deemed to be paid or credited as, on account or in lieu of payment of, or in satisfaction of, dividends on Subordinate Voting Shares to a U.S. Holder will be subject to Canadian withholding tax. Under the Canadian Tax Act, the rate of withholding is 25% of the gross amount of the dividend. Under the Tax Treaty, the withholding tax rate on any such dividend beneficially owned by a U.S. Holder is generally reduced to 15% or, in the case of an eligible U.S. Holder that is a U.S. company that beneficially owns at least 10% of the voting stock of us, to 5% of the gross amount of such dividends.
Dispositions of Subordinate Voting Shares
A U.S. Holder who disposes, or is deemed to have disposed, of Subordinate Voting Shares will not be subject to income tax under the Canadian Tax Act in respect of any capital gain realized on such disposition or deemed disposition unless, at the time of such disposition or deemed disposition, the Subordinate Voting Shares are or are deemed to be “taxable Canadian property” (as defined in the Canadian Tax Act) to the U.S. Holder, and the gain is not exempt from tax pursuant to the terms of the Tax Treaty.
Provided that the Subordinate Voting Shares are listed on a “designated stock exchange” as defined in the Canadian Tax Act (which currently includes the Canadian Securities Exchange) at the time of disposition, the Subordinate Voting Shares will generally not constitute taxable Canadian property of the U.S. Holder at that time, unless at any time during the 60-month period immediately preceding the disposition, the following two conditions are met concurrently: (a) one or any combination of (i) the U.S. Holder, (ii) persons with whom the U.S. Holder did not deal at arm’s length, or (iii) partnerships in which the U.S. Holder or such non-arm’s length persons held a membership interest (either directly or indirectly through one or more partnerships), owned 25% or more of the issued shares of any class or series of the capital stock of us; and (b) more than 50% of the fair market value of the Subordinate Voting Shares was derived directly or indirectly from one or any combination of real or immovable property situated in Canada, “Canadian resource properties” (as defined in the Canadian Tax Act), “timber resource properties” (as defined in the Canadian Tax Act) or an option in respect of, an interest in, or for civil law purposes, a right in, any such property, whether or not such property exists. The Subordinate Voting Shares may also be deemed to be taxable Canadian property to a U.S. Holder for purposes of the Canadian Tax Act in certain circumstances.
107
Non-Resident Holders whose Subordinate Voting Shares are taxable Canadian property should consult their own tax advisors.
108
CERTAIN MATERIAL U.S. FEDERAL INCOME TAX
CONSIDERATIONS FOR NON-U.S. HOLDERS
The following discussion is a summary of certain U.S. federal income tax considerations applicable to Non-U.S. Holders (as defined below) with respect to their purchase, ownership and disposition of Subordinate Voting Shares. This discussion is for general information only and is not tax advice. Accordingly, all prospective Non-U.S. Holders of our Subordinate Voting Shares should consult their tax advisors with respect to the U.S. federal, state and local and non-U.S. tax consequences of the purchase, ownership and disposition of our Subordinate Voting Shares.
This discussion is based on current provisions of the Code, existing and proposed U.S. Treasury Regulations promulgated thereunder, current administrative rulings and judicial decisions, all as in effect as of the date of this prospectus, all of which are subject to change or to differing interpretation, possibly with retroactive effect. Any change could alter the tax consequences to Non-U.S. Holders described in this prospectus. We assume in this discussion that a Non-U.S. Holder holds shares of our Subordinate Voting Shares as a capital asset (generally, property held for investment).
This discussion does not address all aspects of U.S. federal income taxation that may be relevant to a particular Non-U.S. Holder in light of that Non-U.S. Holder’s individual circumstances nor does it address, except to the limited extent discussed below, any aspects of U.S. federal estate or gift taxes, or state, local or non-U.S. taxes. This discussion also does not consider any specific facts or circumstances that may apply to a Non-U.S. Holder and does not address the special tax rules applicable to particular Non-U.S. Holders, such as:
| • | banks, insurance companies and other financial institutions; |
| • | tax-exempt organizations; | |
| • | holders that own or are deemed to own more than 5% of our capital stock |
| • | financial institutions; |
| • | brokers traders or dealers in securities or currencies; |
| • | regulated investment companies; |
| • | pension plans; |
| • | controlled foreign corporations, passive foreign investment companies and corporations that accumulate earnings to avoid U.S. federal income tax; |
| • | passive foreign investment companies; |
| • | persons subject to the U.S. federal alternative minimum tax or the 3.8% tax on net investment income; | |
| • | holders who hold or receive our securities pursuant to the exercise of employee stock options or otherwise as compensation; |
109
| • | “qualified foreign pension funds” as defined in Section 897(l)(2) of the Code and entities all of the interests of which are held by one or more qualified foreign pension funds; |
| • | owners that hold our Subordinate Voting Shares as part of a straddle, hedge, conversion transaction, synthetic security or other integrated investment; and |
| • | certain U.S. expatriates. |
In addition, this discussion does not address the tax treatment of partnerships (or entities or arrangements treated as partnerships) or other pass-through entities, or persons who hold our Subordinate Voting Shares through partnerships (or entities or arrangements treated as partnerships) or other pass-through entities, in each case for U.S. federal income tax purposes. A partner in a partnership or other pass-through entity that will hold our Subordinate Voting Shares should consult his, her or its tax advisor regarding the tax consequences of acquiring, holding and disposing of our Subordinate Voting Shares through a partnership (or entity or arrangement treated as a partnership) or other pass-through entity, as applicable.
We have not sought and will not seek any ruling from the IRS with respect to the statements made and the conclusions reached in the following discussion. There can be no assurance that the IRS will not challenge one or more of the tax consequences described herein, or that any such challenge would not be sustained by a court.
NON-U.S. HOLDERS ARE URGED TO CONSULT THEIR TAX ADVISORS WITH RESPECT TO THE APPLICATION OF THE U.S. FEDERAL TAX LAWS TO THEIR PURCHASE, OWNERSHIP AND DISPOSITION OF OUR SUBORDINATE VOTING SHARES IN LIGHT OF THEIR PARTICULAR SITUATIONS, AS WELL AS ANY TAX CONSEQUENCES ARISING UNDER THE LAWS OF ANY STATE, LOCAL, NON-U.S. OR OTHER TAXING JURISDICTION OR UNDER ANY APPLICABLE TAX TREATY.
Non-U.S. Holder Defined
For purposes of this discussion, a Non-U.S. Holder means a beneficial owner of our Subordinate Voting Shares that, for U.S. federal income tax purposes, is an individual, corporation, estate or trust that is not a U.S. person. For purposes of this discussion, a U.S. person is:
| • | an individual who is a citizen or resident of the U.S. for U.S. federal income tax purposes; |
| • | a corporation, or any other entity or organization taxable as a corporation for U.S. federal income tax purposes, created or organized in the U.S. or under the laws of the U.S., any state thereof or the District of Columbia; |
| • | an estate, the income of which is subject to U.S. federal income tax regardless of its source; or |
| • | a trust if (1) a U.S. court is able to exercise primary supervision over the trust’s administration and one or more U.S. persons have the authority to control all of the trust’s substantial decisions or (2) the trust has a valid election in effect to be treated as a U.S. person. |
We are and expected to continue to be a Canadian corporation as of the date of this registration statement. We are treated as a Canadian resident company under the Canadian Tax Act, as amended, and are subject to Canadian income taxes.
We are also treated as a U.S. corporation subject to U.S. federal income tax pursuant to Section 7874 of the Code and are also subject to U.S. federal income tax on our worldwide income. As a result, we are subject to taxation both in Canada and the U.S. A number of material U.S. federal income tax consequences may result from our classification under Section 7874 of the Code, and this summary is not intended to describe all such U.S. federal income tax consequences. Section 7874 of the Code and the Treasury Regulations promulgated thereunder do not address all the possible tax consequences that arise from our treatment as a U.S. corporation for U.S. federal income tax purposes. Accordingly, there may be additional or unforeseen U.S. federal income tax consequences that are not discussed in this summary. Each holder should seek tax advice, based on such shareholder’s particular circumstances, from an independent tax advisor.
110
Distributions on Our Subordinate Voting Shares
As described in the section entitled “Dividend Policy,” we have not made distributions on our Subordinate Voting Shares in the past and currently do not plan to make any distributions for the foreseeable future. However, if we do make distributions of cash or property on our Subordinate Voting Shares, those payments generally will constitute dividends for U.S. federal income tax purposes to the extent paid from our current or accumulated earnings and profits, as determined under U.S. federal income tax principles. If a distribution exceeds our current and accumulated earnings and profits, the excess will be treated as a tax-free return of the Non-U.S. Holder’s investment, up to such holder’s tax basis in the Subordinate Voting Shares. Any remaining excess will be treated as capital gain, subject to the tax treatment described below in “—Gain on Sale, Exchange or Other Disposition of Our Subordinate Voting Shares.”
Subject to the discussion below on backup withholding and FATCA (defined below), dividends paid to a Non-U.S. Holder generally will be subject to withholding of U.S. federal income tax at a 30.0% rate or such lower rate as may be specified by an applicable income tax treaty. A Non-U.S. Holder of our Subordinate Voting Shares who claims the benefit of an applicable income tax treaty generally will be required to provide a properly executed IRS Form W-8BEN or W-8BEN-E (or other appropriate version of IRS Form W-8 or successor form) and satisfy applicable certification and other requirements. Non-U.S. Holders are urged to consult their tax advisors regarding their entitlement to benefits under any applicable income tax treaty.
Dividends that are treated as effectively connected with a trade or business conducted by a Non-U.S. Holder within the U.S. and, if an applicable income tax treaty so provides, that are attributable to a permanent establishment or a fixed base maintained by the Non-U.S. Holder within the U.S., are generally exempt from the 30.0% withholding tax if the Non-U.S. Holder satisfies applicable certification and disclosure requirements by providing a properly executed IRS Form W-8ECI (or successor form). However, such U.S. effectively connected income, net of specified deductions and credits, is taxed at the same graduated U.S. federal income tax rates applicable to U.S. persons. In addition, any U.S. effectively connected income received by a Non-U.S. Holder that is a corporation (or other entity that is treated as a corporation for U.S. federal income tax purposes) may also, under certain circumstances, be subject to an additional U.S. federal branch profits tax at a 30.0% rate or such lower rate as may be specified by an applicable income tax treaty.
A Non-U.S. Holder that is eligible for a reduced rate of U.S. withholding tax under an income tax treaty may obtain a refund or credit of any excess amounts withheld by timely filing a U.S. federal income tax return with the IRS.
Gain on Sale, Exchange or Other Disposition of Our Subordinate Voting Shares
Subject to the discussion below on backup withholding and FATCA, a Non-U.S. Holder generally will not be subject to U.S. federal income tax on any gain realized upon such holder’s sale, exchange or other disposition of our Subordinate Voting Shares unless:
| • | the gain is effectively connected with the Non-U.S. Holder’s conduct of a U.S. trade or business and, if an applicable income tax treaty so provides, is attributable to a permanent establishment or a fixed base maintained by such Non-U.S. Holder in the U.S., in which case the Non-U.S. Holder generally will be taxed at the graduated U.S. federal income tax rates applicable to U.S. persons and, if the Non-U.S. Holder is a corporation (or an entity treated as a corporation for U.S. federal income tax purposes), it also may be subject to a U.S. federal branch profits tax at a rate of 30.0% (or such lower rate as may be specified by an applicable income tax treaty) on such effectively connected gain; |
111
| • | the Non-U.S. Holder is a nonresident alien individual for U.S. federal income tax purposes who is present in the U.S. for 183 days or more in the taxable year of the disposition and certain other conditions are met, in which case the Non-U.S. Holder will be subject to a 30.0% tax (or such lower rate as may be specified by an applicable income tax treaty) on the net gain derived from the disposition, which may be offset by certain U.S. source capital losses of the Non-U.S. Holder, if any; or |
| • | we are, or have been, at any time during the five-year period preceding such disposition (or the Non-U.S. Holder’s holding period, if shorter) a U.S. real property holding corporation for U.S. federal income tax purposes. Generally, a corporation is a U.S. real property holding corporation only if the fair market value of its U.S. real property interests equals or exceeds 50.0% of the sum of the fair market value of its worldwide real property interests plus the fair market value of any other of its assets used or held for use in a trade or business. Although there can be no assurance, we do not believe that we are, or have been, a U.S. real property holding corporation, or that we are likely to become one in the future. Even if we are or were to become a U.S. real property holding corporation, gains realized by a Non-U.S. Holder on a disposition of our Subordinate Voting Shares will not be subject to U.S. federal income tax under this rule if our Subordinate Voting Shares is regularly traded on an established securities market and the Non-U.S. Holder holds no more than 5.0% of our outstanding Subordinate Voting Shares, directly or indirectly, during the shorter of the 5-year period ending on the date of the disposition or the period that the Non-U.S. Holder held our Subordinate Voting Shares. No assurance can be provided that our Subordinate Voting Shares will be regularly traded on an established securities market for purposes of the rules described above. |
U.S. Federal Estate Tax
Property having a U.S. situs generally is includible in the gross estate of an individual Non-U.S. Holder for U.S. federal estate tax purposes. Because we are a U.S. corporation, our Subordinate Voting Shares will be U.S. situs property for U.S. federal estate tax purposes and, therefore, generally will be included in the gross estate of an individual who is a Non-U.S. Holder at the time of his or her death, unless an applicable estate tax treaty provides otherwise.
Backup Withholding and Information Reporting
We must report annually to the IRS and to each Non-U.S. Holder payments of dividends on our Subordinate Voting Shares to such holder and the tax withheld, if any, with respect to such dividends, along with certain other information. Non-U.S. Holders may have to comply with specific certification procedures to establish that the holder is not a U.S. person in order to avoid backup withholding with respect to dividends on our Subordinate Voting Shares. Dividends paid to Non-U.S. Holders subject to the U.S. withholding tax, as described above in “—Distributions on our Subordinate Voting Shares,” generally will be exempt from U.S. backup withholding.
Information reporting and backup withholding generally will apply to the proceeds of a disposition of our Subordinate Voting Shares by a Non-U.S. Holder effected by or through the U.S. office of any broker, U.S. or non-U.S., unless the holder certifies its status as a Non-U.S. Holder and satisfies certain other requirements, or otherwise establishes an exemption. Generally, information reporting and backup withholding will not apply to a payment of disposition proceeds to a Non-U.S. Holder where the transaction is effected outside the U.S. through a non-U.S. office of a broker. However, for information reporting purposes, dispositions effected through a non-U.S. office of a broker with substantial U.S. ownership or operations generally will be treated in a manner similar to dispositions effected through a U.S. office of a broker. Non-U.S. Holders should consult their tax advisors regarding the application of the information reporting and backup withholding rules to them.
Copies of information returns may be made available to the tax authorities of the country in which the Non-U.S. Holder resides or is incorporated under the provisions of a specific treaty or other agreement.
Backup withholding is not an additional tax. Any amounts withheld under the backup withholding rules from a payment to a Non-U.S. Holder can be refunded or credited against the Non-U.S. Holder’s U.S. federal income tax liability, if any, provided that an appropriate claim is timely filed with the IRS.
112
Foreign Account Tax Compliance Act (FATCA)
Sections 1471 through 1474 of the Code and related Treasury regulations and guidance, commonly referred to as FATCA, generally imposes a U.S. federal withholding tax at a rate of 30.0% on certain payments (including on dividends on our Subordinate Voting Shares) that are made to certain non-U.S. entities (including foreign financial institutions and non-financial foreign entities, both as specifically defined under FATCA), unless such non-U.S. entities establish that they are compliant with or exempt from FATCA. To comply with FATCA, a foreign financial institution generally is required to register with the IRS, collect and provide to tax authorities information regarding U.S. account holders of such institution (including certain equity and debt holders of such institution, as well as certain account holders that are non-U.S. entities with U.S. owners), and provide withholding agents with a certification that it is compliant with FATCA. A non-financial foreign entity generally is required to provide withholding agents with either a certification that it does not have any substantial direct or indirect U.S. owners or information regarding substantial direct and indirect U.S. owners of the entity, or otherwise establishes an exemption from FATCA. An intergovernmental agreement between the U.S. and an applicable foreign country may, however, modify these requirements and these requirements are different from and in addition to the certification requirements described elsewhere in this discussion.
Subject to the recent Treasury Regulations described in the following sentence, FATCA applies to dividends paid on our Subordinate Voting Shares and to gross proceeds from sales or other dispositions of our Subordinate Voting Shares. The U.S. Treasury Department has proposed regulations, on which taxpayers generally may rely until final Treasury Regulations are issued, and which eliminate FATCA federal withholding tax of 30% applicable to the gross proceeds of a sale or other disposition of our Subordinate Voting Shares. Amounts withheld under FATCA with respect to income that is also subject to the general U.S. federal withholding tax, as discussed above in “—Distributions on Our Subordinate Voting Shares,” will be applied against and reduce the amount of such other withholding tax. Prospective investors should consult their tax advisors regarding the possible implications of FATCA on their investment in our Subordinate Voting Shares.
The Subordinate Voting Shares beneficially owned by the Selling Shareholders covered by this prospectus may be offered and sold from time to time by the Selling Shareholders. The term “Selling Shareholders” includes donees, pledgees, transferees or other successors in interest selling shares received after the date of this prospectus from a Selling Shareholder as a gift, pledge, partnership distribution or other non-sale related transfer. The Selling Shareholders will act independently of us in making decisions with respect to the timing, manner and size of each sale. Such sales may be made on one or more exchanges or in the over-the-counter market or otherwise, at prices and under terms then prevailing or at prices related to the then current market price or in negotiated transactions. The Selling Shareholders may sell or dispose of their shares by one or more of, or a combination of, the following methods:
| • | purchases by a broker-dealer as principal and resale by such broker-dealer for its own account pursuant to this prospectus; |
| • | ordinary brokerage transactions and transactions in which the broker solicits purchasers; |
| • | block trades in which the broker-dealer so engaged will attempt to sell the shares as agent but may position and resell a portion of the block as principal to facilitate the transaction; |
| • | an exchange distribution in accordance with the rules of any stock exchange on which the securities are listed; |
113
| • | through trading plans entered into by a Selling Shareholder pursuant to Rule 10b5-1 under the Exchange Act that are in place at the time of an offering pursuant to this prospectus and any applicable prospectus supplement hereto that provide for periodic sales of their securities on the basis of parameters described in such trading plans; |
| • | to or through underwriters; |
| • | in “at the market” offerings, as defined in Rule 415 under the Securities Act, at negotiated prices, at prices prevailing at the time of sale or at prices related to such prevailing market prices, including sales made directly on a national securities exchange or sales made through a market maker other than on an exchange or other similar offerings through sales agents; |
| • | in privately negotiated transactions; |
| • | in short sales; |
| • | through the distribution of the securities by any Selling Shareholder to its partners, members or shareholders; |
| • | in options transactions; and |
| • | through a combination of any of the above methods of sale and, in addition, any shares that qualify for sale pursuant to Rule 144 may be sold under Rule 144 rather than pursuant to this prospectus. |
The Selling Shareholders will pay all brokerage fees and commissions and similar expenses. We will pay all expenses (except brokerage fees and commissions and similar expenses) relating to the registration of the Subordinate Voting Shares with the SEC.
To the extent required, this prospectus may be amended or supplemented from time to time to describe a specific plan of distribution. In connection with distributions of the shares or otherwise, the Selling Shareholders may enter into hedging transactions with broker-dealers or other financial institutions. In connection with such transactions, broker-dealers or other financial institutions may engage in short sales of Subordinate Voting Shares in the course of hedging the positions they assume with Selling Shareholders. The Selling Shareholders may also sell the Subordinate Voting Shares short and redeliver the shares to close out such short positions. The Selling Shareholders may also enter into option or other transactions with broker-dealers or other financial institutions which require the delivery to such broker-dealer or other financial institution of shares offered by this prospectus, which shares such broker-dealer or other financial institution may resell pursuant to this prospectus (as supplemented or amended to reflect such transaction). The Selling Shareholders may also pledge shares to a broker-dealer or other financial institution, and, upon a default, such broker-dealer or other financial institution, may effect sales of the pledged shares pursuant to this prospectus (as supplemented or amended to reflect such transaction).
A Selling Shareholder may enter into derivative transactions with third parties, or sell securities not covered by this prospectus to third parties in privately negotiated transactions. If the applicable prospectus supplement indicates, in connection with those derivatives, the third parties may sell securities covered by this prospectus and the applicable prospectus supplement, including in short sale transactions. If so, the third party may use securities pledged by any Selling Shareholder or borrowed from any Selling Shareholder or others to settle those sales or to close out any related open borrowings of stock, and may use securities received from any Selling Shareholder in settlement of those derivatives to close out any related open borrowings of stock. The third party in such sale transactions will be an underwriter and will be identified in the applicable prospectus supplement (or a post-effective amendment). In addition, any Selling Shareholder may otherwise loan or pledge securities to a financial institution or other third party that in turn may sell the securities short using this prospectus. Such financial institution or other third party may transfer its economic short position to investors in our securities or in connection with a concurrent offering of other securities.
114
In effecting sales, broker-dealers or agents engaged by the Selling Shareholders may arrange for other broker-dealers to participate. Broker-dealers or agents may receive commissions, discounts or concessions from the Selling Shareholders in amounts to be negotiated immediately prior to the sale.
In offering the shares covered by this prospectus, the Selling Shareholders and any broker-dealers who execute sales for the Selling Shareholders may be deemed to be “underwriters” within the meaning of the Securities Act in connection with such sales. Any profits realized by the Selling Shareholders and the compensation of any broker-dealer may be deemed to be underwriting discounts and commissions.
In order to comply with the securities laws of certain states, if applicable, the shares must be sold in such jurisdictions only through registered or licensed brokers or dealers. In addition, in certain states the shares may not be sold unless they have been registered or qualified for sale in the applicable state or an exemption from the registration or qualification requirement is available and is complied with.
We will make copies of this prospectus available to the Selling Shareholders for the purpose of satisfying the prospectus delivery requirements of the Securities Act. The Selling Shareholders may indemnify any broker-dealer that participates in transactions involving the sale of the shares against certain liabilities, including liabilities arising under the Securities Act.
At the time a particular offer of shares is made, if required, a prospectus supplement will be distributed that will set forth the number of shares being offered and the terms of the offering, including the name of any underwriter, dealer or agent, the purchase price paid by any underwriter, any discount, commission and other item constituting compensation, any discount, commission or concession allowed or reallowed or paid to any dealer, and the proposed selling price to the public.
The validity of the securities being offered hereby as a matter of Canadian law will be passed upon for us by Stikeman Elliott LLP.
The consolidated financial statements appearing in this prospectus and registration statement as of and for the years ended December 31, 2021 and 2020 have been audited by Marcum LLP, an independent registered public accounting firm, as stated in their report appearing elsewhere herein and are included in reliance upon such report and upon the authority of such firm as experts in accounting and auditing.
WHERE YOU CAN FIND MORE INFORMATION
We have filed with the SEC a registration statement on Form S-1 under the Securities Act with respect to the Subordinate Voting Shares sold in this offering. This prospectus, which constitutes part of the registration statement, does not include all of the information contained in the registration statement and the exhibits, schedules and amendments to the registration statement. Some items are omitted in accordance with the rules and regulations of the SEC. For further information with respect to us and our Subordinate Voting Shares, we refer you to the registration statement and to the exhibits and schedules to the registration statement filed as part of the registration statement. Statements contained in this prospectus about the contents of any contract or any other document filed as an exhibit are not necessarily complete, and, and in each instance, we refer you to the copy of the contract or other documents filed as an exhibit to the registration statement. Each of these statements is qualified in all respects by this reference.
115
The SEC maintains an internet website, which is located at www.sec.gov, that contains reports, proxy and information statements and other information regarding issuers that file electronically with the SEC. You may access the registration statement of which this prospectus is a part at the SEC’s internet website. Information contained on or accessible through the SEC’s website is not a part of this prospectus, and the inclusion of the SEC’s website address in this prospectus is an inactive textual reference only.
Upon the date the registration statement of which this prospectus forms a part is declared effective, we will become subject to the informational and periodic reporting requirements of the Exchange Act. We will fulfill our obligations with respect to such requirements by filing periodic reports and other information with the SEC. We intend to furnish our shareholders with annual reports containing financial statements certified by an independent registered public accounting firm. We also maintain a website at www.jushico.com, at which you may access these materials free of charge as soon as reasonably practicable after they are electronically filed with, or furnished to, the SEC. We do not incorporate the information on our website into this prospectus or any supplement to this prospectus and you should not consider any information on, or that can be accessed through, our website as part of this prospectus or any supplement to this prospectus.
In addition, our periodic and current reports are available, free of charge, after the material is electronically filed with, or furnished to, the Canadian securities regulators on SEDAR at www.sedar.com.
116
JUSHI HOLDINGS INC.
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
| F-1 |
CONDENSED CONSOLIDATED BALANCE SHEETS
(in thousands of U.S. dollars, except share amounts)
March 31, 2022 (unaudited) | December 31, 2021 | |||||||
| ASSETS | ||||||||
| CURRENT ASSETS: | ||||||||
| Cash and cash equivalents | $ | 75,717 | $ | 94,962 | ||||
| Accounts receivable, net | 1,460 | 3,200 | ||||||
| Inventories | 39,282 | 43,319 | ||||||
| Prepaid expenses and other current assets | 6,174 | 12,875 | ||||||
| Total current assets | $ | 122,633 | $ | 154,356 | ||||
| NON-CURRENT ASSETS: | ||||||||
| Property, plant and equipment, net | $ | 152,943 | $ | 137,280 | ||||
| Right-of use assets - finance leases | 94,087 | 94,008 | ||||||
| Other intangible assets, net | 189,931 | 182,466 | ||||||
| Goodwill | 61,392 | 52,920 | ||||||
| Other non-current assets | 29,133 | 27,586 | ||||||
| Non-current restricted cash | 525 | 525 | ||||||
| Total non-current assets | $ | 528,011 | $ | 494,785 | ||||
| Total assets | $ | 650,644 | $ | 649,141 | ||||
| LIABILITIES AND EQUITY | ||||||||
| CURRENT LIABILITIES: | ||||||||
| Accounts payable | $ | 21,611 | $ | 10,539 | ||||
| Accrued expenses and other current liabilities | 32,773 | 47,972 | ||||||
| Income tax liabilities - current | 9,170 | 6,614 | ||||||
| Debt, net - current portion (including related party principal amounts of $3,669 as of March 31, 2022) | 65,588 | 6,181 | ||||||
| Finance lease obligations - current | 13,197 | 12,620 | ||||||
| Total current liabilities | $ | 142,339 | $ | 83,926 | ||||
| NON-CURRENT LIABILITIES: | ||||||||
| Non-current debt, net (including related party principal amounts of $4,578 as of December 31, 2021) | $ | 77,384 | $ | 122,971 | ||||
| Finance lease obligations - non-current | 88,539 | 88,297 | ||||||
| Operating lease liabilities - non-current | 14,933 | 15,163 | ||||||
| Derivative liabilities | 68,975 | 92,435 | ||||||
| Income tax liabilities - non-current | 58,372 | 57,143 | ||||||
| Contingent consideration liabilities - non-current | 6,945 | 8,223 | ||||||
| Total non-current liabilities | $ | 315,148 | $ | 384,232 | ||||
| Total liabilities | $ | 457,487 | $ | 468,158 | ||||
| COMMITMENTS AND CONTINGENCIES (Note 20) | ||||||||
| EQUITY: | ||||||||
| Common stock (no par value; authorized shares - unlimited; issued shares - 189,728,625 and 182,707,359 Subordinate Voting Shares (including 2,757,290 and 2,859,151 unvested stock awards) as of March 31, 2022 and December 31, 2021, respectively | $ | — | $ | — | ||||
| Paid-in capital | 456,719 | 424,788 | ||||||
| Accumulated deficit | (262,175 | ) | (242,418 | ) | ||||
| Total Jushi shareholders' equity | $ | 194,544 | $ | 182,370 | ||||
| Non-controlling interests | (1,387 | ) | (1,387 | ) | ||||
| Total equity | $ | 193,157 | $ | 180,983 | ||||
| Total liabilities and equity | $ | 650,644 | $ | 649,141 | ||||
The accompanying notes are an integral part of these unaudited interim condensed consolidated financial statements.
| F-2 |
UNAUDITED INTERIM CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS AND
COMPREHENSIVE INCOME (LOSS)
(in thousands of U.S. dollars, except share and per share amounts)
| Three
Months Ended March 31, | ||||||||
| 2022 | 2021 | |||||||
| REVENUE, NET | $ | 61,888 | $ | 41,675 | ||||
| COST OF GOODS SOLD | (42,776 | ) | (22,934 | ) | ||||
| GROSS PROFIT | $ | 19,112 | $ | 18,741 | ||||
| OPERATING EXPENSES | $ | 37,308 | $ | 21,911 | ||||
| LOSS FROM OPERATIONS | $ | (18,196 | ) | $ | (3,170 | ) | ||
| OTHER (EXPENSE) INCOME: | ||||||||
| Interest expense, net | $ | (10,116 | ) | $ | (6,835 | ) | ||
| Fair value gains (losses) on derivatives | 14,309 | (9,358 | ) | |||||
| Other, net | (703 | ) | (3,376 | ) | ||||
| Total net other income (expense) | $ | 3,490 | $ | (19,569 | ) | |||
| INCOME (LOSS) BEFORE PROVISION FOR INCOME TAXES | $ | (14,706 | ) | $ | (22,739 | ) | ||
| Provision for income taxes | (5,051 | ) | (8,312 | ) | ||||
| NET INCOME (LOSS) AND COMPREHENSIVE INCOME (LOSS) | $ | (19,757 | ) | $ | (31,051 | ) | ||
| Net loss attributable to non-controlling interests | — | (175 | ) | |||||
| NET INCOME (LOSS) AND COMPREHENSIVE INCOME (LOSS) ATTRIBUTABLE TO JUSHI SHAREHOLDERS | $ | (19,757 | ) | $ | (30,876 | ) | ||
| NET INCOME (LOSS) AND COMPREHENSIVE INCOME (LOSS) PER SHARE ATTRIBUTABLE TO JUSHI SHAREHOLDERS - BASIC | $ | (0.11 | ) | $ | (0.20 | ) | ||
| Weighted average shares outstanding - basic | 183,226,027 | 157,176,375 | ||||||
NET (LOSS) AND COMPREHENSIVE (LOSS) PER SHARE ATTRIBUTABLE TO JUSHI SHAREHOLDERS - DILUTED | $ | (0.16 | ) | $ | (0.20 | ) | ||
| Weighted average shares outstanding - diluted | 207,838,906 | 157,176,375 | ||||||
The accompanying notes are an integral part of these unaudited interim condensed consolidated financial statements.
| F-3 |
UNAUDITED INTERIM CONDENSED CONSOLIDATED STATEMENTS OF CHANGES IN EQUITY
(in thousands of U.S. dollars, except share amounts)
| Number of Shares | ||||||||||||||||||||||||||||||||
| Super Voting Shares | Multiple Voting Shares | Subordinate Voting Shares | Paid-In Capital | Accumulated Deficit | Total Jushi Shareholders' Equity | Non-Controlling Interests | Total Equity | |||||||||||||||||||||||||
| Balances - January 1, 2022 | — | — | 182,707,359 | $ | 424,788 | $ | (242,418 | ) | $ | 182,370 | $ | (1,387 | ) | $ | 180,983 | |||||||||||||||||
| Private placement offerings | — | — | 3,717,392 | 13,680 | — | 13,680 | — | 13,680 | ||||||||||||||||||||||||
| Shares issued for Apothecarium acquisition | — | — | 527,704 | 1,594 | — | 1,594 | — | 1,594 | ||||||||||||||||||||||||
| Restricted stock grants and vesting, net of forfeitures (including related parties) | — | — | 5,952 | 832 | — | 832 | — | 832 | ||||||||||||||||||||||||
| Warrant expense, net of cancellations (including related parties) | — | — | — | 287 | — | 287 | — | 287 | ||||||||||||||||||||||||
| Stock option expense, net of forfeitures (including related parties) | — | — | — | 5,845 | — | 5,845 | — | 5,845 | ||||||||||||||||||||||||
| Shares issued upon exercise of warrants | — | — | 2,676,303 | 9,693 | — | 9,693 | — | 9,693 | ||||||||||||||||||||||||
| Shares issued upon exercise of stock options | — | — | 93,915 | — | — | — | — | — | ||||||||||||||||||||||||
| Net loss | — | — | — | — | (19,757 | ) | (19,757 | ) | — | (19,757 | ) | |||||||||||||||||||||
| Balances - March 31, 2022 | — | — | 189,728,625 | $ | 456,719 | $ | (262,175 | ) | $ | 194,544 | $ | (1,387 | ) | $ | 193,157 | |||||||||||||||||
| Number of Shares | ||||||||||||||||||||||||||||||||
| Super Voting Shares | Multiple Voting Shares | Subordinate Voting Shares | Paid-In Capital | Accumulated Deficit | Total Jushi Shareholders' Equity | Non-Controlling Interests | Total Equity | |||||||||||||||||||||||||
| Balances - January 1, 2021 | 149,000 | 4,000,000 | 132,396,064 | $ | 262,145 | $ | (262,669 | ) | $ | (524 | ) | $ | 2,947 | $ | 2,423 | |||||||||||||||||
| Public offerings | — | — | 13,685,000 | 85,660 | — | 85,660 | — | 85,660 | ||||||||||||||||||||||||
| Purchase of non-controlling interests | — | — | 500,000 | 1,562 | — | 1,562 | (1,562 | ) | — | |||||||||||||||||||||||
| Shares issued for Grover Beach acquisition | — | — | 49,348 | 368 | — | 368 | — | 368 | ||||||||||||||||||||||||
| Restricted stock grants and vesting, net of forfeitures (including related parties) | — | — | — | 2,538 | — | 2,538 | — | 2,538 | ||||||||||||||||||||||||
| Warrant expense, net of cancellations (including related parties) | — | — | — | 547 | — | 547 | — | 547 | ||||||||||||||||||||||||
| Stock option expense, net of forfeitures (including related parties) | — | — | — | 928 | — | 928 | — | 928 | ||||||||||||||||||||||||
| Shares issued upon exercise of warrants | — | — | 3,898,180 | 13,135 | — | 13,135 | — | 13,135 | ||||||||||||||||||||||||
| Shares issued upon exercise of stock options | — | — | 15,000 | 30 | — | 30 | — | 30 | ||||||||||||||||||||||||
| Net loss | — | — | — | — | (30,876 | ) | (30,876 | ) | (175 | ) | (31,051 | ) | ||||||||||||||||||||
| Balances - March 31, 2021 | 149,000 | 4,000,000 | 150,543,592 | $ | 366,913 | $ | (293,545 | ) | $ | 73,368 | $ | 1,210 | $ | 74,578 | ||||||||||||||||||
The accompanying notes are an integral part of these unaudited interim condensed consolidated financial statements.
| F-4 |
UNAUDITED INTERIM CONDENSED CONSOLIDATED STATEMENTS OF CASH FLOWS
(in thousands of U.S. dollars)
| Three
Months Ended March 31, | ||||||||
| 2022 | 2021 | |||||||
| CASH FLOWS FROM OPERATING ACTIVITIES: | ||||||||
| Net income (loss) | $ | (19,757 | ) | $ | (31,051 | ) | ||
| Adjustments to reconcile net income (loss) to net cash used in operating activities: | ||||||||
| Depreciation and amortization, including amounts in cost of goods sold | 3,248 | 1,579 | ||||||
| Share-based compensation | 6,964 | 4,013 | ||||||
| Fair value changes in derivatives | (14,309 | ) | 9,358 | |||||
| Non-cash interest expense | 4,551 | 3,905 | ||||||
| Deferred income taxes | (1,125 | ) | (280 | ) | ||||
| Loss on debt modification/extinguishment/redemption | — | 3,815 | ||||||
| (Gains) losses on investments and financial assets | — | (1,149 | ) | |||||
| Non-cash other (income) expense, net | 438 | (110 | ) | |||||
| Changes in operating assets and liabilities, net of acquisitions: | ||||||||
| Accounts receivable | 1,770 | (16 | ) | |||||
| Prepaid expenses and other current assets | 6,718 | 512 | ||||||
| Inventory | 5,917 | (5,680 | ) | |||||
| Accounts payable, accrued expenses and other current liabilities | 1,746 | 12,095 | ||||||
| Other assets | 1,173 | (112 | ) | |||||
| Net cash flows used in operating activities | $ | (2,666 | ) | $ | (3,121 | ) | ||
| CASH FLOWS FROM INVESTING ACTIVITIES: | ||||||||
| Payments for acquisitions, net of cash acquired | $ | (6,592 | ) | $ | (3,592 | ) | ||
| Payments for property, plant and equipment | (26,476 | ) | (8,566 | ) | ||||
| Proceeds from investments and financial assets | — | 3,252 | ||||||
| Net cash flows used in investing activities | $ | (33,068 | ) | $ | (8,906 | ) | ||
| CASH FLOWS FROM FINANCING ACTIVITIES: | ||||||||
| Issuance of shares for cash, net | $ | 13,680 | $ | 85,660 | ||||
| Proceeds from exercise of warrants and options | 541 | 9,886 | ||||||
| Redemptions of senior notes (including related party redemptions of $8 and $3,072 for three months ended March 31, 2022 and 2021, respectively) | (258 | ) | (8,134 | ) | ||||
| Payments on lease obligations | (601 | ) | (251 | ) | ||||
| Proceeds from other debt | 3,265 | 1,134 | ||||||
| Repayments of other debt | (130 | ) | — | |||||
| Net cash flows provided by financing activities | $ | 16,497 | $ | 88,295 | ||||
| Effect of currency translation on cash and cash equivalents | (9 | ) | (40 | ) | ||||
| NET CHANGE IN CASH AND CASH EQUIVALENTS AND RESTRICTED CASH | $ | (19,246 | ) | $ | 76,228 | |||
| CASH AND CASH EQUIVALENTS AND RESTRICTED CASH, BEGINNING OF PERIOD | 95,487 | 85,857 | ||||||
| CASH AND CASH EQUIVALENTS AND RESTRICTED CASH, END OF PERIOD | $ | 76,241 | $ | 162,085 | ||||
| F-5 |
| Three
Months Ended March 31, | ||||||||
| 2022 | 2021 | |||||||
| SUPPLEMENTAL CASH FLOW INFORMATION: | ||||||||
| Cash paid for interest (excluding capitalized interest) | $ | 5,580 | $ | 3,027 | ||||
| Cash paid for income taxes | $ | 3,867 | $ | 686 | ||||
| NON-CASH INVESTING AND FINANCING ACTIVITIES: | ||||||||
| Accrued capital expenditures | $ | 7,566 | $ | 3,531 | ||||
| Right of use assets from finance lease liabilities (excluding from acquisitions) | $ | — | $ | 928 | ||||
| Fair value of note obligations and warrant liabilities from acquisitions and acquisitions of non-controlling interests | $ | 6,922 | $ | — | ||||
| Fair value of shares issued for acquisitions and acquisitions of non-controlling interests | $ | 1,594 | $ | 1,930 | ||||
| Assets acquired and liabilities assumed in acquisitions: | ||||||||
| Cash and cash equivalents | $ | 25 | $ | — | ||||
| Other current assets | $ | 731 | $ | — | ||||
| Property, plant and equipment, including right-of-use assets | $ | 3,051 | $ | 2,319 | ||||
| Other non current assets | $ | 301 | $ | 19 | ||||
| Other intangible assets | $ | 8,200 | $ | 3,654 | ||||
| Goodwill | $ | 8,472 | $ | — | ||||
| Accounts payable and accrued liabilities | $ | (502 | ) | $ | — | |||
| Lease obligations | $ | (2,544 | ) | $ | (2,032 | ) | ||
| Deferred tax liabilities | $ | (2,601 | ) | $ | — | |||
The accompanying notes are an integral part of these unaudited interim condensed consolidated financial statements.
| F-6 |
|
Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| 1. | NATURE OF OPERATIONS |
Jushi Holdings Inc. (the “Company” or “Jushi”) is incorporated under the British Columbia’s Business Corporations Act (“BCBCA”). The Company is a vertically integrated, multi-state cannabis operator engaged in retail, distribution, cultivation, and processing operations in both medical and adult-use markets. As of March 31, 2022, Jushi, through its subsidiaries, owns or manages cannabis operations and/or holds licenses in the adult-use and/or medicinal cannabis marketplace in Illinois, Pennsylvania, Virginia, Massachusetts, Nevada, California and Ohio.
The Company is listed on the Canadian Securities Exchange (the “CSE”) and trades its subordinated voting shares (“SVS”) under the ticker symbol “JUSH", and trades on the U.S. Over the Counter Stock Market (“OTCQX”) under the symbol JUSHF.
The Company’s head office is located at 301 Yamato Road, Suite 3250, Boca Raton, Florida 33431, U.S.A., and its registered address is Suite 1700, Park Place, 666 Burrard Street, Vancouver, British Columbia V6C 2X8, Canada.
| 2. | BASIS OF PRESENTATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES |
Basis of Presentation and Consolidation
The accompanying unaudited interim condensed consolidated financial statements present the consolidated financial position and operations of Jushi Holdings Inc. and its subsidiaries and entities over which the Company has control, in accordance with generally accepted accounting principles in the U.S. (“GAAP”) for interim financial information and in accordance with the rules and regulations of the U.S. Securities and Exchange Commission (“SEC”). Accordingly, they do not include all of the information and footnotes required by GAAP for complete financial statements. The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the amounts reported in the financial statements and footnotes. Actual results could differ materially from those estimates.
In the opinion of management, the unaudited condensed interim consolidated financial statements include all adjustments, of a normal recurring nature, that are necessary to present fairly the financial position, results of operations and cash flows of the Company for the periods, and at the dates, presented. The results for interim periods are not necessarily indicative of results that may be expected for any other interim period or for the full year.
These unaudited interim condensed consolidated financial statements should be read in conjunction with the Company’s audited consolidated financial statements and notes thereto for the year ended December 31, 2021 included elsewhere in this filing. Consolidated balance sheet information as of December 31, 2021 presented herein are derived from the Company’s audited consolidated financial statements for the year ended December 31, 2021.
These unaudited interim condensed consolidated financial statements have been prepared assuming the Company will continue as a going concern. GAAP requires an entity to look forward 12 months from the date of the financial statements (the “look-forward” period) when assessing whether the going concern assumption can be used. The going concern assumption contemplates the realization of assets and satisfaction of liabilities in the normal course of business. However, substantial doubt about the Company’s ability to continue as a going concern exists.
As reflected in these unaudited interim condensed consolidated financial statements, the Company has incurred losses from operations for the three months ended March 31, 2022, has an accumulated deficit of $262,175 as of March 31, 2022, and cash and cash equivalents of $75,717 as of March 31, 2022. As discussed in Note 9 - Debt, the Company’s 10% senior notes (the “Senior Notes”), which as of March 31, 2022 had an aggregate principal amount outstanding of $74,935, mature on January 15, 2023, and the Acquisition Facility (refer to Note 9 - Debt) required the Company to maintain certain covenants which the Company may not have been in compliance with if the court accepted Jushi Europe’s petition for bankruptcy. The Company was also projected to violate certain financial covenants further within the next twelve months. The Company has been working with various lenders to refinance the Senior Notes at terms most favorable to the Company. Given the changing conditions in the debt capital markets, refinancing term sheets are still being negotiated. These conditions raised substantial doubt regarding the Company’s ability to continue as a going concern.
In April 2022, the Company entered into an amendment with the lender of the Acquisition Facility, which included a waiver related to Jushi Europe’s bankruptcy and a change to the terms of the Total Leverage ratio, as defined in the Acquisition Facility agreement, and deferred the commencement date of leverage testing under the Acquisition Facility. The Company is pursuing strategies to obtain the required additional funding primarily to fund the Senior Notes and future operations. These strategies may include, but are not limited to: (i) ongoing efforts with certain lenders to refinance the Senior Notes; (ii) deferral of certain expenditures, including capital projects, and reallocate funds for debt repayment, if the need arose; (iii) alternative sources of debt and equity financing, including secured borrowings and through a base shelf prospectus, which allows the Company to offer up to C$500,000 in securities in Canada through the end of 2023. However, there can be no assurance that the Company will be able to refinance the Senior Notes, generate positive results from operations, or obtain additional liquidity when needed or under acceptable terms, if at all.
| F-7 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Segments
The Company operates a vertically integrated cannabis business in one reportable segment: the cultivation, manufacturing, distribution and sale of cannabis in the U.S. All revenues for the three months ended March 31, 2022 and 2021 were generated within the U.S., and substantially all long-lived assets are located within the U.S.
COVID-19
During the three-months ended March 31, 2022, the Company’s financial condition and results of operations was not materially impacted by COVID-19. The extent to which the COVID-19 pandemic impacts the Company’s future results will depend on future developments, which are highly uncertain and cannot be predicted with certainty, including possible future outbreaks of new strains of the virus and governmental and consumer responses to such future developments.
Emerging Growth Company
As an emerging growth company (“EGC”), the Jumpstart Our Business Startups Act (“JOBS Act”) allows the Company to delay adoption of new or revised accounting pronouncements applicable to public companies until such pronouncements are applicable to private companies. The Company has elected to use this extended transition period under the JOBS Act until such time the Company is no longer considered to be an EGC. The adoption dates discussed below reflect this election.
Recent Accounting Pronouncements
In June 2020, the FASB issued ASU 2020-06 Debt-Debt with Conversion and Other Options (Subtopic 470-20) and Derivatives and Hedging-Contracts in Entity’s Own Equity (Subtopic 815-40): Accounting for Convertible Instruments and Contracts in an Entity’s Own Equity, which simplifies accounting for convertible instruments by removing major separation models required under current GAAP. This ASU also removes certain settlement conditions that are required for equity contracts to qualify for the derivative scope exception and simplifies the diluted earnings per share calculation in certain areas. The amendments in this ASU are effective for annual and interim periods beginning after December 15, 2023, although early adoption is permitted. The Company is in the process of evaluating the impact of this new guidance on its consolidated financial statements.
| F-8 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
In May 2021, the FASB issued ASU 2021-04, “Earnings Per Share (Topic 260), Debt—Modifications and Extinguishments (Subtopic 470-50), Compensation—Stock Compensation (Topic 718), and Derivatives and Hedging—Contracts in Entity’s Own Equity (Subtopic 815-40): Issuer’s Accounting for Certain Modifications or Exchanges of Freestanding Equity-Classified Written Call Options”— The FASB issued guidance to clarify and reduce diversity in an issuer’s accounting for modifications or exchanges of freestanding equity-classified written call options (for example, warrants) that remain equity classified after modification or exchange. The amendments in this ASU are effective for annual and interim periods beginning after December 15, 2021. The adoption of the standard did not have a material impact on the Company’s consolidated financial statements.
In October 2021, the FASB issued ASU 2021-08, Business Combinations (Topic 805): Accounting for Contract Assets and Contract Liabilities from Contracts with Customers. The FASB issued guidance which require that an entity (acquirer) recognize and measure contract assets and contract liabilities acquired in a business combination in accordance with Topic 606. At the acquisition date, an acquirer should account for the related revenue contracts in accordance with Topic 606 as if it had originated the contracts. To achieve this, an acquirer may assess how the acquiree applied Topic 606 to determine what to record for the acquired revenue contracts. Generally, this should result in an acquirer recognizing and measuring the acquired contract assets and contract liabilities consistent with how they were recognized and measured in the acquiree’s financial statements (if the acquiree prepared financial statements in accordance with generally accepted accounting principles). The amendments in this ASU are effective for annual and interim periods beginning after December 15, 2023, although early adoption is permitted. The Company is in the process of evaluating the impact of this new guidance on its consolidated financial statements.
In June 2022, the FASB issued ASU 2022-03, Fair Value Measurement (Topic 820): Fair Value Measurement of Equity Securities Subject to Contractual Sale Restrictions. The FASB issued guidance clarifies that a contractual restriction on the sale of an equity security is not considered part of the unit of account of the equity security and, therefore, is not considered in measuring fair value. The amendments also clarify that an entity cannot, as a separate unit of account, recognize and measure a contractual sale restriction. The amendments in this ASU are effective for annual and interim periods beginning after December 15, 2023, although early adoption is permitted. The Company is in the process of evaluating the impact of this new guidance on its consolidated financial statements.
| 3. | REVENUE |
The Company has three revenue streams: (i) cannabis retail (ii) cannabis wholesale; and (iii) other. The retail revenues are comprised of cannabis operations for medical and adult-use dispensaries. The Company’s wholesale revenues are comprised of cannabis cultivation, processing, production and distribution of cannabis for medical and adult-use. The Company’s other operations primarily include the Company’s hemp/cannabidiol (“CBD”) retail operations. Any intercompany revenue and any costs between entities are eliminated to arrive at consolidated totals.
The following table summarizes revenue disaggregated by revenue stream:
| Three Months Ended March 31, | ||||||||||||||||||||||||
| 2022 | 2021 | |||||||||||||||||||||||
| Gross Revenue | Intercompany Revenue | Revenue to External Customers | Gross Revenue | Intercompany Revenue | Revenue to External Customers | |||||||||||||||||||
| Retail cannabis | $ | 57,955 | $ | — | $ | 57,955 | $ | 39,277 | $ | — | $ | 39,277 | ||||||||||||
| Wholesale cannabis | 9,443 | (5,595 | ) | 3,848 | 4,192 | (1,883 | ) | 2,309 | ||||||||||||||||
| Other | 85 | — | 85 | 89 | — | 89 | ||||||||||||||||||
| Eliminations | (5,595 | ) | 5,595 | — | (1,883 | ) | 1,883 | — | ||||||||||||||||
| Consolidated revenue | $ | 61,888 | $ | — | $ | 61,888 | $ | 41,675 | $ | — | $ | 41,675 | ||||||||||||
| F-9 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| 4. | INVENTORIES |
The components of inventories, net, are as follows:
March 31, 2022 (unaudited) |
December 31, 2021 | |||||||
| Cannabis plants | $ | 3,101 | $ | 6,347 | ||||
| Harvested cannabis and packaging | 9,180 | 5,180 | ||||||
| Total raw materials | $ | 12,281 | $ | 11,527 | ||||
| Work in process | 6,136 | 8,756 | ||||||
| Finished goods | 20,865 | 23,036 | ||||||
| Total inventories, net | $ | 39,282 | $ | 43,319 | ||||
| 5. | PREPAID EXPENSES AND OTHER CURRENT ASSETS |
The components of prepaid expenses and other current assets are as follows:
March 31, 2022 (unaudited) | December 31, 2021 | |||||||
| Prepaid expenses and deposits | $ | 3,844 | $ | 3,837 | ||||
| Landlord receivables for leasehold improvements | 394 | 7,357 | ||||||
| Employee receivable | 46 | 248 | ||||||
| Other current assets | 1,890 | 1,433 | ||||||
| Total prepaid expenses and other current assets | $ | 6,174 | $ | 12,875 | ||||
| 6. | PROPERTY, PLANT AND EQUIPMENT |
The components of property, plant and equipment (PP&E) are as follows:
March 31, 2022 (unaudited) | December 31, 2021 | |||||||
| Buildings and building components | $ | 51,020 | $ | 49,697 | ||||
| Land | 12,380 | 12,380 | ||||||
| Leasehold improvements | 23,323 | 24,042 | ||||||
| Machinery and equipment | 12,743 | 12,656 | ||||||
| Computer equipment | 2,281 | 2,221 | ||||||
| Furniture and fixtures | 8,518 | 8,000 | ||||||
| Construction-in-process | 52,521 | 35,625 | ||||||
| Total property, plant and equipment - gross | $ | 162,786 | $ | 144,621 | ||||
| Less: Accumulated depreciation | (9,843 | ) | (7,341 | ) | ||||
| Total property, plant and equipment - net | $ | 152,943 | $ | 137,280 | ||||
Construction-in-process (“CIP”) represents assets under construction for manufacturing and retail build-outs not yet ready for use.
For the three months ended March 31, 2022 and 2021, total depreciation, including depreciation from assets held under finance leases (which are reflected separately in the consolidated balance sheets), was $3,693 and $1,169, respectively, of which $2,168 and $604, respectively, was absorbed into inventory production. Interest expense capitalized to PP&E totaled $744 and $85 for the three months ended March 31, 2022 and 2021, respectively.
| F-10 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| 7. | ACQUISITIONS |
2022 Business Combination
In March 2022, the Company closed on the acquisition of 100% of the equity interest of an entity operating an adult-use and medical retail dispensary under the name, The Apothecarium in Las Vegas, Nevada (“Apothecarium”). The Apothecarium acquisition, together with the prior acquisition of Franklin Bioscience NV, LLC, a holder of medical and adult-use cannabis cultivation, processing, and distribution licenses, enables the Company to become vertically integrated in Nevada, as well as provide significant branding exposure for Jushi’s high-quality product lines.
The following table summarizes the preliminary purchase price allocation for Apothecarium as of the acquisition date:
| Cash and cash equivalents | $ | 25 | ||
| Prepaids and other assets | 32 | |||
| Inventory | 699 | |||
| Property, plant and equipment | 498 | |||
| Right-of-use assets | 2,553 | |||
| Intangible asset - licenses (1) | 8,200 | |||
| Deposits | 301 | |||
| Total assets | $ | 12,308 | ||
| Accounts payable and accrued liabilities | $ | (502 | ) | |
| Lease liabilities | (2,544 | ) | ||
| Deferred tax liabilities | (2,601 | ) | ||
| Total liabilities | $ | (5,647 | ) | |
| Net assets acquired | $ | 6,661 | ||
| Goodwill | 8,472 | |||
| Total | $ | 15,133 | ||
| Consideration paid in cash, as adjusted for working capital adjustments | $ | 6,617 | ||
| Consideration paid in promissory notes (fair value) (2) | 6,922 | |||
| Consideration paid in shares (3) | 1,594 | |||
| Fair value of consideration | $ | 15,133 |
| (1) | The licenses acquired have indefinite useful lives. | |
| (2) | The Company issued a $9,853 promissory note. Refer to “Acquisition-Related Promissory Notes” in Note 9 - Debt for details on the seller notes. | |
| (3) | The Company issued 527,704 Subordinate Voting Shares (“SVS”) with a grant date fair value of $3.02 each. |
In addition, the Company may pay up to $2,000 in potential earn-out consideration based on the achievement of certain financial metrics. As of the date of acquisition, the Company has not recognized a contingent consideration liability for this acquisition as the probability is unlikely. The estimated range of such potential additional consideration is between $0 and $2,000.
| F-11 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
The estimated fair value of the licenses was determined using the multi-period excess earnings method under the income approach based on projections extended to 2036 assuming a 5.3% revenue growth rate in 2023 and a 3% long-term revenue growth rate thereafter. The goodwill recognized from the acquisition is attributable to synergies expected from integrating Apothecarium into the Company’s existing business. The goodwill acquired is not deductible for tax purposes.
Preliminary Purchase Price Allocations for Business Combinations
The consideration has been allocated to the estimated fair values of the assets acquired and liabilities assumed at the dates of the acquisitions and remain preliminary as of March 31, 2022. These estimated fair values involve significant judgement and estimates. The primary area of judgement involves the valuation of the retail licenses acquired, which requires management to estimate value based on future cash flows from these assets. The primary areas of the preliminary purchase price allocations that are not yet finalized relate to: licenses acquired, inventories, leases, contingent consideration, promissory notes, deferred tax liabilities, and residual goodwill. The Company expects to continue to obtain information to assist in determining the fair value of the net assets acquired at the acquisition date during the measurement period.
Business Combinations - Acquisition and Deal Costs
For the three months ended March 31, 2022, acquisition and deal costs relating to Apothecarium totaled $34 and are included within operating expenses in the consolidated statements of operations and comprehensive loss. The remaining acquisition and deal costs included in operating expenses were incurred either for acquisitions not completed or not expected to be completed.
2021 Business Combinations and Asset Acquisitions
The Company had the following acquisitions during the year ended December 31, 2021: (i) Nature’s Remedy; (ii) OSD; (iii) OhiGrow; and (iv) Grover Beach (all defined below).
| F-12 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
The following table summarizes the purchase price allocations as of their respective acquisition dates:
| Business Combinations | Asset Acquisitions | |||||||||||||||||||
| Nature’s Remedy | OSD | OhiGrow | Grover Beach | Total | ||||||||||||||||
| Assets Acquired: | ||||||||||||||||||||
| Cash and cash equivalents | $ | 3,195 | $ | 259 | $ | — | $ | — | $ | 3,454 | ||||||||||
| Prepaids | 325 | 53 | — | — | 378 | |||||||||||||||
| Accounts receivable, net | 263 | — | — | — | 263 | |||||||||||||||
| Inventory | 15,882 | 184 | — | — | 16,066 | |||||||||||||||
| Indemnification assets (1) | 1,322 | 1,411 | — | — | 2,733 | |||||||||||||||
| Property, plant and equipment | 19,470 | — | 3,165 | 269 | 22,904 | |||||||||||||||
| Right-of-use assets - finance leases | 27,305 | — | — | 2,050 | 29,355 | |||||||||||||||
| Right-of-use assets - operating leases | 1,337 | 1,859 | — | — | 3,196 | |||||||||||||||
| Intangible assets - license (2) | 46,000 | 2,160 | 1,817 | 3,654 | 53,631 | |||||||||||||||
| Intangible assets - tradenames (2) | 4,400 | — | — | — | 4,400 | |||||||||||||||
| Intangible assets - customer database (2) | 2,100 | — | — | — | 2,100 | |||||||||||||||
| Deposits | 20 | 6 | — | 19 | 45 | |||||||||||||||
| Total assets acquired | $ | 121,619 | $ | 5,932 | $ | 4,982 | $ | 5,992 | $ | 138,525 | ||||||||||
| Liabilities Assumed: | ||||||||||||||||||||
| Accounts payable and accrued liabilities | $ | 7,004 | $ | 1,601 | $ | — | $ | — | $ | 8,605 | ||||||||||
| Finance lease obligations | 27,052 | — | — | 2,032 | $ | 29,084 | ||||||||||||||
| Operating lease obligations | 1,267 | 1,859 | — | — | $ | 3,126 | ||||||||||||||
| Deferred tax liabilities | 19,876 | 648 | — | — | $ | 20,524 | ||||||||||||||
| Total liabilities | $ | 55,199 | $ | 4,108 | $ | — | $ | 2,032 | $ | 61,339 | ||||||||||
| Net assets acquired | $ | 66,420 | $ | 1,824 | $ | 4,982 | $ | 3,960 | $ | 77,186 | ||||||||||
| Goodwill | 33,178 | 2,432 | — | — | 35,610 | |||||||||||||||
| Total | $ | 99,598 | $ | 4,256 | $ | 4,982 | $ | 3,960 | $ | 112,796 | ||||||||||
| Consideration: | ||||||||||||||||||||
| Consideration paid in cash, as adjusted for working capital adjustments | $ | 40,360 | $ | 1,827 | $ | 4,949 | $ | 3,592 | $ | 50,728 | ||||||||||
| Consideration paid in promissory notes (fair value) | 15,345 | 2,429 | — | — | $ | 17,774 | ||||||||||||||
| Consideration paid in shares | 35,670 | — | — | 368 | $ | 36,038 | ||||||||||||||
| Contingent consideration | 8,223 | — | — | $ | — | $ | 8,223 | |||||||||||||
| Capitalized costs | — | — | 33 | $ | — | $ | 33 | |||||||||||||
| Fair value of consideration | $ | 99,598 | $ | 4,256 | $ | 4,982 | $ | 3,960 | $ | 112,796 | ||||||||||
| (1) | As part of the OSD and Nature’s Remedy acquisition agreements, the sellers contractually agreed to indemnify the Company for certain amounts that may become payable, including for taxes that relate to periods prior to the date of acquisition. Accordingly, the Company recorded indemnification assets and corresponding estimated accrued tax liabilities, at fair value, for a total of $2,733 as of the dates of the acquisitions. The range of total estimated potential indemnification assets is from $0 to $6,322; however, there is no limit on the Nature’s Remedy indemnification asset. Additional subsequent changes in the amounts recognized for the indemnification assets may occur in relation to the provision for the corresponding tax liabilities, according to changes in the range of outcomes or the assumptions used to develop the estimates of the liabilities at the time of the acquisition. |
| (2) | The licenses acquired have indefinite useful lives. The customer relationships have a useful life of 15 years and the tradenames have a useful life of 5 years. |
| F-13 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
2021 Business Combinations
Nature’s Remedy
On September 10, 2021, the Company acquired 100% of the equity of Nature’s Remedy of Massachusetts, Inc. and certain of its affiliates (collectively, “Nature’s Remedy”), for upfront consideration comprised of cash, net of working capital adjustments, 8,700,000 subordinate voting shares (with a grant date fair value of $4.10 each), an $11,500 unsecured three-year note and a $5,000 unsecured five-year note.
Nature’s Remedy is a vertically integrated single state operator in Massachusetts and currently operates two retail dispensaries, in Millbury, MA and Tyngsborough, MA, and a 50,000 sq. ft. cultivation and production facility in Lakeville, MA. The goodwill is not tax deductible.
The Company also agreed to issue a $5,000 increase to the principal balance of the three-year note and up to an additional $5,000 in Company SVS upon the occurrence or non-occurrence of certain events after the closing date. The payment of the contingent consideration depends on whether or not a competitor opens a competing dispensary within a certain radius of the Company’s dispensary in the Town of Tyngsborough, MA during the first 12 months following the acquisition (The “First Milestone Period”) or during the 18 months following the end of the First Milestone Period. As of the date of acquisition, the Company recognized a contingent consideration liability of $8,223, a Level 3 measurement amount, which was based on the weighted-average probability of the potential outcomes. The estimated range of such additional consideration is between $0 and $10,800 (which also includes the interest on the additional principal for the three-year note). Contingent consideration is measured at its acquisition-date fair value and included as part of the consideration transferred for the business combination. Contingent consideration that is classified as a liability is remeasured at subsequent reporting dates with the corresponding gain or loss being recognized in Other, net in the consolidated statements of operations and comprehensive income (loss).
During the three months ended March 31, 2022, the Company remeasured the contingent consideration and recognized a loss of $405 primarily related to accretion. The remeasurement increased the total contingent consideration liability from $8,223 on December 31, 2021, to $8,628 on March 31, 2022. The Company utilized the cash flows associated with the weighted-average probability of the potential outcomes to determine the potential cash outflows that are short-term vs. long-term. As a result, the Company classified $1,683 as a short-term contingent liability and $6,945 as a long-term contingent liability as of March 31, 2022.
OSD
On April 30, 2021, the Company acquired 100% of the equity of Organic Solutions of the Desert, LLC (“OSD”), an operating dispensary located in Palm Springs, California, for consideration comprised of cash, as adjusted for working capital adjustments, and $3,100 principal amount of promissory notes. Refer to “Promissory Notes Payable” in Note 9 - Debt for details on the seller notes. The goodwill is not tax deductible.
Preliminary Purchase Price Allocations for Business Combinations
The consideration for Nature’s Remedy has been allocated to the estimated fair values of the assets acquired and liabilities assumed at the dates of the acquisitions and remain preliminary as of March 31, 2022. These estimated fair values involve significant judgement and estimates. The primary areas of judgement involved are the valuation of the intangible assets acquired, which requires management to estimate value based on future cash flows from these assets. The primary areas of the preliminary purchase price allocations that are not yet finalized relate to: intangible assets acquired, property, plant and equipment, indemnification assets, contingent consideration, deferred tax liabilities, and residual goodwill. The Company expects to continue to obtain information to assist in determining the fair value of the net assets acquired as of the respective acquisition dates during the measurement period.
| F-14 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
2021 Asset Acquisitions
The Company determined that the OhiGrow and Grover Beach acquisitions described below did not qualify as business combinations because, for OhiGrow, the assets acquired did not constitute a business, and for Grover Beach, under the concentration test, substantially all of the fair value of the acquisition is concentrated in a single identifiable asset – the license.
OhiGrow
In July 2021, the Company acquired OhiGrow, LLC, a licensed cultivator in Ohio, and Ohio Green Grow LLC (collectively, “OhiGrow”), inclusive of an approximately 10,000 sq. ft. facility and 1.35 acres of land for $4,949 in cash.
Grover Beach
On March 4, 2021, the Company closed on the acquisition of approximately 78% of the equity of a retail license holder located in Grover Beach, California (“Grover Beach”) for $3,592 in cash, as adjusted for working capital adjustments, and 49,348 SVS at a fair value of $7.46 per share, with the rights to acquire the remaining equity for $1 in the future.
Business Combinations Acquisition Results and Unaudited Supplemental Pro Forma Financial Information
The following table summarizes consolidated proforma revenue and consolidated proforma net income (loss) as if the business combinations had occurred at the beginning of the year prior to their actual acquisition for the periods presented:
| Three months ended March 31, | ||||||||
| 2022 | 2021 | |||||||
| Revenue | $ | 64,903 | $ | 55,962 | ||||
| Net income (loss) | $ | (19,560 | ) | $ | (31,463 | ) | ||
These unaudited pro forma financial results do not purport to be indicative of the actual results that would have been achieved by the combined companies for the years indicated, or of the results that may be achieved by the combined companies in the future. These amounts have been calculated using actual results and adding pre-acquisition results, after adjusting for: acquisition costs, additional depreciation and amortization from acquired property, plant and equipment and intangible assets, as well as adjustments for incremental interest expense relating to consideration paid, and changes to conform to the Company’s accounting policies.
For the three months ended March 31, 2022, Apothecarium contributed revenues of $556 and net income of $55 to the Company’s consolidated results.
| F-15 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| 8. ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES |
The components of accrued expenses and other current liabilities are as follows:
| March 31,
2022 (unaudited) | December 31, 2021 | |||||||
| Accrued capital expenditures | $ | 7,566 | $ | 17,599 | ||||
| Goods received not invoiced | 6,501 | 8,007 | ||||||
| Accrued employee related expenses and liabilities | 5,838 | 6,062 | ||||||
| Accrued professional and management fees | 2,007 | 5,139 | ||||||
| Accrued sales and excise taxes | 1,189 | 2,535 | ||||||
| Accrued interest | 1,470 | 1,181 | ||||||
| Deferred revenue (loyalty program) | 1,693 | 1,427 | ||||||
| Operating lease obligations – current portion | 2,705 | 2,745 | ||||||
| Contingent consideration liabilities – current portion(1) | 1,683 | — | ||||||
| Other accrued expenses and current liabilities | 2,121 | 3,277 | ||||||
| Total | $ | 32,773 | $ | 47,972 | ||||
| (1) | Refer to Note 7 - Acquisitions. |
| 9. DEBT |
The components of the Company’s debt are as follows:
| Effective Interest Rate | Maturity Date | March 31, 2022 (unaudited) | December 31, 2021 | |||||||||||
| Principal amounts: | ||||||||||||||
| Senior Notes | 34% | January 2023 | $ | 74,935 | $ | 75,193 | ||||||||
| Acquisition Facility | 14% | October 2026 | 40,000 | 40,000 | ||||||||||
| Acquisition-related promissory notes payable | 9% - 25% | November 2022 - April,2027 | 35,614 | 25,767 | ||||||||||
| Other debt (1) | 6% - 12% | March 2022 - July 2050 | 14,965 | 11,728 | ||||||||||
| Total debt - principal amounts | $ | 165,514 | $ | 152,688 | ||||||||||
| Less: debt issuance costs and original issue discounts | (22,542 | ) | (23,536 | ) | ||||||||||
| Total debt - carrying amounts | $ | 142,972 | $ | 129,152 | ||||||||||
| Debt - current portion | $ | 65,588 | $ | 6,181 | ||||||||||
| Debt - non-current portion | $ | 77,384 | $ | 122,971 | ||||||||||
(1) Includes Jushi Europe debt. Refer to Note 15 - Non-Controlling Interests.
| F-16 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
As of March 31, 2022, aggregate future contractual maturities of the Company’s debt are as follows:
| Remainder of the year | 2023 | 2024 | 2025 | 2026 | Thereafter | Total | ||||||||||||||||||||||
| Senior Notes | $ | — | $ | 74,935 | $ | — | $ | — | $ | — | $ | — | $ | 74,935 | ||||||||||||||
| Acquisition Facility | — | — | 4,000 | 4,000 | 32,000 | — | 40,000 | |||||||||||||||||||||
| Acquisition-related promissory notes payable (1) | 2,412 | 3,941 | 17,385 | 1,970 | 6,806 | 3,100 | 35,614 | |||||||||||||||||||||
| Other debt | 3,718 | 149 | 139 | 109 | 4,579 | 6,271 | 14,965 | |||||||||||||||||||||
| Total | $ | 6,130 | $ | 79,025 | $ | 21,524 | $ | 6,079 | $ | 43,385 | $ | 9,371 | $ | 165,514 | ||||||||||||||
(1) The Promissory Note that matures in 2022 is a mandatorily convertible note that will be settled in 910,000 shares.
Interest expense, net is comprised of the following:
| Three Months Ended March 31, | ||||||||
| 2022 | 2021 | |||||||
| Interest and accretion - Senior Notes | 5,398 | 5,204 | ||||||
| Interest - Finance lease liabilities | 2,901 | 1,389 | ||||||
| Interest and accretion - Promissory notes | 737 | 311 | ||||||
| Interest and accretion - Acquisition Facility | 1,360 | — | ||||||
| Interest and accretion - Other debt | 479 | 113 | ||||||
| Capitalized interest | (744 | ) | (85 | ) | ||||
| Total interest expense | 10,131 | 6,932 | ||||||
| Interest income | $ | (15 | ) | $ | (97 | ) | ||
| Total interest expense, net | 10,116 | 6,835 | ||||||
Other Debt - PAMS Sale-leaseback Transactions
During 2021, the Company acquired land and buildings that are adjacent to the Company’s Pennsylvania Medical Solutions, LLC (“PAMS”) cultivation facility in order to expand the facility. In February 2022, the Company then entered into a sale-leaseback agreement. The Company concluded that control, including the significant risks and rewards of ownership, did not transfer to the buyer-lessor at the inception of the sale-leaseback transaction. As a result, the transaction did not meet the accounting criteria for a successful sale-leaseback transaction and therefore represents a financing obligation with a maturity date of April 2048. The Company recognized a liability of $3,265 and will accrue interest using an incremental borrowing rate of 11.6%. Annual payments are $88 and subject to certain escalation.
Unsecured Promissory Notes – Apothecarium
In March 2022, in connection with the Apothecarium acquisition, the Company issued to the seller two unsecured promissory notes with a total principal amount of $9,853, with no stated interest and both maturing on March 16, 2027. The promissory notes provide for a principal payment of $3,448 on the 21st month anniversary, followed by 39 equal monthly payments for the remaining balance.
Amendments to the Acquisition Facility
In April 2022, the Company entered into an amendment to the Acquisition Facility pursuant to which: (i) the commencement of leverage testing was pushed back by four quarters (now beginning March 31, 2023 as reflected in the table below), (ii) certain leverage ratios were revised; and (iii) the Company may proceed with a reorganization pursuant to a petition for bankruptcy in Switzerland with respect to Jushi Europe without defaulting under the Acquisition Facility. Refer to Note 15 - Non-Controlling Interests for additional information on Jushi Europe.
Total Leverage Ratio, calculated as the ratio of Total Funded Indebtedness to EBITDA (all such terms are defined in the Acquisition Facility agreement) not to exceed the correlative ratio below:
| Applicable Ratio | Fiscal Quarter Ending | |
| 6.00 to 1.00 | March 31, 2023 | |
| 5.00 to 1.00 | June 30, 2023 | |
| 4.00 to 1.00 | September 30 and December 31, 2023 | |
| 3.50 to 1.00 | March 31, 2024 and all fiscal quarters ending thereafter |
| F-17 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| 10. LEASE OBLIGATIONS |
The Company leases certain business facilities for corporate, retail and cultivation operations from third parties under lease agreements that specify minimum rentals. In addition, the Company leases certain equipment for use in cultivation and extraction activities. The Company determines whether a contract is or contains a lease at the inception of the contract. The expiry dates of the leases, including reasonably certain estimated renewal periods, are between 2022 and 2050. The Company’s lease agreements do not contain any material residual value guarantees or material restrictive covenants.
The following table provides the components of lease cost recognized in the consolidated statements of operations and comprehensive income (loss) for the periods presented:
| Three Months Ended March 31, | ||||||||
| 2022 | 2021 | |||||||
| Operating lease cost | $ | 886 | $ | 457 | ||||
| Finance lease cost: | ||||||||
| Amortization of lease assets | 1,143 | 366 | ||||||
| Interest on lease liabilities | 2,901 | 1,389 | ||||||
| Total finance lease cost | $ | 4,044 | $ | 1,755 | ||||
| Variable lease cost | 93 | $ | 75 | |||||
| Total lease cost | $ | 5,023 | $ | 2,287 | ||||
Other information related to operating and finance leases as of the balance sheet dates presented are as follows:
| March 31,
2022 (unaudited) | December 31, 2021 | |||||||||||||||
| Finance Leases | Operating Leases | Finance Leases | Operating Leases | |||||||||||||
| Weighted average discount rate | 11.73 | % | 11.50 | % | 11.75 | % | 11.50 | % | ||||||||
| Weighted average remaining lease term (in years) | 22.2 | 14.4 | 22.6 | 14.6 | ||||||||||||
| F-18 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
The maturities of the contractual undiscounted lease liabilities as of March 31, 2022 are as follows:
| Finance Leases | Operating Leases | |||||||
| Remainder of the year | $ | 11,711 | $ | 2,150 | ||||
| 2023 | 11,031 | 2,974 | ||||||
| 2024 | 11,153 | 2,709 | ||||||
| 2025 | 11,773 | 2,522 | ||||||
| 2026 | 11,436 | 2,292 | ||||||
| Thereafter | 251,898 | 27,866 | ||||||
| 309,002 | 40,513 | |||||||
| Interest on lease liabilities | (207,266 | ) | (22,875 | ) | ||||
| Total present value of minimum lease payments | $ | 101,736 | $ | 17,638 | ||||
| Lease liabilities – current portion | $ | 13,197 | $ | 2,705 | ||||
| Lease liabilities – non-current | $ | 88,539 | $ | 14,933 | ||||
| 11. DERIVATIVE LIABILITIES |
The continuities of the Company’s derivative liabilities are as follows:
| Total
Derivative Liabilities (1)(3) | ||||
| Carrying amounts as of January 1, 2022 | $ | 92,435 | ||
| Fair value changes (2) | (14,309 | ) | ||
| Derivative Warrants exercises | (9,151 | ) | ||
| Carrying amounts as of March 31, 2022 | $ | 68,975 | ||
| (1) | Refer to Note 12 - Equity for the continuity of the number of these warrants outstanding. | |
| (2) | Included in other income (expense), net in the consolidated statements of operations and comprehensive income (loss). | |
| (3) | Includes mandatory prepayment option on the Senior Notes, which had a fair value of $174 as of March 31, 2022. |
The Company’s derivative liabilities are primarily comprised of derivative warrants. These are warrants to purchase SVS of the Company which were issued in connection with the Senior Notes (the “Derivative Warrants”) have an expiration date of December 23, 2024 and an exercise price of US$1.25. There were 36,674,355 and 40,124,355 Derivative Warrants outstanding as of March 31, 2022 and December 31, 2021, respectively. The Derivative Warrants may be net share settled.
| F-19 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
These warrants are considered derivative financial liabilities measured at fair value with all gains or losses recognized in profit or loss as the settlement amount for the warrants may be adjusted during certain periods for variables that are not inputs to standard pricing models for forward or option equity contracts, i.e., the "fixed for fixed" criteria under ASC 815-40. The estimated fair value of the Derivative Warrants is measured at each reporting period and an adjustment is reflected in fair value changes in derivatives in the consolidated statements of operations and comprehensive income (loss). These are Level 3 recurring fair value measurements. The estimated fair value of the Derivative Warrants as of March 31, 2022 and December 31, 2021 was determined using the Black-Scholes model and Monte Carlo simulation model. The assumptions used in the fair value calculations as of the balance sheet dates presented include the following:
| March 31, 2022 (unaudited) | December 31, 2021 | |||
| Stock price | $2.85 | $3.25 | ||
| Risk-free annual interest rate | 2.40% | 0.97% | ||
| Range of estimated possible exercise price | $1.25 | $0.04 - $1.25 | ||
| Volatility | 67% | 73% | ||
| Remaining life (years) | 2.7 years | 3 years | ||
| Forfeiture rate | 0% | 0% | ||
| Expected annual dividend yield | 0% | 0% |
Volatility was estimated by using a weighting of the Company’s volatility and the average historical volatility of comparable companies from a representative peer group of publicly traded cannabis companies. The risk-free interest rate for the expected life of the Warrants was based on the yield available on government benchmark bonds with an approximate equivalent remaining term. The expected life is based on the contractual term. If any of the assumptions used in the calculation were to increase or decrease, this could result in a material or significant increase or decrease in the estimated fair value of the derivative liability. For example, the following table illustrates an increase or decrease in certain significant assumptions as of the balance sheet dates:
| As of March 31,
2022 (unaudited) | As of December 31, 2021 | |||||||||||||||||||||||
| Input | Effect of
10% Increase | Effect of
10% Decrease | Input | Effect of
10% Increase | Effect of
10% Decrease | |||||||||||||||||||
| Stock price | $ | 2.85 | $ | 9,433 | $ | (9,284 | ) | $ | 3.25 | $ | 12,781 | $ | (10,834 | ) | ||||||||||
| Volatility | 67 | % | $ | 1,818 | $ | (1,755 | ) | 73 | % | $ | 4,473 | $ | (3,210 | ) | ||||||||||
| 12. EQUITY |
Authorized, Issued and Outstanding
The authorized share capital of the Company consists of common shares with an unlimited number of Subordinate Voting Shares (“SVS”), an unlimited number of Multiple Voting Shares (“MVS”), and an unlimited number of Super Voting Shares (“SV”). As of March 31, 2022, the Company had 189,728,625 SVS issued and outstanding and no MVS or SV outstanding. On August 9, 2021, all of the 149,000 previously issued and outstanding SV and all of the 4,000,000 previously outstanding MVS were converted into SVS in accordance with their terms as described in Jushi Holdings Inc.’s Articles of Incorporation. All previously outstanding warrants to acquire SV and MVS were also converted into warrants to acquire SVS, without any other amendment to the terms of such warrants.
| F-20 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Private Placements
In January 2022, the Company closed non-brokered private placement offerings for an aggregate 3,717,392 SVS at a price of $3.68 per share to an existing investor group for aggregate gross proceeds of $13,680.
Restricted Stock and Stock Options
Refer to Note 13 - Share-Based Compensation and Other Benefits for details of restricted stock awards and stock option grants.
Other Equity
Refer to Note 9 - Debt for details of a convertible promissory note classified as equity.
Warrants
Each warrant entitles the holder to purchase one share of the same class of common share. The following table summarizes all warrants outstanding as of March 31, 2022:
| Expiration Date | Exercise
Price ($) | Number of Warrants | ||||||
| 2022 | 1.25 - 1.31 | 322,298 | ||||||
| 2023 | 1.47 - 1.50 | 337,500 | ||||||
| 2024 | 1.25 | 36,040,922 | ||||||
| 2025 | 1.25 - 2.97 | 2,068,508 | ||||||
| 2026 | 4.18 | 300,000 | ||||||
| 2029 | 0.50 - 2.00 | 26,367,627 | ||||||
| Total warrants | 65,436,855 | |||||||
As of March 31, 2022, warrants issued and outstanding have a weighted-average remaining contractual life of 4.5 years. Certain warrants may be net share settled.
The activity of warrants outstanding was as follows:
| Non-Derivative Warrants | Derivative Warrants (2) | Total Number
of Warrants | Weighted
- Average Exercise Price | |||||||||||||
| Balance as of January 1, 2022 | 29,156,048 | 40,124,355 | 69,280,403 | $ | 1.19 | |||||||||||
| Exercised (1) | (393,548 | ) | (3,450,000 | ) | (3,843,548 | ) | 1.26 | |||||||||
| Balance as of March 31, 2022 | 28,762,500 | 36,674,355 | 65,436,855 | $ | 1.19 | |||||||||||
| Exercisable as of March 31, 2022 | 26,899,164 | 36,674,355 | 63,573,519 | $ | 1.15 | |||||||||||
| (1) | The weighted average share price as of the dates of exercise was $3.71. The Company issued 2,676,303 SVS and received $541 in cash proceeds during the three months ended March 31, 2022 for warrants exercised. | |
| (2) | Derivative warrants, which were issued to the Senior Notes holders, have an exercise price of $1.25. These warrants represent a derivative liability and are therefore not classified as equity in the statement of financial position. Refer to Note 11 - Derivative Liabilities. |
| F-21 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated)
|
 |
| 13. | SHARE-BASED COMPENSATION AND OTHER BENEFITS |
The components of share-based compensation expense are as follows:
| Three Months Ended March 31, | ||||||||
| 2022 | 2021 | |||||||
| Stock options | $ | 5,845 | $ | 928 | ||||
| Restricted stock | 832 | 2,538 | ||||||
| Warrants | 287 | 547 | ||||||
| Total share-based compensation expense | $ | 6,964 | $ | 4,013 | ||||
Equity Incentive Plan
Under the Company’s 2019 Equity Incentive Plan, as amended (the “Plan”), non-transferable options to purchase SVS Shares and restricted SVS of the Company may be issued to directors, officers, employees, or consultants of the Company. The Plan authorizes the issuance of up to 15% (plus an additional 2% inducements for hiring employees and senior management) of the number of outstanding shares of common stock (of all classes) of the Company (the “Share Reserve”). Incentive stock options are limited to the Share Reserve as of June 6, 2019. As of March 31, 2022, the maximum number of incentive awards available for issuance under the 2019 Plan, including additional awards available for certain new hires, was 9.1 million.
| (a) | Stock Options |
The stock options issued by the Company are options to purchase SVS of the Company. All stock options issued have been issued to employees of certain subsidiaries of the Company under the Company’s Plan. Such options generally expire in ten years from the date of grant and generally vest ratably over three years from the grant date. The options may be net share settled.
The continuity of stock options outstanding is as follows:
| Number of Stock Options | Weighted- Average Per Share Exercise Price | |||||||
| Issued and Outstanding as of January 1, 2022 | 20,429,120 | $ | 3.20 | |||||
| Granted (1) | 545,000 | $ | 4.20 | |||||
| Exercised (2) | (249,998 | ) | $ | 1.70 | ||||
| Forfeited/expired | (216,668 | ) | $ | 3.63 | ||||
| Issued and Outstanding as of March 31, 2022 | 20,507,454 | $ | 3.24 | |||||
| Exercisable as of March 31, 2022 | 5,872,425 | $ | 2.02 | |||||
| (1) | The weighted-average per share grant date fair value was $4.20. |
| (2) | The weighted-average share price at the date of exercise was $4.03. There were no cash proceeds as all the exercises were net share settled. |
| F-22 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated)
|
 |
The following table summarizes the issued and outstanding stock options as of March 31, 2022:
| Expiration Year | Stock Options Outstanding | Exercise Price | Stock Options Exercisable | |||||||||||
| 2028 | 720,000 | $1.00 - $1.35 | 720,000 | |||||||||||
| 2029 | 7,024,668 | $1.26 - $2.75 | 4,670,322 | |||||||||||
| 2030 | 920,834 | $0.91 - $ 3.98 | 345,824 | |||||||||||
| 2031 | 11,321,952 | $3.70 - $6.53 | 136,279 | |||||||||||
| 2032 | 520,000 | $4.20 | — | |||||||||||
| 20,507,454 | 5,872,425 | |||||||||||||
As of March 31, 2022, stock options outstanding have a weighted-average remaining contractual life of 8.6 years.
In determining the amount of share-based compensation expense related to stock options issued, the Company used the Black-Scholes option-pricing model to establish the measurement date fair value of stock options granted during the period. The following assumptions were applied at the time of grant:
| Three Months Ended March 31, | ||||||||
| 2022 | 2021 | |||||||
| Stock price | $3.94 | $6.13 | ||||||
| Risk-free annual interest rate | 1.83% - 1.90% | 0.45% - 0.69% | ||||||
| Expected annual dividend yield | 0% | 0% | ||||||
| Volatility | 73% | 78% | ||||||
| Expected life of stock options | 5.3 - 7.5 years | 5 - 6.5 years | ||||||
| Forfeiture rate | 0% | 0% | ||||||
Volatility was estimated by using the Company volatility and a weighting of the average historical volatility of comparable companies from a representative peer group of publicly traded cannabis companies. The expected life in years represents the period of time that stock options issued are expected to be outstanding, using the simplified method. The simplified method represents the Company’s best estimate of the expected term of the options, given the Company’s limited history available. The risk-free rate is based on U.S. Treasury bills with a remaining term equal to the expected life of the options. The Company does not anticipate paying dividends in the foreseeable future, and as a result, the expected annual dividend yield is expected to be 0%.
| F-23 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated)
|
 |
| (b) | Restricted Stock Grants |
The Company grants restricted SVS to independent directors, management, former owners of acquired businesses or assets, and to consultants and other employees. The restricted SVS are included in the issued and outstanding SVS. The activity for unvested restricted stock grants is as follows:
| Number of
Restricted Subordinate Voting Shares | ||||
| Unvested restricted stock as of January 1, 2022 | 2,859,151 | |||
| Granted (1) | 5,952 | |||
| Cancelled | (7,813 | ) | ||
| Vested | (100,000 | ) | ||
| Unvested restricted stock as of March 31, 2022 | 2,757,290 | |||
| (1) | The weighted-average per share grant date fair value was $4.20 |
Generally, restricted stock awards will vest either one-third on each anniversary of service from the vesting start date or will be fully vested on the completion of one year of full service from the vesting start date, depending on the award. As of March 31, 2022, unvested restricted stock awards have a weighted-average remaining vesting period of 0.9 years.
| 14. | INCOME TAXES |
The following table summarized the Company’s income tax expense and effective tax rates for the three months ended March 31, 2022 and 2021:
| Three Months Ended March 31, | ||||||||
| 2022 | 2021 | |||||||
| Income (Loss) Before Income Taxes | $ | (14,706 | ) | $ | (22,739 | ) | ||
| Income Tax Expense | $ | 5,051 | $ | 8,312 | ||||
| Effective Tax Rate | (34.3 | )% | (36.6 | )% | ||||
The Company has computed its provision for income taxes based on the actual effective rate for the quarter as the Company believes this is the best estimate for the annual effective tax rate.
Due to its cannabis operations, the Company is subject to the limitation of U.S. Internal Revenue Code of 1986, as amended (“IRC”) Section 280E under which the Company is only allowed to deduct “costs of goods sold”. This results in permanent differences between ordinary and necessary business expenses deemed non-allowable under IRC Section 280E. Therefore, the effective tax rate can be highly variable and may not necessarily correlate with pre-tax income which provides for effective tax rates that are well in excess of statutory tax rates.
The Company’s tax returns benefited from not applying IRC Section 280E to certain entities of the consolidated group either due to the entity not yet starting operations or because the entity had a separate trade or business that was not medical or recreational cannabis operations. The Company determined that it is not more likely than not these tax positions would be sustained under examination.
As a result, the Company has an uncertain tax liability of $41,743 and $41,990 as of March 31, 2022 and December 31, 2021, respectively, inclusive of interest and penalties, which is included in non-current income tax liabilities in the consolidated balance sheets.
| F-24 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated)
|
 |
| 15. | NON-CONTROLLING INTERESTS |
Jushi Europe
The Company’s non-controlling interests are comprised primarily of the non-controlling interest in Jushi Europe. In March 2020, the Company finalized its agreement to expand internationally through the establishment of Jushi Europe. Jushi Europe planned to build out its European business through a combination of strategic acquisitions, partnerships, and license applications, focused on supplying the highest-quality medical cannabis products to patients throughout Europe. During the first quarter of 2020, the Company received $2,000 in cash from the 49% investor partner. The Company owns 51% of Jushi Europe and is exposed, or has rights, to variable returns from Jushi Europe and has the power to govern the financial and operating policies of Jushi Europe through voting control so as to obtain economic benefits, and therefore the Company has consolidated Jushi Europe from the date of acquisition.
During the fourth quarter of 2020, Jushi Europe entered into a credit agreement with a sibling of the Jushi Europe non-controlling partner and received €500 (approximately $614) principal amount. In January 2021, Jushi Europe received €1,000 approximately ($1,214 as of December 31, 2021) principal amount pursuant to a credit agreement with an individual. These credit agreements accrue interest at 5% per annum, payable annually in arrears, and mature on November 11, 2024. The outstanding balance may be prepaid at any time prior to maturity without penalty and may be offset with receivables from the lender. Subsequent to December 31, 2021, it was determined that Jushi Europe was insolvent. The insolvency created an event of default under the unsecured credit agreements and the notes are immediately due and payable.
In April 2021, Jushi Europe entered into an unsecured bridge loan with the Company (51% owner) and an investment partner for a total of €1,800 (~$2,141) principal amount, of which €900 (~$1,070) was contributed by the Company and is eliminated in consolidation. In September 2021, the parties amended the loan agreement and an additional €1,200 (~$1,390) in funding was provided for Jushi Europe, of which 51% was contributed by the Company and is eliminated in consolidation. The bridge loans, as amended, currently accrue interest at 0.5% per annum, which is the foreign marginal lending facility rate plus 25 basis points. All payments including interest are due on maturity, which is 180 days post amendment. These loans had not yet been repaid and are delinquent.
On February 22, 2022, Jushi Europe filed a notice of over-indebtedness with the Swiss courts. The Swiss court declared Jushi Europe’s bankruptcy on May 19, 2022. As a result of the impending proceedings, the Company determined that the assets of Jushi Europe were impaired and recognized an impairment loss of $4,561 for the year ended December 31, 2021.
| 16. | NET INCOME (LOSS) PER SHARE |
The reconciliations of the earnings (loss) and the weighted average number of shares used in the computations of basic and diluted EPS are as follows:
| Three Months Ended March 31, | ||||||||
| 2022 | 2021 | |||||||
| Numerator: | ||||||||
| Net income (loss) and comprehensive income (loss) attributable to Jushi shareholders - basic | $ | (19,757 | ) | $ | (30,876 | ) | ||
| Dilutive effect of net income from derivative warrants liability | (14,309 | ) | — | |||||
| Net loss and comprehensive loss attributable to Jushi shareholders - diluted | $ | (34,066 | ) | $ | (30,876 | ) | ||
| Denominator: | ||||||||
| Weighted-average shares of common stock - basic | 183,226,027 | 157,176,375 | ||||||
| Dilutive effect of derivative warrants | 24,612,879 | — | ||||||
| Weighted-average shares of common stock - diluted | 207,838,906 | 157,176,375 | ||||||
| Net income (loss) per common share attributable to Jushi: | ||||||||
| Basic | $ | (0.11 | ) | $ | (0.20 | ) | ||
| Diluted | $ | (0.16 | ) | $ | (0.20 | ) | ||
On August 9, 2021, all of the 149,000 previously issued and outstanding Super Voting Shares and all of the 4,000,000 previously outstanding Multiple Voting Shares were converted into Subordinate Voting Shares in accordance with their terms as described in Jushi Holdings Inc.’s Articles of Incorporation. Refer to Note 12 - Equity. The number of basic and diluted weighted-average shares outstanding for the three months ended March 31, 2022 and 2021, assumes the conversion of the Multi Voting Share and Super Voting Shares into Subordinate Voting Shares as of the beginning of the year. Other than voting rights, the Multi Voting Shares and Super Voting Shares had the same rights as the Subordinate Voting Shares and therefore all these shares are treated as the same class of common stock for purposes of the earnings (loss) per share calculations.
| F-25 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated)
|
 |
The weighted-average shares outstanding for the three months ended March 31, 2022 and 2021 exclude unvested restricted stock awards as they are not considered participating securities under ASC 260 Earnings Per Share, because although the holders have dividend rights regardless of vesting, they are not obligated to participate in losses until the shares are vested. Shares issued to employees for which a corresponding non-recourse promissory note receivable with the employee is outstanding are not included in the weighted average shares outstanding in accordance with ASC 260 until the notes are repaid.
For purposes of computing diluted earnings (loss) per share for the three months ended March 31, 2022, and 2021, the outstanding stock options, warrants, restricted stock awards, and the convertible notes presented in the table below are considered anti-dilutive because the Company was in a net loss position, or was in a net loss position after adjusting for the effect of the derivative warrants liability. The following table summarizes equity instruments that may, in the future, have a dilutive effect on earnings (loss) per share, but were excluded from consideration in the computation of diluted net loss per share for the three months ended March 31, 2022 and 2021, because the impact of including them would have been anti-dilutive:
| March 31, | ||||||||
| 2022 | 2021 | |||||||
| Stock options | 20,507,454 | 9,658,834 | ||||||
| Warrants | 28,762,500 | 74,883,956 | ||||||
| Restricted stock (1) | 4,307,290 | 7,359,600 | ||||||
| Convertible promissory notes | 910,000 | 930,000 | ||||||
| 54,487,244 | 92,832,390 | |||||||
| (1) | Includes 1,550,000 and 1,583,333 vested restricted stock awards related to outstanding non-recourse promissory notes due from employees as of March 31, 2022 and 2021, respectively |
| 17. | OPERATING EXPENSES |
The major components of operating expenses are as follows:
| Three Months Ended March 31, | ||||||||
| 2022 | 2021 | |||||||
| Salaries, wages and employee related expenses | $ | 17,336 | $ | 9,882 | ||||
| Other general and administrative expenses (1) | 10,611 | 6,804 | ||||||
| Share-based compensation expense | 6,964 | 4,013 | ||||||
| Depreciation and amortization expense | 2,256 | 974 | ||||||
| Acquisition and deal costs | 141 | 238 | ||||||
| Total operating expenses | $ | 37,308 | $ | 21,911 | ||||
| (1) | Other general and administrative expenses are primarily comprised of rent and related expenses, professional fees and legal expenses, marketing and selling expenses, insurance costs, administrative and application fee, software and technology costs, travel, entertainment and conferences and other. |
| F-26 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated)
|
 |
| 18. | OTHER INCOME (EXPENSE) |
The components of other expense, net are as follows:
| Three Months Ended March 31, | ||||||||
| 2022 | 2021 | |||||||
| Gains (losses) on investments and financial assets | — | 1,149 | ||||||
| Losses on debt redemptions/extinguishments/modifications | — | (3,815 | ) | |||||
| Gains (losses) on legal settlements | 24 | (807 | ) | |||||
| Other | (727 | ) | 97 | |||||
| Other expense, net | $ | (703 | ) | $ | (3,376 | ) | ||
| 19. | RELATED PARTY TRANSACTIONS |
The Company had the following related party transactions:
| Three Months Ended March 31,
(unaudited) | As of | |||||||||||||||
| 2022 | 2021 | March 31, 2022 (unaudited) | December 31, 2021 | |||||||||||||
| Nature of transaction | Related Party Income (Expense) | Related Party Prepaid/Receivable (Payable) | ||||||||||||||
| Management services agreements (1) | $ | — | $ | (10 | ) | $ | — | $ | — | |||||||
| Senior Notes - interest expense and principal amount (2) | $ | (9 | ) | $ | (46 | ) | $ | (342 | ) | $ | (1,194 | ) | ||||
| Loans to senior key management - interest charged and principal plus accrued interest outstanding (3) | $ | — | $ | 31 | $ | — | $ | — | ||||||||
| (1) | Includes fees paid to entities controlled by the Company’s Chief Executive Officer, James Cacioppo, for shared costs of administrative services, the provision of financial and research-related advice, and sourcing and assisting in mergers, acquisitions and capital transactions. These amounts are included in operating expenses within the consolidated statements of operations and comprehensive income (loss). Excludes expense from previously issued warrants, which is included in stock-based compensation expense. For the three months ended March 31, 2022 and 2021, total expense for warrants issued in connection with these management services agreements was $445 and $359, respectively. |
| (2) | Interest expense includes amounts related to certain senior key management, directors and other employees as well as a significant investor. |
| (3) | In January 2021, an executive received a loan from the Company of $174 for withholding tax requirements for RSAs issued to the executive, which was repaid in full via payroll deductions. In April 2019, the Company entered into promissory notes with certain executives for the purchase of restricted stock, pursuant to which those executives borrowed an aggregate of $1,813 at a rate of 2.89% per annum, compounded annually. As these loans were non-recourse loans under the accounting guidance they were not reflected in the consolidated balance sheet or table above. As of December 31, 2021, all these balances plus accrued interest have been settled. The balances including accrued interest were settled as part of the executive’s regular pay and bonus, severance or via shares repurchased by the Company. During the year ended December 31, 2021, the Company received 471,757 shares from key management personnel in full settlement of principal amount $2,007 outstanding promissory notes and related interest. |
Refer to Note 15 - Non-Controlling Interests for details of other loans from related parties.
| F-27 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated)
|
 |
| 20. | COMMITMENTS AND CONTINGENCIES |
Contingencies
Although the possession, cultivation and distribution of cannabis for medical use is permitted in certain states, cannabis is classified as a Schedule-I controlled substance under the U.S. Controlled Substances Act and its use remains a violation of federal law. The Company’s operations are subject to a variety of local and state regulations. Failure to comply with one or more of those regulations could result in fines, restrictions on its operations, or losses of permits that could result in the Company ceasing operations. While management believes that the Company is in material compliance with applicable local and state regulations as of March 31, 2022, marijuana regulations continue to evolve and are subject to differing interpretations. As a result, the Company could be subject to regulatory fines, penalties or restrictions at any time. Since federal law criminalizing the use of cannabis preempts state laws that legalize its use, strict enforcement of federal law regarding cannabis would likely result in the Company’s inability to proceed with the Company’s business plans. In addition, the Company’s assets, including real property, cash and cash equivalents, equipment, inventory and other goods, could be subject to asset forfeiture because cannabis is still federally illegal.
Refer to Note 14 - Income Taxes for certain tax-related contingencies and to Note 7 - Acquisitions for an acquisition-related contingent consideration liability.
Claims and Litigation
Any proceeding that may be brought against the Company could have a material adverse effect on the Company’s business plans, financial condition and results of operations. From time to time, the Company may be involved in litigation relating to claims arising out of operations in the normal course of business. Except as disclosed below, as of March 31, 2022, there were no pending or threatened lawsuits that could reasonably be expected to have a material effect on the Company’s financial results. There are also no proceedings in which any of the Company’s directors, officers or affiliates is an adverse party or has a material interest adverse to the Company’s interest.
Refer to Note 15 - Non-Controlling Interests for the information regarding the bankruptcy of Jushi Europe.
Commitments
In addition to the contractual obligations outlined in Note 9 - Debt and Note 10 - Lease Obligations, the Company has the following commitments as of March 31, 2022:
(i) Acquisitions
NuLeaf
In November 2021, the Company entered into a definitive agreement to acquire NuLeaf, Inc. together with its subsidiaries and related companies (collectively, “NuLeaf”), a Nevada-based vertically integrated operator. As of March 31, 2022, NuLeaf currently operates two high-performing adult-use and medical retail dispensaries in Las Vegas, NV and Lake Tahoe, NV, in addition to a 27,000 sq. ft. cultivation facility in Sparks, NV, as well as a 13,000 sq. ft. processing facility in Reno, NV. Additionally, NuLeaf owns a third licensed retail dispensary located directly on Las Vegas Boulevard. Refer to Note 22 - Subsequent Events.
| F-28 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated)
|
 |
(ii) Property and Construction Commitments
In connection with various license applications, the Company may enter into conditional leases or other property commitments which will be executed if the Company is successful in obtaining the applicable license and/or resolving other contingencies related to the license or application.
In addition, the Company expects to incur capital expenditures for leasehold improvements and construction of buildouts of certain locations, including for properties for which the lease is conditional on obtaining the applicable related license or for which other contingencies exist. If the Company were to be unsuccessful in obtaining a particular license or certain other conditions are not met, the previously capitalized improvements and buildouts relating to that license may need to be expensed in future periods in the statements of operations and comprehensive income (loss).
| 21. | FINANCIAL INSTRUMENTS |
The Company applies fair value accounting for all financial assets and liabilities that are recognized or disclosed at fair value in the financial statements on a recurring basis. Fair value is defined as the price that would be received from selling an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date. When determining the fair value measurements for assets and liabilities that are required to be recorded at fair value, the Company considers all related factors of the asset by market participants in which the Company would transact and the market-based risk measurements or assumptions that market participants would use in pricing the asset or liability, such as inherent risk, transfer restrictions, and credit risk.
The Company applies the following fair value hierarchy, which prioritizes the inputs used to measure fair value into three levels, and bases the categorization within the hierarchy upon the lowest level of input that is available and significant to the fair value measurement:
(i) Level 1 – Observable inputs such as unadjusted, quoted prices in active markets for identical assets or liabilities at the measurement date;
(ii) Level 2 – Observable inputs other than Level 1 prices, such as quoted prices for similar assets or liabilities; quoted prices in markets that are not active; or other inputs that are observable or can be corroborated by the observable market data for substantially the full term of the assets or liabilities;
(iii) Level 3 – Unobservable inputs that are supported by little or no market activity and that are significant to the fair value of the assets or liabilities.
The following table sets forth the Company’s financial assets and liabilities, subject to fair value measurements on a recurring basis by level within the fair value hierarchy as of March 31, 2022:
| Level 1 | Level 2 | Level 3 | Total | |||||||||||||
| Financial assets: | ||||||||||||||||
| Equity investment | $ | — | $ | — | $ | 1,500 | $ | 1,500 | ||||||||
| $ | — | $ | — | $ | 1,500 | $ | 1,500 | |||||||||
| Financial liabilities: | ||||||||||||||||
| Derivative liabilities (1) | $ | — | $ | — | $ | 68,975 | $ | 68,975 | ||||||||
| Contingent consideration liabilities | $ | — | $ | — | $ | 8,628 | $ | 8,628 | ||||||||
| $ | — | $ | — | $ | 77,603 | $ | 77,603 | |||||||||
| F-29 |
JUSHI HOLDINGS INC. Notes to the Unaudited Interim Condensed Consolidated Financial Statements (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated)
|
 |
The following table sets forth the Company’s financial assets and liabilities, subject to fair value measurements on a recurring basis by level within the fair value hierarchy as of December 31, 2021:
| Level 1 | Level 2 | Level 3 | Total | |||||||||||||
| Financial assets: | ||||||||||||||||
| Equity investment | $ | — | $ | — | $ | 1,500 | $ | 1,500 | ||||||||
| $ | — | $ | — | $ | 1,500 | $ | 1,500 | |||||||||
| Financial liabilities: | ||||||||||||||||
| Derivative liabilities (1) | $ | — | $ | — | $ | 92,435 | $ | 92,435 | ||||||||
| Contingent consideration liabilities (2) | — | — | 8,223 | 8,223 | ||||||||||||
| $ | — | $ | — | $ | 100,658 | $ | 100,658 | |||||||||
(1) Refer to Note 11 - Derivative Liabilities
(2) Refer to Note 7 - Acquisitions
The carrying amounts of certain financial instruments, including cash and cash equivalents, accounts receivable, accounts payable and certain accrued expenses, and certain other assets and liabilities held at amortized cost, approximate their fair values due to the short-term nature of these instruments. The equity investment approximates its fair value at March 31, 2022 and December 31, 2021. The carrying amounts of the promissory notes approximate their fair values as the effective interest rates are consistent with market rates. The fair value of the 10% Senior Notes as of March 31, 2022 and December 31, 2021 approximated the principal amount.
There were no transfers between hierarchy levels during the three months ended March 31, 2022.
| 22. | SUBSEQUENT EVENTS |
NuLeaf Acquisition
In April 2022, the Company closed on the NuLeaf acquisition. The Company paid upfront consideration comprised of $15,750 in cash (which was drawn down from the acquisition facility in April 2022), subject to working capital adjustments, issued 4,662,384 SVS (with a grant date fair value of $2.87 each), issued $15,750 in an unsecured five-year note. The Company will also pay an additional $10,000 ($3,000 in cash, $3,000 as an addition to the five-year note and the balance in shares) contingent on the opening of a third retail dispensary. In June 2022, the Company opened the third retail dispensary. In July 2022, the Company paid $3,000 in cash, amended the five-year note for an additional $3,000 and issued 888,880 SVS to settle the $10,000 contingent liability. The Company is still in the process of gathering and preparing additional disclosure information for this acquisition, including preliminary purchase price allocations and pro forma financial information. Due to the timing of the acquisition, the Company is not able to provide additional information at this time.
Drawdown on Acquisition Facility
Additionally, in April 2022, the Company drew down $25,000 from the Acquisition Facility to fund the cash portions of the NuLeaf and Apothecarium acquisitions.
Results of Annual and Special Meeting of Shareholders
On June 29, 2022, the Company’s shareholders approved the following:
| - | An amendment to the Company’s 2019 Equity Incentive Plan (the “Plan”) to increase the number of shares that may be issued pursuant to the exercise of incentive stock options under the Plan to an amount equal to 15% (plus an additional 2% inducements for hiring employees and senior management) of the outstanding Shares as of May 31, 2022; and |
| - | An amendment to the Company’s articles to create a new class of preferred shares issuable in series, with the special rights and restrictions attached to each series to be as determined by the board of directors of the Company, if, as and when created and issued, voted to create a class of preferred shares. |
| F-30 |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Shareholders and Board of Directors of Jushi Holdings Inc.
Opinion on the Consolidated Financial Statements
We have audited the accompanying consolidated balance sheets of Jushi Holdings, Inc. and Subsidiaries (collectively known as the “Company”) as of December 31, 2021 and 2020, the related consolidated statements of operations and comprehensive (income) loss, changes in equity, and cash flows for each of the years in the two year period ended December 31, 2021, and the related notes (collectively referred to as the “financial statements”). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2021 and 2020 and the results of its operations and its cash flows for each of the years in the two year period ended December 31, 2021, in conformity with accounting principles generally accepted in the U.S.
Restatement
As discussed in Note 25 to the financial statements, the 2021 Consolidated Statement of Cash Flows has been restated to correct a misstatement.
Basis for Our Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (“PCAOB”) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audit, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ Marcum LLP
Marcum LLP (PCAOB ID 688)
We have served as the Company’s auditor since 2021.
Chicago, Illinois
May 16, 2022, except for Note 25 as to which the date is July 6, 2022
| F-31 |
JUSHI HOLDINGS INC.
(in thousands of U.S. dollars, except share amounts)
| December 31, | ||||||||
| 2021 | 2020 | |||||||
| ASSETS | ||||||||
| CURRENT ASSETS: | ||||||||
| Cash and cash equivalents | $ | 94,962 | $ | 85,857 | ||||
| Accounts receivable, net | 3,200 | 859 | ||||||
| Investments in securities | — | 7,934 | ||||||
| Inventories | 43,319 | 11,945 | ||||||
| Prepaid expenses and other current assets | 12,875 | 4,691 | ||||||
| Total current assets | $ | 154,356 | $ | 111,286 | ||||
| NON-CURRENT ASSETS: | ||||||||
| Property, plant and equipment, net | $ | 137,280 | $ | 38,619 | ||||
| Right-of use assets - finance leases | 94,008 | 29,439 | ||||||
| Other intangible assets, net | 182,466 | 124,312 | ||||||
| Goodwill | 52,920 | 19,093 | ||||||
| Other non-current assets (including related party receivables of $nil and $2,470 as of December 31, 2021 and 2020, respectively) | 27,586 | 12,916 | ||||||
| Non-current restricted cash | 525 | — | ||||||
| Total non-current assets | $ | 494,785 | $ | 224,379 | ||||
| Total assets | $ | 649,141 | $ | 335,665 | ||||
| LIABILITIES AND EQUITY | ||||||||
| CURRENT LIABILITIES: | ||||||||
| Accounts payable | $ | 10,539 | $ | 3,711 | ||||
| Accrued expenses and other current liabilities | 47,972 | 11,356 | ||||||
| Income tax liabilities - current | 6,614 | 1,541 | ||||||
| Debt, net - current portion | 6,181 | 1,659 | ||||||
| Finance lease obligations - current | 12,620 | 3,674 | ||||||
| Total current liabilities | $ | 83,926 | $ | 21,941 | ||||
| NON-CURRENT LIABILITIES: | ||||||||
| Non-current debt, net (including related party principal amounts of $4,578 and $26,089 as of December 31, 2021 and 2020, respectively) | $ | 122,971 | $ | 57,091 | ||||
| Finance lease obligations - non-current | 88,297 | 29,053 | ||||||
| Operating lease liabilities - non-current | 15,163 | 6,040 | ||||||
| Derivative liabilities | 92,435 | 205,361 | ||||||
| Income tax liabilities - non-current | 57,143 | 13,756 | ||||||
| Contingent consideration liabilities | 8,223 | — | ||||||
| Total non-current liabilities | $ | 384,232 | $ | 311,301 | ||||
| Total liabilities | $ | 468,158 | $ | 333,242 | ||||
| COMMITMENTS AND CONTINGENCIES (Note 22) | ||||||||
| EQUITY: | ||||||||
| Common stock (no par value; authorized shares - unlimited; issued shares - 182,707,359 and 132,396,064 Subordinate Voting Shares (including 2,859,151 and 6,438,186 unvested stock awards) as of December 31, 2021 and 2020, respectively; no Super Voting Shares and no Multi Voting Shares as of December 31, 2021; and 149,000 Super Voting Shares and 4,000,000 Multi Voting Shares as of December 31, 2020) | $ | — | $ | — | ||||
| Paid-in capital | 424,788 | 262,145 | ||||||
| Accumulated deficit | (242,418 | ) | (262,669 | ) | ||||
| Total Jushi shareholders’ equity | $ | 182,370 | $ | (524 | ) | |||
| Non-controlling interests | (1,387 | ) | 2,947 | |||||
| Total equity | $ | 180,983 | $ | 2,423 | ||||
| Total liabilities and equity | $ | 649,141 | $ | 335,665 | ||||
The accompanying notes are an integral part of these consolidated financial statements.
| F-32 |
JUSHI HOLDINGS INC.
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE INCOME (LOSS)
(in thousands of U.S. dollars, except share amounts)
The accompanying notes are an integral part of these consolidated financial statements.
| F-33 |
JUSHI HOLDINGS INC.
CONSOLIDATED STATEMENTS OF CHANGES IN EQUITY
(in thousands of U.S. dollars, except share amounts)
| Number of Shares | ||||||||||||||||||||||||||||||||
| Super Voting Shares | Multiple Voting Shares | Subordinate Voting Shares | Paid-In Capital | Accumulated Deficit | Total Jushi Shareholders’ Equity | Non-Controlling Interests | Total Equity | |||||||||||||||||||||||||
| Balances - January 1, 2020 | 149,000 | 4,000,000 | 91,592,638 | $ | 165,436 | $ | (52,062 | ) | $ | 113,374 | $ | 9,660 | $ | 123,034 | ||||||||||||||||||
| Issuance of shares and warrants for cash, net | — | — | 11,500,000 | 29,243 | — | 29,243 | — | 29,243 | ||||||||||||||||||||||||
| TGS Transaction | — | — | (4,800,000 | ) | (7,125 | ) | — | (7,125 | ) | 4,661 | (2,464 | ) | ||||||||||||||||||||
| Purchases of non-controlling interests | — | — | 3,927,911 | 7,395 | — | 7,395 | (13,020 | ) | (5,625 | ) | ||||||||||||||||||||||
| Non-controlling interests - Jushi Europe and other transactions | — | — | — | — | — | — | 1,994 | 1,994 | ||||||||||||||||||||||||
| Acquisition of Agape | — | — | 769,231 | 1,000 | — | 1,000 | 1,560 | 2,560 | ||||||||||||||||||||||||
| Restricted stock grants and vesting, net of forfeitures (including related parties) | — | — | 4,548,099 | 5,898 | — | 5,898 | — | 5,898 | ||||||||||||||||||||||||
| Warrant expense, net of cancellations (including related parties) | — | — | — | 1,128 | — | 1,128 | — | 1,128 | ||||||||||||||||||||||||
| Stock option expense, net of forfeitures (including related parties) | — | — | — | 2,741 | — | 2,741 | — | 2,741 | ||||||||||||||||||||||||
| Warrant exercises | — | — | 24,456,519 | 55,283 | — | 55,283 | — | 55,283 | ||||||||||||||||||||||||
| Stock option exercises | — | — | 26,666 | 41 | — | 41 | — | 41 | ||||||||||||||||||||||||
| Shares issued for settlements | — | — | 375,000 | 1,105 | — | 1,105 | — | 1,105 | ||||||||||||||||||||||||
| Net loss | — | — | — | — | (210,607 | ) | (210,607 | ) | (1,908 | ) | (212,515 | ) | ||||||||||||||||||||
| Balances - December 31, 2020 | 149,000 | 4,000,000 | 132,396,064 | $ | 262,145 | $ | (262,669 | ) | $ | (524 | ) | $ | 2,947 | $ | 2,423 | |||||||||||||||||
| Public offerings | — | — | 13,685,000 | 85,660 | — | 85,660 | — | 85,660 | ||||||||||||||||||||||||
| Purchases of non-controlling interests - Agape | — | — | 500,000 | 1,562 | — | 1,562 | (1,562 | ) | — | |||||||||||||||||||||||
| Acquisition of Grover Beach | — | — | 49,348 | 368 | — | 368 | — | 368 | ||||||||||||||||||||||||
| Acquisition of Nature’s Remedy | — | — | 8,700,000 | 35,670 | — | 35,670 | — | 35,670 | ||||||||||||||||||||||||
| Conversion of Super Voting Shares and Multiple Voting Shares | (149,000 | ) | (4,000,000 | ) | 18,900,000 | — | — | — | — | — | ||||||||||||||||||||||
| Restricted stock grants and vesting, net of forfeitures (including related parties) | — | — | 65,398 | 5,873 | — | 5,873 | — | 5,873 | ||||||||||||||||||||||||
| Warrant expense, net of cancellations (including related parties) | — | — | — | 1,169 | — | 1,169 | — | 1,169 | ||||||||||||||||||||||||
| Stock option expense, net of forfeitures (including related parties) | — | — | — | 7,464 | — | 7,464 | — | 7,464 | ||||||||||||||||||||||||
| Shares issued upon exercise of warrants | — | — | 8,667,173 | 24,676 | — | 24,676 | — | 24,676 | ||||||||||||||||||||||||
| Shares issued upon exercise of stock options | — | — | 216,133 | 171 | — | 171 | — | 171 | ||||||||||||||||||||||||
| Settlement of promissory notes due from related parties | — | — | (471,757 | ) | 30 | — | 30 | — | 30 | |||||||||||||||||||||||
| Net income (loss) | — | — | — | — | 20,251 | 20,251 | (2,772 | ) | 17,479 | |||||||||||||||||||||||
| Balances - December 31, 2021 | — | — | 182,707,359 | $ | 424,788 | $ | (242,418 | ) | $ | 182,370 | $ | (1,387 | ) | $ | 180,983 | |||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements
| F-34 |
JUSHI HOLDINGS INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
(in thousands of U.S. dollars)
| F-35 |
| For the Year Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Assets acquired and liabilities assumed in business combinations: | ||||||||
| Cash and cash equivalents | $ | 3,454 | 1,102 | |||||
| Other current assets | $ | 19,440 | 5,263 | |||||
| Property, plant and equipment, including right-of-use assets | $ | 55,455 | 19,023 | |||||
| Other intangible assets | $ | 60,131 | 45,505 | |||||
| Goodwill | $ | 35,610 | 1,375 | |||||
| Accounts payable and accrued liabilities | $ | (8,604 | ) | (1,076 | ) | |||
| Lease obligations | $ | (32,210 | ) | (18,895 | ) | |||
| Deferred tax liabilities | $ | (20,524 | ) | (1,410 | ) | |||
| Contingent consideration liabilities | $ | (8,223 | ) | — | ||||
The accompanying notes are an integral part of these consolidated financial statements
| F-36 |
|
Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS |
| 1. NATURE OF OPERATIONS |
Jushi Holdings Inc. (the “Company” or “Jushi”) is incorporated under the British Columbia’s Business Corporations Act (“BCBCA”). The Company is a vertically integrated, multi-state cannabis operator engaged in retail, distribution, cultivation, and processing operations in both medical and adult-use markets. As of December 31, 2021, Jushi, through its subsidiaries, owns or manages cannabis operations and/or holds licenses in the adult-use and/or medicinal cannabis marketplace in Illinois, Pennsylvania, Virginia, Massachusetts, Nevada, California and Ohio.
The Company is listed on the Canadian Securities Exchange (the “CSE”) and trades its subordinated voting shares (“SVS”) under the ticker symbol “JUSH”, and trades on the U.S. Over the Counter Stock Market (“OTCQX”) under the symbol JUSHF.
The Company’s head office is located at 301 Yamato Road, Suite 3250, Boca Raton, Florida 33431, U.S.A., and its registered address is Suite 1700, Park Place, 666 Burrard Street, Vancouver, British Columbia V6C 2X8, Canada.
| 2. BASIS OF PRESENTATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES |
Basis of Presentation and Consolidation
The accompanying consolidated financial statements present the consolidated financial position and operations of Jushi Holdings Inc. and its subsidiaries and entities over which the Company has control, in accordance with accounting principles generally accepted in the U.S. (“GAAP”). The accounts of the subsidiaries are prepared for the same reporting period using consistent accounting policies. Intercompany balances and transactions are eliminated in consolidation.
These consolidated financial statements have been prepared assuming the Company will continue as a going concern. US GAAP requires an entity to look forward 12 months from the date the financial statements are available to be issued, (the “look-forward” period) when assessing whether the going concern assumption can be used. The going concern assumption contemplates the realization of assets and satisfaction of liabilities in the normal course of business.
As reflected in these consolidated financial statements, the Company has incurred losses from operations for the years ended December 31, 2021 and 2020 and has an accumulated deficit of $242,418 as of December 31, 2021. As discussed in Note 11 - Debt, the Company’s 10% senior notes (the “Senior Notes”), which as of December 31, 2021 had an aggregate principal amount outstanding of $75,193, mature on January 15, 2023, and its Acquisition Facility (refer to Note 11 - Debt) required the Company to maintain certain covenants which the Company may not have been in compliance with if the court accepted Jushi Europe’s petition for bankruptcy and was projected to violate certain financial covenants further within the next twelve months. This condition raised a substantial doubt about the Company’s ability to continue as a going concern.
| F-37 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Management believes that this substantial doubt has been alleviated due to: (i) an amendment entered into with the lender in April 2022, which included a waiver related to Jushi Europe’s bankruptcy and a change to the terms of the Total Leverage ratio, as defined in the Acquisition Facility agreement, and deferred the commencement date of leverage testing under the Acquisition Facility (ii) available cash and cash equivalents on hand, which were $94,962 as of December 31, 2021; (iii) ongoing efforts with certain lenders to refinance the Senior Notes; (iv) expected continued growth of sales, gross profit and cash flows from operations; and (v) the ability to defer certain capital projects and reallocate funds for debt repayment, if the need arose. The Company also has access to alternative sources of debt and equity financing, including secured borrowings and through a base shelf prospectus, which allows the Company to offer up to C$500,000 in securities in Canada through the end of 2023.
Management’s evaluation of the Company’s liquidity position involved significant judgment. The most significant judgments involved: (i) a probability assessment of the successful refinancing of the Senior Notes; and (ii) the underlying estimates and assumptions for projected sales, gross profits and cash flow. Management believes its plans are achievable. However, there can be no assurance that the Company will be able to refinance the Senior Notes, generate positive results from operations, or obtain additional liquidity when needed or under acceptable terms, if at all.
Functional and Reporting Currency
The functional currency of the Company and its subsidiaries, as determined by management, is the U.S. dollar. The Company’s reporting currency is the U.S. Dollar. These consolidated financial statements are presented in thousands of U.S. dollars unless otherwise noted. Transactions in foreign currencies are recorded at a rate of exchange approximating the prevailing rate at the date of the transaction. Monetary assets and liabilities denominated in foreign currencies at the statement of financial position date are translated into the functional currency at the foreign exchange rate in effect at that date. Realized and unrealized exchange gains and losses are recognized through profit and loss.
Use of Estimates
The preparation of these consolidated financial statements and accompanying notes requires us to make estimates and assumptions that affect amounts reported. Estimates are used to account for certain items such as the valuation of inventories, including stage of growth of cannabis plants, the likelihood the plants will grow to full maturity, and the estimated yields from harvest and conversion to finished goods; the assessment of business combinations and asset acquisitions and the fair values of the assets and liabilities acquired; the fair value of purchase consideration and contingent consideration; the useful lives of definite lived intangible assets and property and equipment; stock-based compensation, leases, income tax provision and uncertain tax positions, the collectability of receivables and other items requiring judgment. Estimates are based on historical information and other assumptions that management believes are reasonable under the circumstances. Due to the inherent uncertainty involved with estimates, actual results may differ materially.
Cash and Cash Equivalents and Restricted Cash
The Company considers cash deposits and all highly liquid investments with an original maturity of three months or less to be cash equivalents. Cash and cash equivalents include cash deposits in financial institutions and cash held at retail locations. Cash and cash held in money market investments are carried at fair value. When the use of a cash balance is subject to regulatory or contractual restrictions and therefore not available for general use by the Company, the Company classifies the cash as restricted cash. The Company had $525 restricted cash as of December 31, 2021 and had no restricted cash as of December 31, 2020.
The Company maintains cash with various U.S. banks and credit unions with balances in excess of the Federal Deposit Insurance Corporation and National Credit Union Share Insurance Fund limits, respectively. The failure of a bank or credit union where the Company has significant deposits could result in a loss of a portion of such cash balances in excess of the insured limit, which could materially and adversely affect the Company’s business, financial condition and results of operations.
| F-38 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Investments in Securities
Investments in securities consists of investments in mutual funds and equity securities. These investments are measured at fair value with realized and unrealized gains and losses recognized in other income (expense) in the consolidated statement of operations in the period in which they occur.
Accounts Receivable and Expected Credit Losses
Accounts receivable are recorded at the invoiced amount and do not bear interest. Expected credit losses (or “allowance”) reflects the Company’s estimate of amounts in its existing accounts receivable that may not be collected due to customer claims or customer inability or unwillingness to pay. Collectability of accounts receivable is reviewed on an ongoing basis. Expected credit losses are determined based on a combination of factors, including the Company’s risk assessment regarding the specific exposures, credit worthiness of its customers, historical collection experience and length of time the receivables are past due. Account balances are charged off against the allowance when the Company believes it is probable the receivable will not be recovered. During the years ended December 31, 2021 and 2020, the Company’s recorded expected credit losses were not material given that a significant majority of the Company’s sales were collected in cash at the point of sale.
Inventories
Inventories are comprised of raw materials, work in process, finished goods and packaging materials. Inventories primarily consist of cannabis plants, dried cannabis, cannabis trim, and cannabis derivatives such as oils and edible products, and accessories. Inventories are initially recorded at cost and subsequently at the lower of cost or net realizable value. Costs incurred during the growing and production processes are capitalized as incurred, as adjusted for estimated normal capacity and expected yields when incurred. These costs include direct materials, labor and manufacturing overhead used in the growing and production processes. Net realizable value is determined as the estimated selling price in the ordinary course of business less the estimated costs to complete and sell. Cost is primarily determined on an average cost basis. The Company also reviews inventory for obsolete and slow-moving goods and writes down inventory to net realizable value.
Property, Plant and Equipment
Property, plant, and equipment (“PP&E”) are measured at cost less accumulated depreciation, and impairment losses, if applicable. Purchased property and equipment are initially recorded at cost, or, if acquired in a business combination, at the acquisition date fair value. Finance lease right-of-use assets are recognized at inception based on the present value of minimum future lease payments. Depreciation is recognized on a straight-line basis over the following periods:
| Buildings and building components | 10 - 30 years |
| Leasehold improvements | The lesser of the term of the lease or the estimated useful life of the asset: 1-28 years |
| Machinery and equipment | 1 - 10 years |
| Computer equipment | 2 - 3 years |
| Furniture and fixtures | 2 - 7 years |
| Finance lease ROU assets - buildings | 14 - 28 years |
| Finance lease ROU assets - machinery and equipment | 3 - 5 years |
Land has an unlimited useful life and is, therefore, not depreciated. An asset’s residual value, useful life and depreciation method are reviewed annually and adjusted prospectively if necessary.
| F-39 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Construction-in-process (“CIP”) represents assets under construction and is measured at cost, including borrowing costs incurred during the construction of qualifying assets. When construction on a property is complete and available for use, the cost of construction which has been included CIP will be reclassified to buildings and improvements, leasehold improvements or furniture and fixtures, as appropriate, and depreciated.
Impairment of Long-Lived Assets
Property and equipment, as well as right-of-use assets and definite lived intangible assets, are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset or asset group may not be recoverable. If circumstances require these long-lived assets to be tested for possible impairment and the Company’s analysis indicates that a possible impairment exists based on an estimate of undiscounted future cash flows, the Company is required to estimate the fair value of the asset.
An impairment charge is recorded for the excess of the asset’s or asset group’s carrying value over its fair value, if any. Asset groups have identifiable cash flows and are largely independent of other asset groups. The Company assesses the fair value of long-lived assets using commonly accepted techniques, and may use more than one method, including recent third-party comparable sales and discounted cash flow models. The Company’s impairment analyses require management to apply judgment in estimating future cash flows as well as asset fair values, and other assumptions.
Refer to Note 6 - Property, Plant and Equipment for information regarding the disposal/impairment of certain long-lived assets.
Business Combinations
Acquisitions are assessed under ASC 805, Business Combinations (“ASC 805”), and judgement is required to determine whether a transaction qualifies as an asset acquisition or businesses combination. The Company includes the results of operations of the businesses acquired from the acquisition date. Acquisition-related expenses are recognized separately from a business combination and are expensed as incurred.
The Company allocates the purchase price of the business combination to the assets acquired and liabilities assumed based on their estimated fair values. The excess of the purchase price over the fair values of identifiable assets and liabilities is recorded as goodwill. To the extent the fair value of the net assets acquired, including other identifiable assets, exceeds the purchase price, a bargain purchase gain is recognized in the statement of operations and comprehensive income (loss).
Acquisitions of assets or a group of assets that do not meet the definition of a business are accounted for as asset acquisitions using the cost accumulation method, whereby the cost of the acquisition, including certain transaction costs, is allocated to the assets acquired on the basis of relative fair values. No goodwill is recognized in an asset acquisition.
Variable Interest Entities
The Company determines at the inception of each arrangement whether an entity in which the Company has made an investment or in which it has other variable interests is considered a variable interest entity (“VIE”). The Company consolidates VIEs when it is the primary beneficiary. The Company is the primary beneficiary of a VIE when it has the power to direct activities that most significantly affect the economic performance of the VIE and has the obligation to absorb the majority of their losses or benefits. If the Company is not the primary beneficiary in a VIE, the VIE will be accounted for in accordance with other applicable accounting guidance. Periodically, the Company assesses whether any changes in the Company’s interest or relationship with the entity affect the determination of whether the entity is a VIE and, if so, whether the Company is the primary beneficiary.
| F-40 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
The following are the Company’s VIEs as of December 31, 2020 and for which the Company was the primary beneficiary:
| Entity | Location | Ownership | |||
| Franklin Bioscience OH, LLC (1) | Ohio | 0 | % | ||
| Franklin Bioscience NV LLC (2) | Nevada | 0 | % |
(1) In August 2021, the Company completed the acquisition of 100% of Franklin Bioscience OH, LLC (“FBS - OH”). The Company had been operating FBS - OH under a management services agreement (“MSA”) since 2019.
(2) In April 2021, the Company completed the acquisition of 100% of the equity of Franklin Bioscience NV LLC (“FBS Nevada”). The Company had been operating FBS Nevada under a MSA since 2019.
Intangible Assets
Intangible assets are recorded at cost, less accumulated amortization and impairment losses, if any. Intangible assets acquired in a business combination are measured at fair value at the acquisition date. The estimated useful lives, residual values and amortization methods are reviewed annually, and any changes in estimates are accounted for prospectively. Finite lived intangible assets are amortized using the straight-line method over their estimated useful lives. Refer to Note 8 - Goodwill and Other Intangible Assets.
Goodwill and Indefinite Lived Intangibles
In accordance with ASC 350 Intangibles - Goodwill and Other the Company reviews goodwill and indefinite lived intangibles for impairment at the reporting unit level annually, or, when events or circumstances dictate, more frequently. At the time of a business combination, goodwill is either assigned to a specific reporting unit or allocated between reporting units based on the relative fair value of each reporting unit. The Company first performs a qualitative assessment to determine if it is more-likely-than-not that the reporting unit’s carrying value, which includes goodwill and intangibles, is less than its fair value, indicating a potential for impairment, and therefore requiring a quantitative assessment. If the Company determines that a quantitative impairment test is required, the Company typically uses a combination of an income approach, i.e., a discounted cash flow calculation, and a market approach, i.e., using a market multiple method, to determine the fair value of each reporting unit, and then compare the fair value to its carrying amount to determine the amount of impairment, if any. If a reporting unit’s fair value is less than its carrying amount, the Company would record an impairment charge based on that difference, up to the amount of goodwill and intangibles allocated to that reporting unit.
The quantitative impairment test requires the application of a number of significant assumptions, including estimated revenue growth rates, profit margins, terminal value growth rates, market multiples, and discount rates. The projections of future cash flows used to assess the fair value of the reporting units are based on the internal operation plans reviewed by management. The market multiples are based on comparable public company multiples. The discount rates are based on the risk-free rate of interest and estimated risk premiums for the reporting units at the time the impairment analysis is prepared or such evaluation date.
The Company performs its annual impairment tests on goodwill and indefinite-lived intangible assets during the fourth quarter. Refer to Note 8 - Goodwill and Other Intangible Assets.
Leases
In accordance with ASC 842 Leases the Company determines if an arrangement is a lease at inception. When a leasing arrangement is identified, a determination is made at inception as to whether the lease is an operating or a finance lease. Operating lease right-of-use (“ROU”) assets and operating lease (current and non-current) liabilities and finance lease ROU assets and finance lease (current and non-current) liabilities are recognized in the consolidated balance sheets. Leases with an initial term of 12 months or less are not recorded on the consolidated balance sheets and are expensed in the consolidated statements of operations on a straight-line basis over the lease term.
| F-41 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Right of use (“ROU”) assets represent the Company’s right to use an underlying asset in which the Company obtains substantially all of the economic benefits and the right to direct the use of the asset during the lease term. Lease liabilities represent the Company’s obligation to make lease payments arising from the lease. ROU assets and lease liabilities are recognized at the commencement date based on the present value of lease payments over the lease term, using a discount rate equivalent to the Company’s incremental borrowing rate for a term similar to the estimated duration of the lease, as the rates implicit in the Company’s leases are not readily available. Payments that are not fixed at the commencement of the lease are considered variable and are excluded from the ROU asset and lease liability calculations. For finance leases, interest expense on lease liabilities is recognized using the effective interest method, and amortization of the related ROU asset is on a straight-line basis. Refer to Property, Plant and Equipment above for the useful lives of finance lease ROU assets. Operating lease cost, which includes the interest on the lease liability and amortization of the related ROU asset, is recognized on a straight-line basis over the lease term.
Topic 842 requires lessees to discount lease payments using the rate implicit in the lease if that rate is readily available in accordance with Topic 842. If that rate cannot be readily determined, the lessee is required to use its incremental borrowing rate. The Company generally uses the incremental borrowing rate when initially recording leases. Information from the lessor regarding the fair value of underlying assets and initial direct costs incurred by the lessor related to the leased assets is not available. The Company determines the incremental borrowing rate as the interest rate the Company would pay to borrow over a similar term the funds necessary to obtain an asset of a similar value to the right-of-use asset in a similar economic environment. Topic 842 requires lessees to estimate the lease term. In determining the period which the Company has the right to use an underlying asset, management considers the non-cancellable period along with all facts and circumstances that create an economic incentive to exercise an extension option, or not to exercise a termination option.
Segments
The Company operates a vertically integrated cannabis business in one reportable segment: the cultivation, manufacturing, distribution and sale of cannabis in the U.S. All revenues for the year ended December 31, 2021 and 2020 were generated within the U.S., and substantially all long-lived assets are located within the U.S.
| F-42 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Revenue Recognition
The Company recognizes revenue in accordance with ASC 606 Revenue from Contracts with Customers (“ASC 606”). ASC 606 requires revenue to be recognized when control of the promised goods or services are transferred to customers at an amount that reflects the consideration that the Company expects to receive. Application of ASC 606 requires a five-step model applicable to all product offering revenue streams as follows: (1) Identify a customer along with a corresponding contract; (2) Identify the performance obligation(s) in the contract to transfer goods or provide distinct services to a customer; (3) Determine the transaction price the Company expects to be entitled to in exchange for transferring promised goods or services to a customer; (4) Allocate the transaction price to the performance obligation(s) in the contract; and (5) Recognize revenue when or as the Company satisfies the performance obligation(s).
Contract assets, as defined in ASC 606, include amounts that represent the right to receive payment for goods and services that have been transferred to the customer with rights conditional upon something other than the passage of time. Contract liabilities are defined in the standard to include amounts that reflect obligations to provide goods and services for which payment has been received. There are no contract assets or unsatisfied performance obligations as of December 31, 2021 and 2020.
The Company’s contracts with customers for the sale of dried cannabis, cannabis oil and other cannabis related products may consist of multiple performance obligations. Revenue from the direct sale of cannabis to customers for a fixed price is recognized when the Company transfers control of the goods to the customer at the point of sale and the customer has paid for the goods. Revenue is measured based on the amount of consideration that the Company can expect to receive in exchange for those goods or services, reduced by promotional discounts and estimates for return allowances and refunds. Taxes collected from customers for remittance to governmental authorities are excluded from revenue.
For some of its locations, the Company offers a loyalty reward program to its dispensary customers. A portion of the revenue generated in a sale is allocated to the loyalty points earned. The Company records a reduction in revenue and a liability based on the estimated probability of the point obligation incurred, calculated based on a standalone selling price of each point. Loyalty reward credits issued as part of a sales transaction results in revenue being deferred until the loyalty reward is redeemed by the customer. Loyalty points expire six months from award date and the Company expects outstanding loyalty points to be redeemed within six months.
The Company has three revenue streams: (i) cannabis retail (ii) cannabis wholesale; and (iii) other. The retail revenues are comprised of cannabis operations for medical and adult-use dispensaries. The Company’s wholesale revenues are comprised of cannabis cultivation, processing, production and distribution of cannabis for medical and adult-use. The Company’s other operations primarily include the Company’s hemp/cannabidiol (“CBD”) retail operations. Any intercompany revenue and any costs between entities are eliminated to arrive at consolidated totals.
The following table summarizes revenue disaggregated by revenue stream:
| Year Ended December 31, | ||||||||||||||||||||||||
| 2021 | 2020 | |||||||||||||||||||||||
| Gross Revenue | Intercompany Revenue | Revenue to External Customers | Gross Revenue | Intercompany Revenue | Revenue to External Customers | |||||||||||||||||||
| Retail cannabis | $ | 195,085 | $ | — | $ | 195,085 | $ | 75,499 | $ | — | $ | 75,499 | ||||||||||||
| Wholesale cannabis | 29,969 | (16,177 | ) | 13,792 | 6,639 | (1,901 | ) | 4,738 | ||||||||||||||||
| Other | 415 | — | 415 | 535 | — | 535 | ||||||||||||||||||
| Eliminations | (16,177 | ) | 16,177 | — | (1,901 | ) | 1,901 | — | ||||||||||||||||
| Consolidated revenue | $ | 209,292 | $ | — | $ | 209,292 | $ | 80,772 | $ | — | $ | 80,772 | ||||||||||||
| F-43 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Costs of Goods Sold
Cost of goods sold includes the costs directly attributable to revenue recognition and includes compensation and fees for services, travel and other expenses for services and costs of products and equipment.
Operating Expenses
Operating expenses represent costs incurred at the Company’s corporate and administrative offices, primarily related to: compensation expenses, including share-based compensation; depreciation and amortization; professional fees and legal expenses; marketing, advertising and selling costs; facility-related expenses, including rent and security; insurance; software and technology expenses; impairments; and acquisition and deal costs. Advertising and promotion costs are included as a component of operating expenses and are expensed as incurred.
Stock-Based Payment Arrangements
The Company accounts for stock-based compensation in accordance with the fair value recognition provisions of ASC 718 Compensation – Stock Compensation, which requires that all stock-based compensation to employees and non-employees, including grants of employee stock options, restricted stock awards (“RSAs”) and compensatory warrants, be recognized as an expense in the consolidated financial statements based on their fair values over the vesting period. For the years ended December 31, 2021 and 2020, total stock-based compensation expense is included in operating expenses.
The fair value of stock options and compensatory warrants is estimated using the Black-Scholes option pricing formula that requires assumptions for expected volatility, expected dividends, the risk-free interest rate and the expected term. The Company accounts for forfeitures of stock-based grants as they occur. If any of the assumptions used in the Black-Scholes model or the anticipated number of shares to be awarded change significantly, stock-based compensation expense may differ materially in the future from that recorded in the current period. RSAs are measured at their grant date market value.
Equity-settled stock-based payments under stock-based payments plans are recognized as an expense in profit or loss with a corresponding credit to equity. If vesting periods or other vesting conditions apply, the expense is allocated over the vesting period. No adjustment is made to any expense recognized in prior periods if vested stock options or warrant awards expire without being exercised.
Income Taxes
Income tax expense is the total of the current year income tax due or refundable and the change in deferred tax assets and liabilities. Deferred tax assets and liabilities are the expected future tax amounts for the temporary differences between carrying amounts and tax bases of assets and liabilities, computed using enacted rates. A valuation allowance, if needed, reduces deferred tax assets to the amount expected to be realized.
As the Company operates in the legal cannabis industry, the Company is subject to the limits of Internal Revenue Code (“IRC”) Section 280E for US federal income tax purposes as well as state income tax purposes for all states except for California and Colorado. Under IRC Section 280E, the Company is only allowed to deduct expenses directly related to sales of product, i.e. the cost of producing the products or cost of production. This results in permanent differences between ordinary and necessary business expenses deemed non-allowable under IRC Section 280E.
In accordance with ASC 740 Income Taxes, a tax position is recognized as a benefit only if it is more likely than not that the tax position would be sustained in a tax examination, with a tax examination being presumed to occur. The amount recognized is the largest amount of tax benefit that is greater than 50% likely of being realized upon examination. For tax positions not meeting the more likely than not test, no tax benefit is recorded.
| F-44 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
The Company is treated as a U.S. corporation for US federal income tax purposes under IRC Section 7874 and is subject to US federal income tax on its worldwide income. However, for Canadian tax purposes, the Company, regardless of any application of IRC Section 7874, is treated as a Canadian resident company (as defined in the Income Tax Act (Canada) for Canadian income tax purposes. As a result, the Corporation is subject to taxation both in Canada and the U.S.
Fair Value of Financial Instruments
The Company applies fair value accounting for all financial assets and liabilities that are recognized or disclosed at fair value in the financial statements on a recurring basis. Fair value is defined as the price that would be received from selling an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date. When determining the fair value measurements for assets and liabilities that are required to be recorded at fair value, the Company considers all related factors of the asset by market participants in which the Company would transact and the market-based risk measurements or assumptions that market participants would use in pricing the asset or liability, such as inherent risk, transfer restrictions, and credit risk.
The Company applies the following fair value hierarchy, which prioritizes the inputs used to measure fair value into three levels, and bases the categorization within the hierarchy upon the lowest level of input that is available and significant to the fair value measurement: (i) Level 1 – Observable inputs such as unadjusted, quoted prices in active markets for identical assets or liabilities at the measurement date; (ii) Level 2 – Observable inputs other than Level 1 prices, such as quoted prices for similar assets or liabilities; quoted prices in markets that are not active; or other inputs that are observable or can be corroborated by the observable market data for substantially the full term of the assets or liabilities; (iii) Level 3 – Unobservable inputs that are supported by little or no market activity and that are significant to the fair value of the assets or liabilities. Refer to Note 23 - Financial Instruments.
COVID-19
At the onset of the COVID-19 pandemic, the Company implemented new procedures at all operating locations to better protect the health and safety of its employees, medical patients, and customers across its network of dispensaries at the onset of the COVID-19 outbreak. Depending on the location, some of the initiatives include, but are not limited to: reducing the number of point-of-sale registers, restricting the number of people permitted in-store, limiting store hours to those most susceptible, and offering curbside pick-up. The Company has also directed a significant amount of traffic to its recently launched online educational tool and reservation platform, www.beyond-hello.com, which enables a medical patient or customer to view real-time pricing and product availability, and reserve products for convenient in-store pick-up at BEYOND / HELLO™ locations across Pennsylvania, Illinois, California, and Virginia. Although the Company’s dispensaries and grower-processor facilities have remained open throughout the pandemic, the Company has experienced disruptions related to supply chains, labor supply and delays in expansion projects, and, in certain states, cannabis license applications, and may experience further disruptions as result of COVID-19. In particular:
| · | The Company may experience significant reductions or volatility in demand for its products as customers may not be able to purchase its products due to illness, quarantine or government or self-imposed restrictions placed on its dispensaries’ operations or reduction in operational hours due to labor shortages; | |
| · | Inability to meet production demand if a particular grower-processor or section of the grower-processor is closed due to illness, quarantine or government or self-imposed restrictions placed on our facility operations or reduction in operational hours due to labor shortages; |
| · | Social distancing measures or changes in consumer spending behaviors due to COVID-19 may impact traffic in the Company’s dispensaries and such actions could result in a loss of sales and profit; |
| · | The Company may experience temporary or long-term disruptions in its supply chain; |
| · | Transportation delays and cost increases, closures or disruptions of businesses and facilities, or social, economic, political or labor instability, may impact the Company’s or its suppliers’ operations or its customers; and |
| · | The Company may experience impairments of assets in markets that are disproportionately impacted by these and other effects of the COVID-19 pandemic. |
To date, the Company’s financial condition and results of operations have not been materially impacted by COVID-19. The extent to which the COVID-19 pandemic impacts the Company’s future results will depend on future developments, which are highly uncertain and cannot be predicted with certainty, including possible future outbreaks of new strains of the virus and governmental and consumer responses to such future developments.
Recent Accounting Pronouncements
In June 2016, the Financial Accounting Standards Board (“FASB”) issued Accounting Standards Update (“ASU”) No. 2016-13, “Financial Instruments - Credit Losses (Topic 326) Measurement of Credit Losses on Financial Instruments” (“ASU 2016-13”), which replaces the incurred loss model with a current expected credit loss model and requires consideration of a broader range of reasonable and supportable information to explain credit loss estimates. Under the new standard, the Company recognizes a loss allowance based on lifetime expected credit losses at each reporting date from the date of the trade receivable. The Company is not required to track the changes in credit risk. The guidance must be adopted using a modified retrospective transition method through a cumulative-effect adjustment to retained earnings in the period of adoption. The adoption of the standard did not have a material impact on the Company’s consolidated financial statements.
In August 2018, the FASB issued ASU 2018-13, “Fair Value Measurement (Topic 820): Disclosure Framework— Changes to the Disclosure Requirements for Fair Value Measurement” (“ASU 2018-13”), which removes, modifies and adds certain disclosure requirements in Topic 820 “Fair Value Measurement”. Per ASU 2018-13 certain disclosures are eliminated which relate to transfers and the valuations process, modifies disclosures for investments that are valued based on net asset value, clarifies the measurement uncertainty disclosure, and requires additional disclosures for Level 3 fair value measurements. ASU 2018-13 must be applied prospectively and is effective for the Company for fiscal years beginning after December 15, 2019. The adoption of the standard did not have a material impact on the Company’s Consolidated Financial Statements.
| F-45 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
In December 2019, the FASB issued ASU 2019-12 Income Taxes (Topic 740), which simplifies certain aspects of accounting for income taxes. The guidance is effective for interim and annual reporting periods beginning after December 15, 2020, and early adoption is permitted. The adoption of this ASU did not have a material effect on the Company’s consolidated financial statements.
In June 2020, the FASB issued ASU 2020-06 Debt-Debt with Conversion and Other Options (Subtopic 470-20) and Derivatives and Hedging-Contracts in Entity’s Own Equity (Subtopic 815-40): Accounting for Convertible Instruments and Contracts in an Entity’s Own Equity, which simplifies accounting for convertible instruments by removing major separation models required under current GAAP. This ASU also removes certain settlement conditions that are required for equity contracts to qualify for the derivative scope exception and simplifies the diluted earnings per share calculation in certain areas. The amendments in this ASU are effective for annual and interim periods beginning after December 15, 2021, although early adoption is permitted. The Company is in the process of evaluating the impact of this new guidance on its consolidated financial statements for the convertible Promissory Notes issued at a discount and with a Beneficial Conversion Feature.
In May 2021, the FASB issued ASU 2021-04, “Earnings Per Share (Topic 260), Debt—Modifications and Extinguishments (Subtopic 470-50), Compensation—Stock Compensation (Topic 718), and Derivatives and Hedging—Contracts in Entity’s Own Equity (Subtopic 815-40): Issuer’s Accounting for Certain Modifications or Exchanges of Freestanding Equity-Classified Written Call Options”— The FASB issued guidance to clarify and reduce diversity in an issuer’s accounting for modifications or exchanges of freestanding equity-classified written call options (for example, warrants) that remain equity classified after modification or exchange. The amendments in this ASU are effective for annual and interim periods beginning after December 15, 2021, although early adoption is permitted. The Company is in the process of evaluating the impact of this new guidance on our consolidated financial statements.
In October 2021, the FASB issued ASU 2021-08, Business Combinations (Topic 805): Accounting for Contract Assets and Contract Liabilities from Contracts with Customers. The FASB issued guidance which require that an entity (acquirer) recognize and measure contract assets and contract liabilities acquired in a business combination in accordance with Topic 606. At the acquisition date, an acquirer should account for the related revenue contracts in accordance with Topic 606 as if it had originated the contracts. To achieve this, an acquirer may assess how the acquiree applied Topic 606 to determine what to record for the acquired revenue contracts. Generally, this should result in an acquirer recognizing and measuring the acquired contract assets and contract liabilities consistent with how they were recognized and measured in the acquiree’s financial statements (if the acquiree prepared financial statements in accordance with generally accepted accounting principles). The amendments in this ASU are effective for annual and interim periods beginning after December 15, 2022, although early adoption is permitted. The Company is in the process of evaluating the impact of this new guidance on our consolidated financial statements.
| F-46 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| 3. INVESTMENTS IN SECURITIES |
A summary of investments in securities is as follows:
| Level within the Fair Value Hierarchy (Note 23) | December 31, 2021 | December 31, 2020 | ||||||||
| Investments in mutual funds | Level 1 | $ | — | $ | 5,783 | |||||
| Cresco shares and warrants | Level 1 & Level 2 | — | 2,151 | |||||||
| Total investments in securities | $ | — | $ | 7,934 | ||||||
Investments in Mutual Funds
During the year ended December 31, 2021, the Company sold all of its investments in mutual funds and recognized gains of $115. The Company owned 603,749 shares in mutual funds with a fair value of $9.58 per share as of December 31, 2020. For the year ended December 31, 2020, the Company recognized a net investment loss of $451.
Cresco Shares and Warrants
In October 2019, as consideration for its sale of its 16.5% equity investment in Gloucester Street Capital, LLC (“GSC”), the parent company of New York state licensed cannabis operator Valley Agriceuticals, LLC, to Cresco Labs Inc. “Cresco”), the Company was issued 7,180 of Cresco proportionate voting shares (which were converted in January 2020 into a total of 1,436,000 subordinate voting shares), 1,657 warrants for proportionate voting shares (which were convertible into 331,400 warrants for subordinate voting shares) and received short-term secured notes (the “Cresco Notes”) and cash. During the year ended December 31, 2020, the Company received payment of $5,193 for the Cresco Notes and was also granted 330 proportionate voting shares (which were subsequently converted on a 200:1 basis into 66,000 subordinate voting shares) as payment for the interest accrued and recognized $66 in related investment losses.
During the year ended December 31, 2020, the Company sold the majority of its Cresco subordinate voting shares for aggregate proceeds of $13,404. As of December 31, 2020, the Company owned a remaining 24,600 subordinate voting shares with a fair value of $9.87 each, for a total of $243, and the Cresco warrants were valued at $1,908. The fair value of the tradable shares was determined based on the share price. The fair value of the warrants was determined based on a Black-Scholes model using the following assumptions:
| December 31, 2020 | |
| Quoted market price | $9.87 |
| Strike price | $4.24 |
| Estimated life | 0.75 years |
| Volatility | 70% |
| Risk-free interest rate | 0.10% |
| Dividend rate | —% |
| F-47 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
The Company recognized net investment losses of $1,550 relating to the Cresco subordinate voting shares and warrants during the year ended December 31, 2020. The warrants had cumulative unrealized gains of $1,085 relating to securities still held by the Company as of December 31, 2020.
During the year ended December 31, 2021, the Company net exercised the warrants and received 207,599 Cresco subordinate voting shares. The Company subsequently sold all its outstanding Cresco shares during the year ended December 31, 2021.
The Company is also eligible to receive certain contingency payouts of up to a total $1,650 in cash plus 440,000 Cresco shares, from the sale of its minority interest in GSC, which are tied to both the performance of the GSC operations as well as the development of the New York market. The Company records a gain contingency at the earlier of when the contingency is realized or becomes realizable. As of December 31, 2021 and 2020, the Company had not recognized a receivable as the gain contingency was not determined to be realizable.
Organigram
During the year ended December 31, 2020, the Company realized $407 in investment losses when it sold the Organigram, Inc. (“OGI”) shares received in connection with the acquisition of two dispensaries in Illinois in 2020 (refer to Note 7 - Acquisitions).
Refer to Note 9 - Other Non-Current Assets for a non-current equity investment.
| 4. INVENTORIES |
The components of inventories, net, are as follows:
| December 31, 2021 | December 31, 2020 | |||||||
| Cannabis plants | $ | 6,347 | $ | 1,553 | ||||
| Harvested cannabis | 5,180 | 1,454 | ||||||
| Total raw materials | $ | 11,527 | $ | 3,007 | ||||
| Work in process | 8,756 | 996 | ||||||
| Finished goods | 23,036 | 7,942 | ||||||
| Total inventories, net | $ | 43,319 | $ | 11,945 | ||||
| 5. PREPAID EXPENSES AND OTHER CURRENT ASSETS |
The components of prepaid expenses and other current assets are as follows:
| December 31, 2021 | December 31, 2020 | |||||||
| Prepaid expenses and deposits | $ | 3,837 | $ | 3,318 | ||||
| Landlord receivables for leasehold improvements | 7,357 | 806 | ||||||
| Employee receivable | 248 | — | ||||||
| Other current assets | 1,433 | 567 | ||||||
| Total prepaid expenses and other current assets | $ | 12,875 | $ | 4,691 | ||||
| F-48 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| 6. PROPERTY, PLANT AND EQUIPMENT |
The components of property, plant and equipment (PP&E) and accumulated depreciation are as follows:
| December 31, 2021 | December 31, 2020 | |||||||
| Buildings and building components | $ | 49,697 | $ | 6,679 | ||||
| Land | 12,380 | 1,994 | ||||||
| Leasehold improvements | 24,042 | 22,261 | ||||||
| Machinery and equipment | 12,656 | 1,218 | ||||||
| Computer equipment | 2,221 | 770 | ||||||
| Furniture and fixtures | 8,000 | 3,405 | ||||||
| Construction-in-process | 35,625 | 4,351 | ||||||
| Total property, plant and equipment - gross | $ | 144,621 | $ | 40,678 | ||||
| Less: Accumulated depreciation | (7,341 | ) | (2,059 | ) | ||||
| Total property, plant and equipment - net | $ | 137,280 | $ | 38,619 | ||||
Construction-in-process (“CIP”) represents assets under construction for manufacturing and retail build-outs not yet ready for use.
For the years ended December 31, 2021 and 2020, total depreciation, including depreciation from assets held under finance leases (which are reflected separately in the consolidated balance sheets), was $8,808 and $2,293, respectively, of which $4,970 and $341, respectively, was absorbed into inventory production. Interest expense capitalized to PP&E totaled $977 and $1,074 for the years ended December 31, 2021 and 2020, respectively.
For the year ended December 31, 2021, the Company recognized fixed asset impairment charges of $4,451 related to construction-in-process and land for Jushi Europe, which the Company determined will no longer be used and were an insignificant part of the Company’s business. Refer to Note 17 - Non-Controlling Interests for additional information.
| 7. ACQUISITIONS |
2021 Business Combinations and Asset Acquisitions
The Company had the following acquisitions during the year ended December 31, 2021: (i) Nature’s Remedy; (ii) OSD; (iii) OhiGrow; and (iv) Grover Beach (all defined below).
| F-49 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
The following table summarizes the purchase price allocations as of their respective acquisition dates:
| Business Combinations | Asset Acquisitions | |||||||||||||||||||
| Nature’s Remedy | OSD | OhiGrow | Grover Beach | Total | ||||||||||||||||
| Assets Acquired: | ||||||||||||||||||||
| Cash and cash equivalents | $ | 3,195 | $ | 259 | $ | — | $ | — | $ | 3,454 | ||||||||||
| Prepaids | 325 | 53 | — | — | 378 | |||||||||||||||
| Accounts receivable, net | 263 | — | — | — | 263 | |||||||||||||||
| Inventory | 15,882 | 184 | — | — | 16,066 | |||||||||||||||
| Indemnification assets (1) | 1,322 | 1,411 | — | — | 2,733 | |||||||||||||||
| Property, plant and equipment | 19,470 | — | 3,165 | 269 | 22,904 | |||||||||||||||
| Right-of-use assets - finance leases | 27,305 | — | — | 2,050 | 29,355 | |||||||||||||||
| Right-of-use assets - operating leases | 1,337 | 1,859 | — | — | 3,196 | |||||||||||||||
| Intangible assets - license (2) | 46,000 | 2,160 | 1,817 | 3,654 | 53,631 | |||||||||||||||
| Intangible assets - tradenames (2) | 4,400 | — | — | — | 4,400 | |||||||||||||||
| Intangible assets - customer database (2) | 2,100 | — | — | — | 2,100 | |||||||||||||||
| Deposits | 20 | 6 | — | 19 | 45 | |||||||||||||||
| Total assets acquired | $ | 121,619 | $ | 5,932 | $ | 4,982 | $ | 5,992 | $ | 138,525 | ||||||||||
| Liabilities Assumed: | ||||||||||||||||||||
| Accounts payable and accrued liabilities | $ | 7,004 | $ | 1,601 | $ | — | $ | — | $ | 8,605 | ||||||||||
| Finance lease obligations | 27,052 | — | — | 2,032 | $ | 29,084 | ||||||||||||||
| Operating lease obligations | 1,267 | 1,859 | — | — | $ | 3,126 | ||||||||||||||
| Deferred tax liabilities | 19,876 | 648 | — | — | $ | 20,524 | ||||||||||||||
| Total liabilities | $ | 55,199 | $ | 4,108 | $ | — | $ | 2,032 | $ | 61,339 | ||||||||||
| Net assets acquired | $ | 66,420 | $ | 1,824 | $ | 4,982 | $ | 3,960 | $ | 77,186 | ||||||||||
| Goodwill | 33,178 | 2,432 | — | — | 35,610 | |||||||||||||||
| Total | $ | 99,598 | $ | 4,256 | $ | 4,982 | $ | 3,960 | $ | 112,796 | ||||||||||
| Consideration: | ||||||||||||||||||||
| Consideration paid in cash, as adjusted for working capital adjustments | $ | 40,360 | $ | 1,827 | $ | 4,949 | $ | 3,592 | $ | 50,728 | ||||||||||
| Consideration paid in promissory notes (fair value) (3) | 15,345 | 2,429 | — | — | $ | 17,774 | ||||||||||||||
| Consideration paid in shares | 35,670 | — | — | 368 | $ | 36,038 | ||||||||||||||
| Contingent consideration | 8,223 | — | — | $ | — | $ | 8,223 | |||||||||||||
| Capitalized costs | — | — | 33 | $ | — | $ | 33 | |||||||||||||
| Fair value of consideration | $ | 99,598 | $ | 4,256 | $ | 4,982 | $ | 3,960 | $ | 112,796 | ||||||||||
| (1) | As part of the OSD and Nature’s Remedy acquisition agreements, the sellers contractually agreed to indemnify the Company for certain amounts that may become payable, including for taxes that relate to periods prior to the date of acquisition. Accordingly, the Company recorded indemnification assets and corresponding estimated accrued tax liabilities, at fair value, for a total of $2,733 as of the dates of the acquisitions. The range of total estimated potential indemnification assets is from $0 to $6,322; however, there is no limit on the Nature’s Remedy indemnification asset. Additional subsequent changes in the amounts recognized for the indemnification assets may occur in relation to the provision for the corresponding tax liabilities, according to changes in the range of outcomes or the assumptions used to develop the estimates of the liabilities at the time of the acquisition. |
| (2) | The licenses acquired have indefinite useful lives. The customer relationships have a useful life of 15 years and the tradenames have a useful life of 5 years. |
| (3) | Refer to “Promissory Notes Payable” in Note 11 - Debt for details on the seller notes. |
| F-50 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
2021 Business Combinations
Nature’s Remedy
On September 10, 2021, the Company acquired 100% of the equity of Nature’s Remedy of Massachusetts, Inc. pursuant to a merger and certain of its affiliates pursuant to member interest purchases (collectively, “Nature’s Remedy”), for upfront consideration comprised of cash, net of working capital adjustments, 8,700,000 subordinate voting shares (with a grant date fair value of $4.10 each), an $11,500 unsecured three-year note and a $5,000 unsecured five-year note. Refer to “Promissory Notes Payable” in Note 11 - Debt for details on the seller notes.
Nature’s Remedy is a vertically integrated single state operator in Massachusetts and currently operates two retail dispensaries, in Millbury, MA and Tyngsborough, MA, and a 50,000 sq. ft. cultivation and production facility in Lakeville, MA. The goodwill is not tax deductible.
The Company also agreed to issue a $5,000 increase to the principal balance of the three-year note and up to an additional $5,000 in Company Subordinate Voting Shares (“SVS”) upon the occurrence or non-occurrence of certain events after the closing date. The payment of the contingent consideration depends on whether or not a competitor opens a competing dispensary within a certain radius of the Company’s dispensary in the Town of Tyngsborough, MA during the first 12 months following the acquisition (The “First Milestone Period”) or during the 18 months following the end of the First Milestone Period. As of the date of acquisition, the Company recognized a contingent consideration liability of $8,223, a Level 3 measurement amount (refer to Note 23 - Financial Instruments), which was based on the weighted-average probability of the potential outcomes. The estimated range of such additional consideration is between $0 and $10,800 (which also includes the interest on the additional principal for the three-year note). Contingent consideration is measured at its acquisition-date fair value and included as part of the consideration transferred for the business combination. Contingent consideration that is classified as a liability is remeasured at subsequent reporting dates with the corresponding gain or loss being recognized in the consolidated statements of operations and comprehensive income (loss). There was no change in the fair value of the contingent consideration from the date of acquisition through December 31, 2021.
OSD
On April 30, 2021, the Company acquired 100% of the equity of Organic Solutions of the Desert, LLC (“OSD”), an operating
dispensary located in Palm Springs, California, for consideration comprised of cash, as adjusted for working capital adjustments, and
$3,100 principal amount of promissory notes. Refer to “Promissory Notes Payable” in Note 11 - Debt for details on the seller
notes. The goodwill is not tax deductible.
Preliminary Purchase Price Allocations for Business Combinations
The consideration has been allocated to the estimated fair values of the assets acquired and liabilities assumed at the dates of the acquisitions and remain preliminary as of December 31, 2021. These estimated fair values involve significant judgement and estimates. The primary areas of judgement involved are the valuation of the intangible assets acquired, which requires management to estimate value based on future cash flows from these assets. The primary areas of the preliminary purchase price allocations that are not yet finalized relate to: intangible assets acquired, property, plant and equipment, indemnification assets, contingent consideration, deferred tax liabilities, and residual goodwill. The Company expects to continue to obtain information to assist in determining the fair value of the net assets acquired as of the respective acquisition dates during the measurement period.
| F-51 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
2021 Asset Acquisitions
The Company determined that the OhiGrow and Grover Beach acquisitions described below did not qualify as business combinations because, for OhiGrow, the assets acquired did not constitute a business, and for Grover Beach, under the concentration test, substantially all of the fair value of the acquisition is concentrated in a single identifiable asset – the license.
OhiGrow
In July 2021, the Company acquired OhiGrow, LLC, a licensed cultivator in Ohio, and Ohio Green Grow LLC (collectively, “OhiGrow”), inclusive of an approximately 10,000 sq. ft. facility and 1.35 acres of land.
Grover Beach
On March 4, 2021, the Company closed on the acquisition of approximately 78% of the equity of a retail license holder located in Grover Beach, California (“Grover Beach”) for $3,592 in cash, as adjusted for working capital adjustments, and 49,348 SVS at a fair value of $7.46 per share, with the rights to acquire the remaining equity for $1 in the future.
2020 Business Combinations and Asset Acquisitions
The Company had the following acquisitions during the year ended December 31, 2020: (i) PAMS; (ii) PADS; (iii) BHILH; (iv) GSG Santa Barbara; and (v) Agape (all defined below).
The following table summarizes the purchase price allocations for the acquisitions completed during the year ended December 31, 2020, as of their respective acquisition dates:
| Business Combinations | Asset Acquisitions | |||||||||||||||||||||||
| PAMS | PADS | BHILH | Agape | GSG Santa Barbara | Total | |||||||||||||||||||
| Cash and cash equivalents | $ | 118 | $ | 971 | $ | 13 | $ | — | $ | — | $ | 1,102 | ||||||||||||
| Prepaid expenses and other current assets | 214 | 5 | 84 | 10 | — | 313 | ||||||||||||||||||
| Accounts receivable | 407 | — | — | — | — | 407 | ||||||||||||||||||
| Inventory | 4,251 | 192 | 100 | — | — | 4,543 | ||||||||||||||||||
| Property, plant and equipment | 579 | 1,075 | 465 | — | — | 2,119 | ||||||||||||||||||
| Right of use assets - finance leases | 15,017 | 234 | 466 | 15,717 | ||||||||||||||||||||
| Right of use assets - operating leases | — | 310 | 877 | — | — | 1,187 | ||||||||||||||||||
| Intangible assets - license(s) (1) | 19,189 | 4,182 | 8,500 | 7,881 | 5,328 | 45,080 | ||||||||||||||||||
| Intangible assets - patient/customer database (1) | 425 | — | — | — | — | 425 | ||||||||||||||||||
| Deposits | 540 | 15 | — | — | — | 555 | ||||||||||||||||||
| Total assets | $ | 40,740 | $ | 6,984 | $ | 10,505 | $ | 7,891 | $ | 5,328 | $ | 71,448 | ||||||||||||
| Accounts payable, accrued expenses and other current liabilities | $ | 335 | $ | 156 | $ | 585 | $ | — | $ | — | $ | 1,076 | ||||||||||||
| Note payable | — | — | — | 90 | — | 90 | ||||||||||||||||||
| Finance lease obligations | 17,013 | 230 | 465 | — | — | 17,708 | ||||||||||||||||||
| F-52 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| Business Combinations | Asset Acquisitions | |||||||||||||||||||||||
| PAMS | PADS | BHILH | Agape | GSG Santa Barbara | Total | |||||||||||||||||||
| Operating lease obligations | — | 310 | 877 | — | — | 1,187 | ||||||||||||||||||
| Deferred tax liabilities | 1,410 | — | — | — | — | 1,410 | ||||||||||||||||||
| Total liabilities | $ | 18,758 | $ | 696 | $ | 1,927 | $ | 90 | $ | — | $ | 21,471 | ||||||||||||
| Net assets acquired | $ | 21,982 | $ | 6,288 | $ | 8,578 | $ | 7,801 | $ | 5,328 | $ | 49,977 | ||||||||||||
| Non-controlling interests | — | — | (4,661 | ) | (1,560 | ) | — | (6,221 | ) | |||||||||||||||
| Total net assets acquired net of non-controlling interest | $ | 21,982 | $ | 6,288 | $ | 3,917 | $ | 6,241 | $ | 5,328 | $ | 43,756 | ||||||||||||
| Consideration paid in cash, as adjusted for working capital adjustments (2) | $ | 15,054 | $ | 5,671 | $ | 2,692 | $ | 3,050 | $ | 4,900 | $ | 31,367 | ||||||||||||
| Capitalized acquisition costs | — | — | — | 191 | 428 | 619 | ||||||||||||||||||
| Fair value of PADS purchase option | — | 1,992 | — | — | — | 1,992 | ||||||||||||||||||
| Consideration paid in 10% senior notes (3) | — | — | — | 1,476 | — | 1,476 | ||||||||||||||||||
| Consideration paid in warrants (3) | — | — | — | 524 | — | 524 | ||||||||||||||||||
| Consideration paid in promissory notes (net of discount) (4) | 2,658 | — | — | — | — | 2,658 | ||||||||||||||||||
| Assumption of Beacon Notes and accrued interest | — | — | 9,555 | — | — | 9,555 | ||||||||||||||||||
| Net effect of other related transactions | — | — | (15,740 | ) | — | — | (15,740 | ) | ||||||||||||||||
| Consideration paid in shares | — | — | — | 1,000 | — | 1,000 | ||||||||||||||||||
| Fair value of consideration | $ | 17,712 | $ | 7,663 | $ | (3,493 | ) | $ | 6,241 | $ | 5,328 | $ | 33,451 | |||||||||||
| Goodwill | $ | — | $ | 1,375 | $ | — | $ | — | $ | — | $ | 1,375 | ||||||||||||
| Bargain purchase on business combination (5) | 4,270 | — | 7,410 | — | — | 11,680 | ||||||||||||||||||
| Total | $ | 21,982 | $ | 6,288 | $ | 3,917 | $ | 6,241 | $ | 5,328 | $ | 43,756 | ||||||||||||
| (1) | The licenses acquired have indefinite useful lives. The patient/customer related intangible assets have estimated useful lives of 0.25 - 5 years. |
| (2) | Cash paid for acquisitions includes $2,320 that was paid during prior years and was previously included in deferred acquisition costs as of December 31, 2019. |
| (3) | The consideration for the Agape acquisition included 10% senior notes amounting to $2,000 principal, and related warrants to purchase 0.6 million Subordinate Voting Shares with a $1.25 strike price; and 769,231 Subordinate Voting Shares at a closing date market price of $1.30 per share. Refer to “Senior Notes” in Note 11 - Debt and to Note 13 - Derivative Liabilities for additional details on the 10% senior secured notes and warrants. |
| (4) | Refer to “Promissory Notes Payable” in Note 11 - Debt for details on the seller note. |
| (5) | The bargain purchase gain for BHILH was reduced by asset disposal and other charges of $1,531 to arrive at the total net gain on business combination. The net gains on business combinations are included in other income (expense), net in the consolidated statements of operations and comprehensive income (loss). The asset disposal and other adjustments, were comprised of net write-offs/impairments of $1,681 reflecting the excess of the carrying amounts over the estimated fair values of intangible assets returned to TGS (included as other consideration in the TGS Transaction), offset by other adjustments of $150. |
| F-53 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
2020 Business Combinations
PAMS
On August 11, 2020, the Company closed on the acquisition of 100% of the equity of Pennsylvania Medical Solutions, LLC, a Pennsylvania grower-processor (“PAMS”). The acquisition increased the Company’s presence in Pennsylvania and expanded its supply of cannabis to the BEYOND/HELLOTM retail stores. The estimated fair value of the license was determined using the multi-period excess earnings method under the income approach based on projections extended to 2025 assuming a 3% long-term revenue growth rate. The estimated fair value of the customer relationships was determined using the with-and-without method. The Company purchased PAMS at a favorable price due to the seller’s financial condition and limited sources of funding, which resulted in a bargain purchase gain which is reflected in other income (expense), net in the consolidated statements of operations and comprehensive income (loss).
Prior to closing, in July 2020, the Company entered into an agreement to loan $3,000 to the previous owner of PAMS, which was secured by a first priority lien on, and continuing security interest in Pennsylvania Dispensary Solutions, LLC (“PADS”). The loan bore interest at 12% and the $3,000 was included at closing in the cash purchase price.
PADS
At the time of the PAMS acquisition, as part of the agreements with Vireo Health, Inc (“Vireo”), the seller of PAMS, Jushi received an assignable purchase option (“PADS Purchase Option”) to acquire 100% of the equity of PADS. The PADS Purchase Option had an expiration date of 18 months from closing, and was subject to certain customary closing conditions, including approvals from all applicable regulatory authorities. On November 13, 2020, the Company exercised the PADS Purchase Option and on December 18, 2020, the Company closed on the purchase of 100% of the equity interests of PADS from Vireo.
PADS increased the Company’s retail footprint in Pennsylvania, where PADS is a medical marijuana dispensary permit holder with two dispensaries operating in Bethlehem and Scranton. The goodwill recognized from the acquisition is attributable to synergies expected from integrating PADS into the Company’s existing business. The goodwill acquired is not deductible for tax purposes.
The cash consideration allocated to the PADS Purchase Option was $1,553 based on the relative fair values of the net assets acquired in the PAMS acquisition and the PADS Purchase Option as of August 11, 2020. The difference between the purchase price allocated to the PADS Purchase Option and its fair value at the time of its exercise was a gain of $440 and is included in other income (expense), net in the consolidated statements of operations and comprehensive income (loss) for the year ended December 31, 2020. The fair value of the PADS Purchase Option was estimated using a Black Scholes option pricing model which included the following assumptions:
| December 18, 2020 | ||||
| Fair value of PADS Purchase Option | $ | 1,992 | ||
| Strike price | $ | 5,000 | ||
| Spot price (estimated enterprise value of PADS) | $ | 5,158 | ||
| Risk-free annual interest rate | 0.16 | % | ||
| Volatility | 80 | % | ||
| Estimated term | 1.5 years | |||
The estimated enterprise value of PADS was estimated using a 5-year discounted cash flow analysis using a cost of capital of 29%, and a terminal value at the end of the explicit projection period using the Gordon Growth Model and is considered to be a Level 3 measurement.
| F-54 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
The estimated fair value of the PADS license was determined using the multi-period excess earnings method under the income approach based on projections extended to 2030 assuming a 3% long-term revenue growth rate.
TGS Transaction - Beyond Hello IL Holdings, LLC (“BHILH”) (f/k/a TGS Illinois Holdings LLC (“TGSIH”))
On January 29, 2020, the Company acquired an approximately 75% interest in TGS Illinois Holdings LLC (currently known as Beyond Hello IL Holdings, LLC) (the “TGS Transaction”), and became the owner of two medical cannabis dispensaries in Illinois - one in Sauget, and one in Normal, with each dispensary eligible to seek approval from the Illinois Department of Financial & Professional Regulation to open a second retail location. On April 22, 2020, the names of TGS Illinois Holdings LLC and TGS Illinois, LLC, were changed to Beyond Hello IL Holdings, LLC, and Beyond Hello IL, LLC, respectively. Each change was approved by the State of Illinois.
Due to legalization of adult-use cannabis in the state of Illinois which became effective January 1, 2020, the purchase of BHILH resulted in a bargain purchase gain. The bargain purchase gain is reflected in other income (expense), net in the consolidated statements of operations and comprehensive income (loss).
The estimated fair value of the licenses acquired was determined using the multi-period excess earnings under the income method based on projections extended to 2025 assuming a 3% long-term revenue growth rate and a discount rate of 12%. The estimated fair value of the customer relationships was determined using the with-and-without method.
The fair value of the non-controlling interests was based on the fair value of the consideration paid to purchase the non-controlling interests (the “TGSIH NCI Transaction”). Refer to Note 17 - Non-Controlling Interests.
The TGSIH acquisition was a part of a series of transactions under a favorable settlement agreement between the Company and its respective affiliates, and The Green Solution and its respective affiliates and their owners (“TGS”). The transactions included: (1) the transfer to the Company of approximately 75% interest in the TGSIH units; (2) the Company’s assumption and/or payoff of approximately $12,000 in TGS debt including interest and expenses relating to the debt (see below); (3) the Company returning its 51% majority stake in TGS National Holdings, LLC (“TGS National”) to TGS and terminating the 2018 purchase agreement for TGS National which included certain restrictive covenants, employment agreements and exclusive intellectual property licenses in Jushi’s favor; (4) the return to the Company and cancellation of the 5,000,000 Subordinate Voting Shares of Jushi Holdings, Inc. and warrants with an exercise price of $2.00 to purchase 2,500,000 Subordinate Voting Shares of Jushi Holdings, Inc. which were issued in connection with the 2018 purchase of TGS National; and (5) the transfer to Jushi Inc of 416,060 common shares of Organigram, Inc. (“OGI”) and cash from the liquidation of certain options to purchase common shares of OGI.
The approximately $12,000 in TGS debt noted above includes approximately $2,442 of debt payable to a third party and interest paid off by the Company, which was included in the cash paid at closing, and $9,555 of debt which was negotiated with the holder to be exchanged for Jushi Holdings Inc. Senior Notes and warrants. The $9,555 was comprised of secured notes of an affiliate of TGS, Beacon Holding, LLC (the “Beacon Notes”), plus unpaid accrued interest. The carrying amount of the debt and unpaid accrued interest approximated the fair value. Refer to “Senior Notes” in Note 11 - Debt for further details on the Company’s Senior Notes and the related warrants.
| F-55 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
The net effect of other related transactions from the TGS Transaction, as reflected in the purchase price allocation table above, was comprised of the following:
| Redemption Liability cancelled | $ | 8,440 | ||
| 5,000,000 Subordinate Voting Shares of Jushi Holdings, Inc. and warrants with an exercise price of $2.00 to purchase 2,500,000 Subordinate Voting Shares of Jushi Holdings, Inc. returned to Jushi | 7,075 | |||
| 416,060 common shares of OGI | 1,092 | |||
| Payment to Jushi for the OGI options that were liquidated | 478 | |||
| TGS National fair value of intangibles, net of other items returned and derecognized (1)(2) | (918 | ) | ||
| TGS National cash given up | (427 | ) | ||
| Net effect of other related transactions | $ | 15,740 |
| (1) | The difference of $1,748, net of other adjustments of $217, between the carrying amount of the disposed intangibles of $2,666 (refer to Note 8 - Goodwill and Other Intangible Assets) and the fair value above of $918 was a total of $1,531 and is included in net gains on business combinations in the consolidated statements of operations and comprehensive income (loss). |
| (2) | No goodwill associated with TGS National was written off as a result of this transaction in 2020, since the Company had previously recognized an impairment loss of $8,990 in 2018 related to the total goodwill associated with the original acquisition of TGS National. |
The fair values of the Jushi Holdings, Inc. shares and OGI shares were based on the closing market price as of the date of the transaction and the fair value of the Jushi Holdings, Inc. warrants was calculated using a Black-Scholes model with the following assumptions: stock price of $1.28 (market price); exercise price of $2.00; estimated term 1.35 years; volatility 76%; risk-free rate 1.46%. The fair value of the Jushi securities was accounted for as a reduction to equity. The TGS National net assets returned consisted primarily of cash and intangibles. The fair value of the intangibles returned was calculated using a discounted cash flow model using a discount rate of 12%, a growth rate of zero, and estimated useful lives ranging from 9 - 11 years.
Redemption Liability
At the time of the original acquisition of 51% of TGS National by the Company in 2018, the Company had the exclusive right to purchase the remaining 49% of TGS National for a period of 30 months from the Closing Date (the “Option Period”). The consideration to be paid for this purchase option (the “Redemption Liability”) was $8,500. As a result of the TGS Transaction, the Redemption Liability was cancelled.
The Redemption Liability was initially recorded at fair value and was estimated using the present value of the estimated future purchase price and was therefore considered to be a Level 3 measurement. The adjusted present value of the Redemption Liability was $8,440 as of the date of the TGS Transaction in 2020. The total change in the redemption liability for the year ended December 31, 2020 was insignificant.
2020 Asset Acquisitions
The Company determined that The GSC Santa Barbara and Agape acquisitions below did not qualify as business combinations under ASC 805 Business Combinations, because the assets acquired did not constitute a business, as substantially all of the fair value of each of the acquisitions is concentrated in a single identifiable asset – the license.
GSG Santa Barbara
On July 24, 2020, the Company closed on the acquisition of 100% of the equity of GSG SBCA, Inc. (“GSG Santa Barbara”), a non-operating provisionally licensed Santa Barbara, California dispensary operator for a total purchase price of $4,900 in cash, of which $2,250 was paid in the prior year. The GSG Santa Barbara dispensary commenced retail operations on October 14, 2020.
| F-56 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Agape
On June 25, 2020, the Company closed on the acquisition of 80% of the economic and voting interests in Agape Total Health Care Inc (“Agape”), a Pennsylvania Dispensary Permittee, for consideration in cash and shares. Agape has opened and operates three retail locations in Pennsylvania: one in Philadelphia, one in Reading and one in Pottsville.
Business Combinations - Acquisition and Deal Costs
For the years ended December 31, 2021 and 2020 acquisition and deal costs relating to business combinations totaled $350 and $502 and are included within the selling, general and administrative expenses in the consolidated statements of operations and comprehensive income (loss). The remaining acquisition and deal costs, which are included in operating expenses (refer to Note 19 - Operating Expenses), were incurred either for acquisitions not completed or not expected to be completed.
Business Combinations Acquisition Results and Unaudited Supplemental Pro Forma Financial Information
The following table summarizes consolidated proforma revenue and consolidated proforma net income (loss) as if the 2021 business combinations had occurred at the beginning of the year prior to their actual acquisition:
| Year Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Revenue | $ | 239,937 | $ | 114,622 | ||||
| Net income (loss) | $ | 12,823 | $ | (223,827 | ) | |||
These unaudited pro forma financial results do not purport to be indicative of the actual results that would have been achieved by the combined companies for the years indicated, or of the results that may be achieved by the combined companies in the future. These amounts have been calculated using actual results and adding pre-acquisition results, after adjusting for: acquisition costs, additional depreciation and amortization from acquired property, plant and equipment and intangible assets, as well as adjustments for incremental interest expense relating to consideration paid, and changes to conform to the Company’s accounting policies.
The following table summarizes revenue and net (loss) income for the 2021 acquisitions from the respective acquisition dates through December 31, 2021:
| Nature’s Remedy | OSD | Total | ||||||||||
| Revenue | $ | 13,870 | $ | 1,237 | $ | 15,107 | ||||||
| Net (loss) income | $ | 2,334 | $ | (503 | ) | $ | 1,831 | |||||
| F-57 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
The following table summarizes revenue and net (loss) income for the 2020 acquisitions from the respective acquisition dates through December 31, 2020:
| PAMS | PADS | BHILH | Total | |||||||||||||
| Revenue | $ | 3,048 | $ | 113 | $ | 33,203 | $ | 36,364 | ||||||||
| Net (loss) income | $ | (188 | ) | $ | (85 | ) | $ | 7,940 | $ | 7,667 | ||||||
The above results are after the elimination of intercompany transactions.
| 8. GOODWILL AND OTHER INTANGIBLE ASSETS |
Goodwill
The changes in the carrying amount of goodwill are as follows:
| Goodwill, carrying amount, as of January 1, 2020 | $ | 17,888 | ||
| Additions from new business combinations | 1,375 | |||
| Impairment of goodwill - MEND | (170 | ) | ||
| Goodwill, carrying amount, as of December 31, 2020 | $ | 19,093 | ||
| Additions from new business combinations | 35,610 | |||
| Impairment of goodwill - FBS Nevada | (1,783 | ) | ||
| Goodwill, carrying amount, as of December 31, 2021 | $ | 52,920 |
Goodwill Impairments
During the year ended December 31, 2021, management determined that the lower than expected operating results of the Company’s Nevada operations was an indicator of impairment. The Company utilized the discounted cash flow method for its impairment test, and as a result, recorded a goodwill impairment charge of $1,783. The key inputs and assumptions used in the fair valuation of Nevada include: (i) a five-year cash flow forecast, which is based on the Company’s actual operating results and business plans; (ii) a perpetual growth rate of 3%; and (iii) an estimated discount rate of 16.5%. The goodwill impairment is recorded within operating expenses in the consolidated statements of operations and comprehensive income (loss).
During the year ended December 31, 2020, the Company entered into a confidential settlement agreement with certain parties, including the former owner of Medicinal Excellence for Neurological Disorders, LLC (“MEND”), a business acquired by the Company in 2018. As a result, the Company recognized a loss on settlement of $2,217 (including the write-off of certain intangibles - refer to Other Intangible Assets below) and wrote-off goodwill of $170. The loss on settlement and the goodwill impairment are both recorded in other income (expense), net in the consolidated statements of operations and comprehensive income (loss).
| F-58 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Other Intangible Assets
The components of other intangible assets are as follows:
| December 31, 2021 | December 31, 2020 | Estimated Useful Life | ||||||||
| Licenses | $ | 164,662 | $ | 111,030 | Indefinite | |||||
| Intellectual Property | 9,580 | 9,580 | 10 years | |||||||
| Tradenames | 10,200 | 5,800 | 1 - 15 years | |||||||
| Patient/Customer Database | 2,795 | 695 | 5 - 10 years | |||||||
| Non-compete | 155 | 155 | 3 years | |||||||
| Website development | 61 | 61 | 3 years | |||||||
| Formulations | 50 | 50 | Indefinite | |||||||
| Total gross amount | $ | 187,503 | $ | 127,371 | ||||||
| Less: Accumulated Amortization | (5,037 | ) | (3,059 | ) | ||||||
| Other Intangible Assets, net | $ | 182,466 | $ | 124,312 | ||||||
Amortization expense for the years ended December 31, 2021 and 2020 was $1,977 and $2,202, respectively, and is included in operating expenses in the consolidated statements of operations and comprehensive income (loss).
For the years ended December 31, 2021 and 2020, all additions to intangible assets were from acquisitions. For the year ended December 31, 2020, the Company had net disposals of intangible assets of $2,666 as a result of the TGS Transaction. Refer to Note 7 - Acquisitions. Additionally, during the year ended December 31, 2020, the Company recorded a net disposal of patient databases of $771, which is included in the loss on settlement discussed in Goodwill Impairment above.
The estimated future annual amortization expense related to intangible assets as of December 31, 2021 is as follows:
| 2022 | $ | 2,653 | |||
| 2023 | 2,626 | ||||
| 2024 | 2,604 | ||||
| 2025 | 2,572 | ||||
| 2026 | 2,572 | ||||
| Thereafter | 4,727 | ||||
| Total estimated future amortization expense | $ | 17,754 |
| F-59 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| 9. OTHER NON-CURRENT ASSETS |
The components of other non-current assets are as follows:
| December 31, 2021 | December 31, 2020 | |||||||
| Operating lease assets | $ | 17,288 | $ | 6,075 | ||||
| Deposits - equipment | 4,315 | 768 | ||||||
| Deposits and escrows - properties | 1,343 | 1,630 | ||||||
| Indemnification assets | 2,733 | — | ||||||
| Equity investment | 1,500 | 1,500 | ||||||
| Employee receivables and accrued interest (Refer to Note 21 - Related Party Transactions) | — | 2,470 | ||||||
| Net deferred tax assets (Refer to Note 16 – Income Taxes) | — | 249 | ||||||
| Other | 407 | 224 | ||||||
| Total other non-current assets | $ | 27,586 | $ | 12,916 | ||||
Equity Investment
In December 2020, the Company entered into a lease agreement with a lessor, PV Culver City, LLC, formerly known as Pacific Oakmark TBird, (“PVLLC”). In connection with the lease, and to assist PVLLC in the purchase of the leased property from a third party, the Company contributed $1,500 to PVLLC in exchange for Class C Member shares, or a 23.08% ownership interest. The Company does not have significant influence over, and the Company does not have the right to vote or participate in the management of the PVLLC and therefore the investment is measured at its fair value. The fair value as of December 31, 2021 approximated the initial fair value as there was no change to the business from the date of acquisition through December 31, 2021.
| 10. ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES |
The components of accrued expenses and other current liabilities are as follows:
| December 31, 2021 | December 31, 2020 | |||||||
| Accrued capital expenditures | $ | 17,599 | $ | 2,177 | ||||
| Goods received not invoiced | 8,007 | 2,625 | ||||||
| Accrued employee related expenses and liabilities | 6,062 | 2,303 | ||||||
| Accrued professional and management fees | 5,139 | 638 | ||||||
| Accrued sales and excise taxes | 2,535 | 1,399 | ||||||
| Accrued interest | 1,181 | 45 | ||||||
| Deferred revenue (loyalty program) | 1,427 | 935 | ||||||
| Operating lease obligations - current portion | 2,745 | 314 | ||||||
| Other accrued expenses and current liabilities | 3,277 | 920 | ||||||
| Total | $ | 47,972 | $ | 11,356 | ||||
| F-60 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| 11. DEBT |
The components of the Company’s debt are as follows:
| Year Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Principal amounts: | ||||||||
| 10% Senior Notes | $ | 75,193 | $ | 83,327 | ||||
| Acquisition Facility | 40,000 | — | ||||||
| Acquisition-related promissory notes payable | 25,767 | 7,782 | ||||||
| Other debt | 11,728 | 3,765 | ||||||
| Total debt - principal amounts | $ | 152,688 | $ | 94,874 | ||||
| Less: debt issuance costs and original issue discounts | (23,536 | ) | (36,124 | ) | ||||
| Total debt - carrying amounts | $ | 129,152 | $ | 58,750 | ||||
| Debt - current portion | $ | 6,181 | $ | 1,659 | ||||
| Debt - non-current portion | $ | 122,971 | $ | 57,091 | ||||
As of December 31, 2021, aggregate future contractual maturities of the Company’s debt are as follows:
| 2022 | 2023 | 2024 | 2025 | 2026 | Thereafter | Total | ||||||||||||||||||||||
| Senior Notes | $ | — | $ | 75,193 | $ | — | $ | — | $ | — | $ | — | $ | 75,193 | ||||||||||||||
| Acquisition Facility | — | — | 3,000 | 4,000 | 33,000 | — | 40,000 | |||||||||||||||||||||
| Acquisition-related promissory notes payable (1) | 2,417 | — | 15,250 | — | 5,000 | 3,100 | 25,767 | |||||||||||||||||||||
| Other debt | 3,764 | 156 | 147 | 107 | 113 | 7,441 | 11,728 | |||||||||||||||||||||
| Total | $ | 6,181 | $ | 75,349 | $ | 18,397 | $ | 4,107 | $ | 38,113 | $ | 10,541 | $ | 152,688 | ||||||||||||||
(1) The Promissory Note that matures in 2022 is a mandatorily convertible note that will be settled in 910,000 shares. Refer to Promissory Notes - Dalitso under SENIOR NOTES below.
Interest expense, net is comprised of the following:
| Year Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Interest and accretion - 10% Senior Notes | $ | 19,257 | $ | 12,095 | ||||
| Interest - finance lease liabilities | 9,158 | 2,451 | ||||||
| Interest and accretion - promissory notes | 1,802 | 1,903 | ||||||
| Interest and accretion - Acquisition Facility | 1,106 | — | ||||||
| Interest and accretion - other debt | 507 | 189 | ||||||
| Capitalized interest | (977 | ) | $ | (1,074 | ) | |||
| Total interest expense | $ | 30,853 | $ | 15,564 | ||||
| Interest income | $ | (243 | ) | $ | (231 | ) | ||
| Total interest expense, net | $ | 30,610 | $ | 15,333 | ||||
| F-61 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
SENIOR NOTES
A summary of the carrying amounts of the Company’s 10% senior notes due January 15, 2023 (“Senior Notes”) is as follows:
| December 31, 2021 | December 31, 2020 | |||||||
| Senior Notes - public (1) | $ | 68,218 | $ | 76,352 | ||||
| Senior Notes – private | 6,975 | 6,975 | ||||||
| Senior Notes - principal amount | $ | 75,193 | $ | 83,327 | ||||
| Less: amounts to accrete (from original issue discount and bifurcation of warrants) | (18,995 | ) | (34,133 | ) | ||||
| Less: deferred transaction costs, net | — | (317 | ) | |||||
| Total Senior Notes - carrying amount | $ | 56,198 | $ | 48,877 | ||||
| (1) | The public notes are listed under the symbol “JUSH.DB.U” and were deemed to be tradable at approximately the principal amount as of December 31, 2021 and 2020. |
The Senior Notes bear interest at 10% per annum, payable in cash quarterly on March 31, June 30, September 30 and December 31 of each year to, but excluding, the maturity date of the Notes, which is January 15, 2023. The notes are classified as non-current liabilities in the consolidated balance sheets.
Issuances of Senior Notes
The Senior Notes were originally issued between December 2019 and July 2020. During the first quarter of 2020, the Company closed on the first tranche of the Company’s debt offering (a private placement) announced in December 2019 (“Tranche 1”) and then also closed on the second tranche of the Company’s debt offering (a private placement), in June and July of 2020 (“Tranche 2”).
There were two financing structures offered in connection with the Tranche 1 offering. The first structure was comprised of Senior Notes which were issued with warrants to acquire Subordinate Voting Shares of the Company (“Warrant Notes”) at 75% coverage of the principal amount divided by the original strike price of the warrants. The warrants were issued by the Company in connection with, but were detached from, the Company’s issuance of the Senior Notes. The original exercise price of these warrants was $1.58 but was subsequently adjusted for a “down-round” to $1.25. Refer to Note 14 - Equity for additional information. The second structure was comprised of Senior Notes with no warrants but issued at a discount of approximately 17% (“OID Notes”). The Tranche 2 offering of the Private Senior Notes was comprised of only Warrant Notes, with warrants at 75% coverage.
The original carrying amounts of the Warrant Notes were calculated as the total fair value (cash amount received) less amounts bifurcated and allocated to the warrants. The initial total value of the warrants that were issued with senior notes during Tranche 1 and Tranche 2 was $35,774. Refer to Note 14 - Equity for details of the methods and assumptions used in calculating the fair values of the warrants. The OID Notes were initially recorded at the cash amount received. The carrying amount of each of the Senior Notes is being accreted to the principal amount using the effective interest rate method.
| F-62 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
2020 Modifications
(i) Warrant Notes Exchanges for OID Notes
In January 2020, a certain Tranche 1 holder elected to exchange $500 principal amount of Warrant Notes that had been issued in December 2019 into OID Notes of principal amount $585 in January 2020, and the related 237,537 Warrants that were issued in December 2019 were voided. In addition, Warrant Notes of $5,000 that were issued in January 2020 were subsequently also exchanged for OID Notes of $5,842 in January 2020, and the related 2,375,372 warrants that were issued in connection with the Warrant Notes were also voided. The notes exchanged were determined to have substantially different terms as a result of the cancellation of the Warrants and therefore the exchanges were accounted for as an accounting extinguishment of the original financial liabilities and the recognition of new financial liabilities under ASC 470, Debt. The difference between the fair value of the new OID Notes of $5,500 and the total carrying value of the extinguished Warrants Notes (carrying value of $3,621), and the extinguished Derivative Warrants (carrying value of $1,916) was a gain of $37 and is included in other income (expense), net in the consolidated statements of operations and comprehensive income (loss).
(ii) Down-Round Price Adjustment
The warrants associated with the Tranche 1 Senior Notes contained a down-round price protection feature and were issued at ~$1.58. As a result of the issuance of the Tranche 2 warrants at an exercise price equal to $1.25, the Tranche 1 warrants were adjusted down to an exercise price of $1.25. This resulted in a loss on modification of $1,001 and is included in other income (expense), net in the consolidated statements of operations and comprehensive income (loss).
(iii) “Option 2” Elections
As a result of Most Favored Nation (“MFN”) clauses in certain of the Tranche 1 Senior Notes holders’ offering documents, at the time of the Tranche 2 offering in June 2020, those certain Tranche 1 investors were offered to either retain their existing offering documents, including the Senior Notes, and warrants, if applicable, with an unmodified MFN clause and continued down-round price protection (“Option 1”); or they could elect to exchange their offering documents for the Tranche 2 offering documents with revised terms including an increased number of warrants based on 75% warrant coverage with a strike price of $1.25; but with a substantially narrowed MFN and no down-round price protection going forward (“Option 2”). Certain Tranche 1 Senior Notes holders accepted Option 2 to exchange their Senior Notes and warrants, or their OID Notes, for the new offered terms, with the following results:
OID Notes Exchanges for Warrant Notes
As a result of the selection of Option 2 by certain OID Note Holders, $6,427 principal amount of OID Notes was extinguished and replaced with $5,500 principal amount of Warrant Notes and 3,300,000 warrants. The Company determined that these modifications were substantial under ASC 470-50 Debt - Modification and Extinguishment (“ASC 470-50”) as a result of the change to the MFN, a greater than 10% change to the principal amount of the debt and the addition of warrants, and therefore the exchanges were accounted for as accounting extinguishments of the original financial liabilities and the recognition of new financial liabilities. The difference between the extinguished carrying value of the OID Notes of $5,627, and the total new fair value of $5,500 for the replacement Warrants Notes (fair value of $2,803) and the new Derivative Warrants (fair value of $2,697), was a gain of $127 and is included in other income (expense), net in the consolidated statements of operations and comprehensive income (loss).
Warrant Note Holders
As a result of the election of Option 2 by the Warrant Note holders, the Company issued an additional 2,960,738 warrants. The Company determined that these modifications were substantial under ASC 470-50 as a result of the change to the MFN and the removal of the down-round price protection for the Derivative Warrants, and therefore the exchanges were accounted for as accounting extinguishments of the original financial liabilities and the recognition of new financial liabilities. The difference between the extinguished total carrying value of $25,037 for the existing Warrant Notes (carrying value $15,777) and related Derivative Warrants liabilities (carrying value $9,260), and the total new fair value of $23,700 for the replacement Warrant Notes (fair value of $12,005) and new total Derivative Warrants (fair value of $11,695), was a gain of $1,337 and is included in other income (expense), net in the consolidated statements of operations and comprehensive income (loss).
| F-63 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Subsequent to all Option 2 elections made by the Warrant Notes holders in June and July of 2020, 6,128,459 Derivative Warrants were still subject to the downward price protection.
As a result of the Option 2 extinguishments, the Company expensed previously deferred financing costs of $491 related to Tranche 1 Senior Notes, which is included in other income (expense), net in the consolidated statements of operations and comprehensive income (loss).
(iv) Public Debt Listing
On December 1, 2020, the Company listed for trading on the Canadian Securities Exchange $76,352 of 10%, senior secured notes due January 15, 2023 (the “Public Notes”). The publicly traded senior secured notes were issued to certain existing holders of the Company’s 10% senior secured notes due January 15, 2023 who elected to exchange their then-outstanding non-listed senior secured notes for the new Public Notes in the same principal amount. Other existing holders of the Company’s non-listed 10% senior secured notes due January 15, 2023, representing an aggregate principal amount of $6,975, elected to retain their non-listed/private senior secured notes.
As a result of the public listing of the Senior Notes in December 2020, for the majority of the notes, the Company determined that the modification was not substantial under ASC 470-50. However, there were certain Senior Notes that were modified by removing a different redemption feature, therefore the Company determined that this was a substantial modification under ASC 470-50, and was thus accounted for as an accounting extinguishment of the original financial liabilities and the recognition of new financial liabilities. The difference between the extinguished total carrying value of $8,170 and the total new value of $10,032, was a loss of $1,862 which is included in other income (expense), net in the consolidated statements of operations and comprehensive income (loss).
Senior Notes - Mandatory and Optional Redemptions
Mandatory Redemptions
The Company is required to offer to redeem the Senior Notes using 33% of the net proceeds from any equity offerings by the Company or any of its subsidiaries (including the Guarantors), at a purchase price equal to par plus accrued but unpaid interest (no premiums). The note holder has the option to waive their right to redemption. This mandatory prepayment option (“Prepayment Option”) represents an embedded derivative liability. Refer to Note 13 - Derivative Liabilities for additional information on the Prepayment Option.
Pursuant to these terms of the 10% Senior Notes, in connection with the equity offerings completed during the first quarter of 2021, the Company redeemed a total principal amount of $8,134. The difference between the carrying amounts of the Senior Notes and the principal amount extinguished was $3,815.
| F-64 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Other mandatory redemptions would be required: i) at par plus accrued interest with 100% of the net cash proceeds as result of certain specified dispositions; and/or; ii) 106% of the principal amount plus all accrued interest as a result of certain change of control transactions.
Optional Redemptions
A redemption of the Senior Notes may by initiated by the Company at any time upon 3 days written notice and may redeem all or any portion of the Senior Notes at par plus accrued interest plus a premium equal to 5% of the aggregate principal amount of the Senior Notes being redeemed on or after the first anniversary of the issue date but prior to the second anniversary of the issue date. The Company (at its option) may redeem all or any portion of the Senior Notes at par plus accrued interest (without any premium) on or after the second anniversary of the date of issuance.
Guarantees
The Company’s total obligations under the Senior Notes are secured by substantially all assets of the Company and certain Subsidiaries (subject to certain exclusions) and are guaranteed for non-payment by certain Subsidiaries.
Default Provisions
The Senior Notes agreement provides for customary events of default, as well as customary remedies upon an event of default as defined in the Senior Notes agreement (“Event of Default”), including acceleration of repayment of outstanding amounts. In addition, automatically upon the occurrence and during the continuance of an Event of Default, the interest rate accruing on the outstanding principal amount of the Note shall be 3% more than the rate otherwise payable under the Senior Notes.
Covenants
The Senior Notes are subject to certain customary non-financial provisions and covenants. The covenants, among other things, generally limit the ability of the Company and certain of its subsidiaries, subject to certain exceptions, to (i) incur certain additional debt; (ii) pay dividends or make distributions from certain subsidiaries; (iii) sell certain assets; and (iv) effect certain transactions. As of December 31, 2021 and 2020, the Company was in compliance with the provisions and covenants.
ACQUISITION FACILITY
On October 20, 2021 (the “Closing Date”), the Company entered into definitive documentation in respect of a $100,000 Senior Secured Credit Facility (the “Acquisition Facility”) from Roxbury, LP, a portfolio company of SunStream Bancorp Inc., which is a joint venture sponsored by Sundial Growers Inc. The Company is permitted to borrow amounts under the Acquisition Facility for potential strategic expansion opportunities in both its core and developing markets. After being drawn, loans issued under the Acquisition Facility bear an interest rate of 9.5% per annum, payable quarterly, and will mature five years from the Closing Date. Subject to the approval of the Agent’s investment committee and other conditions, including pro forma compliance with certain financial covenants (further defined below) at the time of borrowing, the Company will be able to make draws under the facility until the 18- month anniversary of the Closing Date (the “Draw Period”), and will have a two-year interest-only period before partial amortization begins on a quarterly basis. The Company also may increase the total commitment of the Acquisition Facility by an aggregate amount of up to $25,000, subject to certain conditions (the “Accordion”). The Acquisition Facility is secured by a first lien over certain Company assets and on a pari passu basis with the collateral securing the indebtedness of the Company evidence by the Senior Notes.
| F-65 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
During the Draw Period, a standby fee of 2.25% per annum of: (1) the undrawn amount of the Acquisition Facility minus the sum of the daily average of the outstanding amount of the Acquisition Facility for the preceding calendar quarter shall be paid quarterly, in arrears, on the first business day of each calendar quarter. The standby fee drops to 1.5% on the date the existing 10% Senior Notes mature or are refinanced. An exit fee of 1.5% of the original term loan amount of $100,000 shall be paid upon the earliest of the maturity date, any repayment of the principal balance of the term loans or the occurrence of an event of default. In the event the existing Senior Notes mature or are refinanced, no exit fee is owed by the Company to the lenders. In the event the Company wishes to refinance the Senior Notes, lenders have a right of first refusal to contribute up to 50% of the amount used to refinance the Senior Notes.
As of December 31, 2021, the Company has drawn down $40,000 from the Acquisition Facility to fund the cash portion of the acquisition of Nature’s Remedy. The original issue discount was $980 and debt issuance costs capitalized were $721, of which $73 has been amortized as of December 31, 2021. The effective interest rate was 10.6% as of December 31, 2021. Beginning April 1, 2024, the Company must repay 10% per annum of the total amount of the term loans funded to date, in quarterly installments on the first business day of each calendar quarter.
The Company will consider borrowing additional amounts in the future under the Acquisition Facility for potential strategic expansion opportunities in both its core and developing markets. As of December 31, 2021, the Company had approximately $60,000 of availability under the Acquisition Facility (excluding the Accordion). Refer to Note 24 - Subsequent Events.
Mandatory and Optional Redemptions
If the Company prepays any of the loans for any reason except for scheduled amortization payments, the Company must also pay a “make-whole premium.” The make-whole premium is the difference between: (1) all fees and interest payments that would be payable if such loan had been outstanding for 27 months and (2) all payments on such loan or portion thereof received by the loan agent prior to the relevant prepayment.
After the Senior Notes mature or are refinanced, the Company is required to repay 100% of net cash proceeds above $5,000 (individually or in the aggregate in any calendar year) from sales of certain assets within 3 business days following the receipt of net cash proceeds.
Covenants
The Acquisition Facility is subject to certain customary non-financial covenants until the Senior Notes mature or are refinanced, including covenants that restrict the Company’s ability to incur or guarantee additional indebtedness or to conduct sales and other dispositions of property or assets. Upon the maturity or refinancing of the Senior Notes, the Acquisition Facility will be subject to additional customary non-financial covenants that, among other things, restrict the Company’s ability to create liens, make investments, consummate acquisitions, mergers, reorganizations or similar transactions, enter into sale-leaseback transactions, prepay subordinated debt, pay dividends or make other payments in respect of capital stock, change the line of business and enter into transactions with affiliates. Upon the occurrence and during the continuance of an event of default (after applicable notice and cure periods) the Acquisition Facility permits the lender to declare all principal, interest and all other obligations immediately due and payable. In addition, the Company is required to comply with certain financial covenants measured on a fiscal quarter-end basis beginning at the earlier of the maturity or refinancing of the Senior Notes as follows:
| F-66 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| • | Total Leverage Ratio, calculated as the ratio of Total Funded Indebtedness to EBITDAR (all such terms are defined in the Acquisition Facility) not to exceed the correlative ratio below: |
| Applicable Ratio | Fiscal Quarter Ending (1) | |
| 6.00 to 1.00 | March 31, 2022 and June 30, 2022 | |
| 5.00 to 1.00 | September 30, 2022 and December 31, 2022 | |
| 4.00 to 1.00 | March 31, 2023 and June 30, 2023 | |
| 3.50 to 1.00 | September 30, 2023, and all fiscal quarters ending thereafter |
| • | Debt Service Coverage Ratio of at least 1.10 to 1.0, calculated as the ratio of Adjusted EBITDA to Debt Service Amounts (all such terms are defined in the Acquisition Facility); and |
| • | Minimum sum of unrestricted cash and cash equivalents of at least 5% of the total loan amount under the Acquisition Facility. |
(1) In April 2022, the Company entered into an amendment to the Acquisition Facility pursuant to which: (i) the commencement of leverage testing was pushed back by four quarters (now beginning March 31, 2023), (ii) certain leverage ratios were revised; and (iii) the Company may proceed with a bankruptcy in Switzerland with respect to Jushi Europe without defaulting under the Acquisition Facility. Refer to Note 17 - Non-Controlling Interests for additional information on Jushi Europe.
As of December 31, 2021, the Company was not subject to any financial covenants and was in compliance with all applicable non-financial covenants.
PROMISSORY NOTES
A summary of the Company’s acquisition-related promissory notes payable is as follows:
| December 31, 2021: | PAMS | Dalitso | OSD | Nature’s Remedy | Total | |||||||||||||||
| Principal amounts of promissory notes | $ | 3,750 | $ | 2,417 | $ | 3,100 | $ | 16,500 | $ | 25,767 | ||||||||||
| Original issuance discount (“OID”) and deferred finance charges, net | (713 | ) | (174 | ) | (596 | ) | (1,056 | ) | (2,539 | ) | ||||||||||
| Beneficial conversion feature, net | — | (272 | ) | — | — | (272 | ) | |||||||||||||
| Carrying amounts as of December 31, 2021 | $ | 3,037 | $ | 1,971 | $ | 2,504 | $ | 15,444 | $ | 22,956 | ||||||||||
| December 31, 2020: | PAMS | FBS Nevada | Dalitso | Total | ||||||||||||
| Principal amounts of promissory notes | $ | 3,750 | $ | 1,500 | $ | 2,532 | $ | 7,782 | ||||||||
| Original issuance discount and deferred finance charges, net | (985 | ) | (2 | ) | (584 | ) | (1,571 | ) | ||||||||
| Beneficial conversion feature, net | — | — | (374 | ) | (374 | ) | ||||||||||
| Carrying amounts as of December 31, 2020 | $ | 2,765 | $ | 1,498 | $ | 1,574 | $ | 5,837 | ||||||||
Accrued interest payable for the promissory notes is included in Accrued expenses and other current liabilities in the consolidated balance sheets. Refer to Note 10 - Accrued Expenses and Other Current Liabilities.
Unsecured Promissory Notes – Nature’s Remedy
In September 2021, in connection with the Nature’s Remedy acquisition, the Company issued a principal amount $11,500, 8% unsecured three-year note maturing September 10, 2024 and a $5,000 8% unsecured five-year note maturing September 10, 2026 to the seller. The effective interest rate on the notes is 9.9%. The promissory notes provide for cash interest payments to be made quarterly and all principal and accrued and unpaid interest are due at their respective maturities. Refer to Note 7 - Acquisitions for additional information on the Nature’s Remedy acquisition and the portion of contingent consideration of $5,000 which would be payable as an addition to the 8% unsecured three-year note.
| F-67 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Secured Promissory Notes – OSD
In April 2021, in connection with the OSD acquisition, the Company issued a principal amount $3,100, 4% secured promissory note to the seller. The promissory note matures on April 30, 2027 and interest is payable quarterly. The effective interest rate on the note is 7.5%. The note is secured by the equity of OSD. Pursuant to the terms of the OSD acquisition, indemnification obligations of the seller may be offset against this promissory note in the future if the Company makes a claim for such indemnification and such right of offset. Refer to Note 7 - Acquisitions.
Unsecured Promissory Notes - PAMS
In August 2020, in connection with the PAMS acquisition, the Company issued an $3,750 8% unsecured promissory note to the seller. The promissory note matures on August 11, 2024 and interest is payable quarterly. The fair value of the promissory note was determined to be $2,658, resulting in a fair value discount on issuance of $1,092, which is being amortized using the effective interest rate of 7.2% over the life of the promissory note. The total effective interest rate on the note is 15.2%.
The initial fair value of the consideration paid in notes was determined using an approach that uses forecast cash flows of the debt instrument according to its contractual terms and then discounts those cash flows to the date of value.
Promissory Notes - Dalitso
During the fourth quarter of 2020, the Company issued a new $2,412 unsecured convertible promissory note (the “Convertible Note”) in connection with the Company acquiring all the remaining 38.24% equity interests of Dalitso (the “Dalitso NCI Transactions”). Refer to Note 17 - Non-Controlling Interests. The convertible note bears interest at 1% per annum and matures on November 19, 2022. The convertible note is classified as a liability based on its legal form, despite the Company’s contractual obligation to settle the note in shares. The entire face value of the convertible note is convertible after the first anniversary into 910,000 Subordinate Voting Shares at a fixed conversion price per share of $2.65. The note will automatically convert at maturity or insolvency, as defined in the note agreement.
The Company determined that the convertible note included a beneficial conversion feature. The beneficial conversion feature is calculated at its intrinsic value (the difference between the conversion price of $2.65 at the date of the note issuance and the fair value of the shares into which the debt is convertible at the commitment date of $3.335, multiplied by the 910,000 shares into which the debt is convertible). The Company recorded $623 as a beneficial conversion discount, which is being amortized on a straight-line basis over the life of the promissory note.
The fair value of the convertible note, net of the beneficial conversion and debt issuance discount, was determined to be $1,391, resulting in a fair value discount on issuance of $397, which is being amortized on a straight-line basis over the life of the promissory note.
The initial fair value of the notes was determined using the expected cash flows of the notes based on its contractual terms and then discounted to the issuance date at a risk-adjusted discount rate that considers the relative risk of achieving those cash flows and the time value of money. The discount rate used was 15%.
| F-68 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Secured Promissory Notes - FBS Penn
In July 2019, in connection with the FBS Penn acquisition, the Company issued $28,122 by way of certain 10% secured notes due to the sellers, with principal payments, together with accrued interest through each such date, due in tranches. These notes were repaid in full, including any accrued interest, prior to maturity during the fourth quarter of 2020.
Secured Promissory Notes - FBS Nevada
In July 2019, in connection with the FBS Nevada management services and purchase agreements and the related acquisition of the associated real estate, the Company issued $2,250 in promissory notes and $375 in other secured notes as part of the consideration. The notes bore interest at 10% per annum. In April 2021, the Company completed the acquisition of 100% of FBS NV equity, which the Company had been operating under an MSA since 2019. In April 2021, the remaining principal balances of the promissory notes (plus accrued interest) were repaid in full.
Secured Promissory Notes - FBS Ohio
In June 2019, the Company entered into a definitive agreement with a FBS Ohio and issued $1,500 in 18-month secured sellers’ notes as part of the consideration. The notes bore interest at 10% per annum and were payable quarterly. The Company repaid these notes in full during the fourth quarter of 2020.
OTHER DEBT
Arlington Facility
In November 2021, the Company closed on the purchase of a property in Arlington, Virginia, for $7,000. On December 28, 2021, the Company entered into $6,900 credit facility (“Arlington Facility”) with a bank to refinance the purchase. As of December 31, 2021, the Company had drawn down $5,000. As of December 31, 2021, the Company had $1,900 of remaining availability under the Arlington Facility. The Arlington Facility bears a fixed interest rate of 5.875% per annum payable monthly and will mature on January 1, 2027, approximately 5 years from the closing date. The effective interest rate was 6.35% as of December 31, 2021.
The Company is subject to a prepayment penalty on the principal balance as follows: year one - 5%, year two - 4%, year three - 3% and year four - 2%. The carrying amount of the Arlington property was $7,028 as of December 31, 2021.
The Arlington Facility is subject to certain financial and non-financial debt covenants. Beginning December 31, 2022, the Company must maintain a Debt Service Coverage Ratio of 1.4 times at the subsidiary level and 1.2 times at the consolidated Company level, both of which are measured at year end. Per the Arlington Facility agreement, the Debt Coverage Ratio is defined as net operating income divided by annual debt service (principal and interest paid on the Arlington Facility) at the subsidiary level. At the consolidated Company level, the Debt Coverage Ratio is defined as Adjusted EBITDA (at the consolidated Company level) divided by annual debt service (principal and interest paid on the Arlington Facility.) As of December 31, 2021, the Company was in compliance with all applicable covenants.
Financing Obligations
The financing obligations primarily relate to a purchase of a Santa Barbara property that closed in July 2020 for $3,100 in cash proceeds. The carrying amount of the property as of December 31, 2021 and 2020 was $2,941 and $3,030, respectively. The initial monthly payment was $27 and monthly payments for the financing obligation increase 2.25% annually and the expected term is 30 years, including renewals. The effective interest rate for this financing obligation is 11.9%.
| F-69 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Unsecured Credit Lines - Jushi Europe
During the fourth quarter of 2020, Jushi Europe entered into a credit agreement with a sibling of the Jushi Europe non-controlling partner and received €500 (approximately $614) principal amount. In January 2021, Jushi Europe received €1,000 approximately ($1,214 as of December 31, 2021) principal amount pursuant to a credit agreement with an individual. These credit agreements accrue interest at 5% per annum, payable annually in arrears, and mature on November 11, 2024. The outstanding balance may be prepaid at any time prior to maturity without penalty and may be offset with receivables from the lender. Subsequent to December 31, 2021, it was determined that Jushi Europe was insolvent. The insolvency created an event of default under the unsecured credit agreements and the notes are immediately due and payable.
In April 2021, Jushi Europe entered into an unsecured bridge loan with the Company (51% owner) and an investment partner for a total of €1,800 (~$2,141) principal amount, of which €900 (~$1,070) was contributed by the Company and is eliminated in consolidation. In September 2021, the parties amended the loan agreement and an additional €1,200 (~$1,390) in funding was provided for Jushi Europe, of which 51% was contributed by the Company and is eliminated in consolidation. The bridge loans, as amended, currently accrue interest at 0.5% per annum, which is the foreign marginal lending facility rate plus 25 basis points. All payments including interest are due on maturity, which is 180 days post amendment. These loans had not yet been repaid and are delinquent. Refer to Note 17 - Non-Controlling Interests for additional information on Jushi Europe.
| 12. LEASE OBLIGATIONS |
The Company leases certain business facilities for corporate, retail and cultivation purposes in Florida, Pennsylvania, Ohio, California, New York, Virginia, Illinois, Colorado and Massachusetts from third parties under lease agreements that specify minimum rentals. In addition, the Company leases certain equipment for use in cultivation and extraction activities. The Company determines whether a contract is or contains a lease at the inception of the contract. The expiry dates of the leases, including reasonably certain estimated renewal periods, are between 2022 and 2050 and contain certain renewal provisions. The Company’s lease agreements do not contain any material residual value guarantees or material restrictive covenants.
| F-70 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Management elected not to separate lease and non-lease components for all of the Company’s leases. For leases with a term of 12 months or less, management elected the short-term lease exemption, which allowed the Company to not recognize right-of-use assets or lease liabilities for qualifying leases existing at transition and new leases the Company may enter into in the future. As of December 31, 2021, estimated future minimum lease payments under non-cancelable operating leases having an initial term of one year or less were $247 and are not capitalized in the consolidated balance sheet. The Company’s rent expense related to short-term leases is included in general and administrative expenses and was $406 and $156 for the years ended December 31, 2021 and 2020, respectively.
The following table provides the components of lease cost recognized in the consolidated statements of operations and comprehensive income (loss) for the year ended December 31, 2021.
| December 31, 2021 | December 31, 2020 | |||||||
| Operating lease cost | $ | 2,585 | $ | 1,555 | ||||
| Finance lease cost: | ||||||||
| Amortization of lease assets | 3,155 | 706 | ||||||
| Interest on lease liabilities | 9,158 | 2,447 | ||||||
| Total finance lease cost | $ | 12,313 | $ | 3,153 | ||||
| Variable lease cost | 355 | $ | 34 | |||||
| Total lease cost | $ | 15,253 | $ | 4,742 | ||||
All extension options that are reasonably certain to be exercised have been included in the measurement of lease obligations. The Company reassesses the likelihood of extension option exercise if there is a significant event or change in circumstances within its control.
Other information related to operating and finance leases as of and for the years ended December 31, 2021 and 2020 are as follows:
| December 31, 2021 | December 31, 2020 | |||||||||||||||
| Finance Leases | Operating Leases | Finance Leases | Operating Leases | |||||||||||||
| Weighted average discount rate | 11.75 | % | 11.50 | % | 16.28 | % | 14.65 | % | ||||||||
| Weighted average remaining lease term (in years) | 22.6 | 14.6 | 23.7 | 13.7 | ||||||||||||
| Cash paid for amounts included in the measurement of lease liabilities | 7,805 | 2,080 | $ | 1,848 | $ | 1,331 | ||||||||||
| F-71 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
The maturities of the contractual undiscounted lease liabilities as of December 31, 2021 are as follows:
| Finance Leases | Operating Leases | ||||||||
| 2022 | $ | 13,897 | $ | 2,927 | |||||
| 2023 | 10,879 | 3,012 | |||||||
| 2024 | 11,004 | 2,719 | |||||||
| 2025 | 11,519 | 2,522 | |||||||
| 2026 | 11,472 | 2,292 | |||||||
| Thereafter | 270,635 | 27,866 | |||||||
| Total undiscounted lease liabilities | 329,406 | 41,338 | |||||||
| Interest on lease liabilities | (228,489 | ) | (23,430 | ) | |||||
| Total present value of minimum lease payments | $ | 100,917 | $ | 17,908 | |||||
| Lease liabilities - current portion | $ | 12,620 | $ | 2,745 | |||||
| Lease liabilities - non-current | $ | 88,297 | $ | 15,163 | |||||
| 13. DERIVATIVE LIABILITIES |
A summary of the Company’s derivative liabilities as of December 31, 2021 and 2020 are as follows:
| December 31, 2021 | December 31, 2020 | |||||||
| Derivative Warrants liability | $ | 92,261 | $ | 204,611 | ||||
| Prepayment Option liability | 174 | 750 | ||||||
| Total derivative liabilities, carrying amount | $ | 92,435 | $ | 205,361 | ||||
The continuities of the Company’s derivative liabilities are as follows:
| Derivative Warrants Liability (1) | Prepayment Option Liability | Total Derivative Liabilities | ||||||||||
| Carrying amounts as of January 1, 2020 | $ | 5,529 | $ | — | $ | 5,529 | ||||||
| Fair value allocated to Derivative Warrants on issuance | 30,245 | — | 30,245 | |||||||||
| Warrant modifications (2) | 4,179 | — | 4,179 | |||||||||
| Fair value changes | 173,397 | 750 | 174,147 | |||||||||
| Derivative Warrants exercised | (8,739 | ) | — | (8,739 | ) | |||||||
| Carrying amounts as of December 31, 2020 | $ | 204,611 | $ | 750 | $ | 205,361 | ||||||
| Fair value changes | (104,594 | ) | (576 | ) | (105,170 | ) | ||||||
| Derivative Warrants exercised | (7,756 | ) | — | (7,756 | ) | |||||||
| Carrying amounts as of December 31, 2021 | $ | 92,261 | $ | 174 | $ | 92,435 | ||||||
| (1) | Refer to Note 14 - Equity for the continuity of the number of these warrants outstanding. |
| (2) | The gains (losses) on warrant modifications are included in other income (expense), net in the consolidated statements of operations and comprehensive income (loss). |
| F-72 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Derivative Warrants Liability
The warrants to purchase Subordinate Voting Shares of the Company which were issued in connection with the Senior Notes (the “Derivative Warrants”) have an expiration date of December 23, 2024 and an exercise price of US$1.25. There were 40,124,355 and 42,017,892 Derivative Warrants outstanding as of December 31, 2021 and 2020, respectively. The Derivative Warrants may be net share settled.
These warrants are considered derivative financial liabilities measured at fair value with all gains or losses recognized in profit or loss (“FVTPL”) as the settlement amount for the warrants may be adjusted during certain periods for variables that are not inputs to standard pricing models for forward or option equity contracts, i.e., the “fixed for fixed” criteria under ASC 815-40. The estimated fair value of the Derivative Warrants is measured at each reporting period and an adjustment is reflected in fair value changes in derivatives in the consolidated statements of operations and comprehensive income (loss). These are Level 3 recurring fair value measurements. The derivatives are classified as non-current liabilities. The estimated fair value of the Derivative Warrants as of the balance sheet date was determined using the Monte Carlo simulation model taking into account the fair value of the Company’s Subordinate Voting Shares on the valuation dates and into the future encompassing a wide range of possible future market conditions. The assumptions used in the calculations as of the balance sheet dates and used in the determination of the initial or settlement fair values during the years presented included the following:
| As of December 31, | Year Ended December 31, | ||||||
| 2021 | 2020 | 2021 | 2020 | ||||
| Stock price | $3.25 | $5.86 | $3.79 - $8.08 | $1.21- $5.58 | |||
| Risk-free annual interest rate | 0.97% | 0.26% | 0.31% - 0.99% | 0.11% - 1.74% | |||
| Range of estimated possible exercise price | $0.04 - $1.25 | $0.01 - $1.25 | $1.25 | $0.03 - $1.58 | |||
| Volatility | 73% | 80% | 73% - 80% | 75% - 85% | |||
| Remaining life (years) | 3 years | 4 years | 3 - 4 years | 4 - 5 years | |||
| Forfeiture rate | 0% | 0% | 0% | 0% | |||
| Expected annual dividend yield | 0% | 0% | 0% | 0% | |||
Volatility was estimated by using a weighting of the Company’s volatility and the average historical volatility of comparable companies from a representative peer group of publicly traded cannabis companies. The risk-free interest rate for the expected life of the Warrants was based on the yield available on government benchmark bonds with an approximate equivalent remaining term. The expected life is based on the contractual term. If any of the assumptions used in the calculation were to increase or decrease, this could result in a material or significant increase or decrease in the estimated fair value of the derivative liability. For example, the following table illustrates an increase or decrease in certain significant assumptions as of the balance sheet dates:
| As of December 31, 2021 | As of December 31, 2020 | ||||||||||||||||||||||||
| Input | Effect of 10% Increase | Effect of 10% Decrease | Input | Effect of 10% Increase | Effect of 10% Decrease | ||||||||||||||||||||
| Stock price | $ | 3.25 | $ | 12,781 | $ | (10,834 | ) | $ | 5.86 | $ | 21,856 | $ | (23,707 | ) | |||||||||||
| Volatility | 73 | % | $ | 4,473 | $ | (3,210 | ) | 80 | % | $ | 1,508 | $ | (4,239 | ) | |||||||||||
| F-73 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Prepayment Option
The mandatory Prepayment Option on the 10% Senior Notes (Refer to “Senior Notes in Note 11 - Debt) represents an embedded derivative which must be bifurcated and recorded at fair value (FVTPL) at each reporting period. The estimated fair value of the Prepayment Option was determined using a probability-weighted discounted cash flow analysis, taking into account the future likelihood of the mandatory redemption. These are Level 3 recurring fair value measurements. The significant assumptions used in the calculations include: the probability of a mandatory redemption, a mid-point redemption date, and the discount rate, which is based on market yields of similar host instruments. If any of the assumptions used in the calculation were to increase or decrease, this could result in a material increase or decrease in the estimated fair value of the Prepayment Option.
| 14. EQUITY |
| (a) | Authorized |
The authorized share capital of the Company consists of common shares with an unlimited number of Subordinate Voting Shares (“SVS”), Multiple Voting Shares (“MVS”), and Super Voting Shares (“SV”). As of December 31, 2021, there were 182,707,359 SVS issued and outstanding and there were no MVS or SV outstanding. On August 9, 2021, all of the 149,000 previously issued and outstanding Super Voting Shares and all of the 4,000,000 previously outstanding Multiple Voting Shares were converted into Subordinate Voting Shares in accordance with their terms as described in Jushi Holdings Inc.’s Articles of Incorporation. All previously outstanding warrants to acquire Super Voting Shares and Multiple Voting Shares were also converted into warrants to acquire Subordinate Voting Shares, without any other amendment to the terms of such warrants.
(i) Subordinate Voting Shares
Holders of SVS (“SVS Holder”) are entitled to notice of and to attend any meeting of the shareholders of the Company, except a meeting of which only holders of another particular class or series of shares of the Company shall have the right to vote. At each such meeting SVS Holders are entitled to one vote in respect of each SVS held.
SVS Holders are entitled to receive as and when declared by the directors, dividends in cash or property of the Company.
| (ii) | Multiple Voting Shares |
The holders of Multiple Voting Shares (the “Multiple Voting Holders”) shall have the right to 10 votes for each Subordinate Voting Share into which such Multiple Voting Shares could then be converted, which for greater certainty, shall initially equal 10 votes per Multiple Voting Share, and with respect to such vote, such holder shall have full voting rights and powers equal to the voting rights and powers of the SVS Holders, and shall be entitled to notice of any shareholders’ meeting and shall be entitled to vote, together with holders of SVS, with respect to any question upon which SVS Holders have the right to vote.
The MVS Holders shall have the right to receive dividends, out of any cash or other assets legally available therefore, pari passu (on an as-converted basis, assuming conversion of all MVS into SVS at the applicable conversion ratio and ignoring any limitations on conversion as outlined in the Company Articles) as to dividends and any declaration or payment of any dividend on the SVS.
| F-74 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| (iii) | Super Voting Shares |
The holders of Super Voting Shares (the “Super Voting Holders”) shall have the right to 10 votes for each Subordinate Voting Share into which such Super Voting Shares could then be converted (as outlined in the Company’s Articles), which for greater certainty, shall initially equal 1,000 votes per Super Voting Share, and with respect to such vote, such holder shall have full voting rights and powers equal to the voting rights and powers of the Subordinate Voting Holders, and shall be entitled, to notice of any shareholders’ meeting and shall be entitled to vote, together with holders of Subordinate Voting Shares, with respect to any question upon which Subordinate Voting Holders have the right to vote.
The Super Voting Holders shall have the right to receive dividends, out of any cash or other assets legally available therefore, pari passu (on an as converted basis, assuming conversion of all Super Voting Shares into Subordinate Voting Shares at the applicable Conversion Ratio and ignoring any limitations on conversion as outlined in the Company Articles) as to dividends and any declaration or payment of any dividend on the Subordinate Voting Shares.
Dividends
The Company has not paid dividends on any of its classes of stock. The Company’s 10% Senior Notes and Acquisition Facility contain covenants that, among other things, limit the Company’s ability to declare or pay dividends.
Public Offerings
On October 23, 2020, the Company issued 11,500,000 subordinate voting shares at a price of C$3.55 per share for total gross proceeds of approximately C$40,825 (~USD$31,076 ), which included the full exercise of the over-allotment option granted to the underwriters. The Company incurred $1,833 of costs directly related to the public offering.
On January 7, 2021, the Company closed on an overnight marketed offering for an aggregate of 6,210,000 subordinate voting shares at a price of C$6.50 per share for total gross proceeds of C$40,365, and total net proceeds of C$37,768 ($29,762). On February 12, 2021, the Company closed on an overnight marketed offering for an aggregate 7,475,000 subordinate voting shares at a price of C$10.00 per share for total gross proceeds of C$74,750 and total net proceeds of C$70,922 ($55,902). These offerings included the full exercise of the over-allotment option granted to the underwriters.
| F-75 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Warrants
Each warrant entitles the holder to purchase one share of the same class of common share. The following table summarizes all warrants outstanding as of December 31, 2021:
| Expiration Date | Exercise Price ($) |
Number of Warrants | ||
| February - July 2022 | 1.25 - 1.58 | 715,846 | ||
| February - September 2023 | 1.47 - 1.50 | 337,500 | ||
| February - December 2024 | 1.25 | 40,124,355 | ||
| October - November 2025 | 2.47 - 2.94 | 1,435,075 | ||
| November 2026 | 4.18 | 300,000 | ||
| January - June 2029 | 0.50 - 2.00 | 26,367,627 | ||
| Total warrants | 69,280,403 |
As of December 31, 2021, warrants issued and outstanding have a weighted-average remaining contractual life of 4.7 years. Certain warrants may be net share settled.
The activity of warrants (on an as-converted basis) outstanding was as follows:
| Non-Derivative Warrants | Derivative Warrants (2) | Total Number of Warrants | Weighted - Average Exercise Price | ||||||||||||||
| Balance as of January 1, 2020 (1) | 59,686,712 | 7,392,157 | 67,078,869 | $ | 1.60 | ||||||||||||
| Granted (3) (4) (5) (6) | 2,821,692 | 39,540,902 | 42,362,594 | 1.26 | |||||||||||||
| Exercised (7) (13) | (22,182,244 | ) | (2,302,258 | ) | (24,484,502 | ) | 1.90 | ||||||||||
| Cancelled (8) | (2,811,916 | ) | — | (2,811,916 | ) | 1.94 | |||||||||||
| Expired | (750,000 | ) | — | (750,000 | ) | 1.50 | |||||||||||
| Voided (3) (9) | — | (2,612,909 | ) | (2,612,909 | ) | 1.58 | |||||||||||
| Balance as of December 31, 2020 (1) | 36,764,244 | 42,017,892 | 78,782,136 | $ | 1.31 | ||||||||||||
| Granted (10) | 300,000 | — | 300,000 | 4.18 | |||||||||||||
| Exercised (11) (13) | (7,658,196 | ) | (1,893,537 | ) | (9,551,733 | ) | 2.26 | ||||||||||
| Cancelled | (250,000 | ) | — | (250,000 | ) | 1.50 | |||||||||||
| Balance as of December 31, 2021 | 29,156,048 | 40,124,355 | 69,280,403 | $ | 1.19 | ||||||||||||
| Exercisable as of December 31, 2021 (12) | 27,292,712 | 40,124,355 | 67,417,067 | $ | 1.15 | ||||||||||||
| (1) | Total number of outstanding warrants on a non-converted basis was 62,669,886 and 50,966,619 as of December 31, 2020 and January 1, 2020, respectively. In August 2021, all outstanding warrants to acquire Super Voting Shares and Multiple Voting Shares were converted into warrants to acquire Subordinate Voting Shares, without any other amendment to the terms of such warrants. The 162,750 outstanding warrants for Super Voting Shares were converted to 16,275,000 warrants. The 6,750,000 warrants for Multi-Voting shares were converted into 6,750,000 warrants for Subordinate Voting Shares. |
| (2) | Derivative warrants which were issued to the 10% Senior Notes holders and which have an exercise price of $1.25. These warrants represent a derivative liability and are therefore not classified as equity in the statement of financial position. Refer to Note 13 - Derivative Liabilities. |
| (3) | Includes 2,375,372 derivative warrants that were issued in 2020 in connection with the issuance of Warrant Notes and subsequently voided when the Warrant Notes were exchanged for OID Notes. Refer to “Senior Notes” in Note 11 - Debt. |
| (4) | Includes 950,148 derivative warrants issued in connection with the Warrant Notes issued for the TGS NCI buyout, 200,000 were issued in connection with the TGS Transaction and 633,433 were issued in connection with the Warrant Notes issued for the Agape acquisition. Refer to Note 7 - Acquisitions. |
| (5) | During the year ended December 31, 2020, the Company issued 301,539 warrants for broker services rendered in connection with the issuance of 10% Senior Notes, with a weighted average per share exercise price of $1.34, and a weighted average grant date fair value of $0.54. |
| F-76 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| (6) | During the year ended December 31, 2020, the Company issued 2,285,075 warrants for other consulting or other services with a weighted average per share exercise price of $2.17 and a weighted average per share grant date fair value of $1.07. Certain of the warrants are subject to vesting and the expense relating to these awards is included in share-based compensation expense in the consolidated statements of operations and comprehensive income (loss). |
| (7) | The weighted average share price as of the dates of exercise was $4.16. Warrants exercised includes warrants exercised as a result of the Company acceleration of the expiration of certain warrants. Warrants exercised includes warrants that were cashless net exercises. The Company received $45,510 in cash proceeds from warrants that were exercised during the year ended December 31, 2020. |
| (8) | Includes the cancellation of 2,500,000 warrants returned in the TGS Transaction. Refer to Note 7 - Acquisitions. |
| (9) | Includes 237,537 derivative warrants that were issued in connection with Warrant Notes during the year ended December 31, 2019 and subsequently voided during the first quarter of 2020 when the Warrant Note was exchanged for an OID Note. |
| (10) | During the year ended December 31, 2021, the Company issued 300,000 warrants for other consulting or other services with a weighted average per share exercise price of $4.18 and a weighted average per share grant date fair value of $2.14. Certain of the warrants are subject to vesting and the expense relating to these awards is included in share-based compensation expense in the consolidated statements of operations and comprehensive income (loss). |
| (11) | The weighted average share price as of the dates of exercise was $5.54. The Company issued 8,667,173 SVS and received $16,922 in cash proceeds during the year ended December 31, 2021. |
| (12) | The weighted average remaining contractual life is 4.7 years. |
| (13) | Several of the warrants contained terms under which the Company could force exercise and many of the warrants contained terms under which the Company could accelerate the expiration date following a liquidity event if the volume weighted average price for any 20 or 30 consecutive trading days, as applicable, equals or exceeds a certain per share price. During the years ended December 31, 2021 and 2020, the Company exercised its rights to accelerate the expiration of certain outstanding warrants issued to participants in the Company’s private placement offerings that closed in March 2019, October 2018, June 2018 and April 2018, respectively. |
Grant Date Fair Values - Non-Derivative Warrants
The fair value of the warrants issued for services or consulting, including for broker services was determined using the Black-Scholes option-pricing model. The following assumptions were used for the calculations at the time of issuance or grant:
| Year Ended December 31, | |||
| 2021 | 2020 | ||
| Stock price | $4.16 | $0.71 -$3.18 | |
| Risk-free annual interest rate | 0.97%-1.16% | 0.11% - 1.55% | |
| Expected annual dividend yield | nil | nil | |
| Expected stock price volatility | 73% | 78% - 85% | |
| Expected life of warrants | 3-4 years | 1 - 4 years | |
| Forfeiture rate | —% | —% | |
Volatility was estimated by using a weighting of the average historical volatility of comparable companies from a representative peer group of publicly traded cannabis companies and the Company volatility. The expected life in years represents the period of time that warrants issued are expected to be outstanding. The risk-free rate is based on U.S. Treasury bills with a remaining term equal to the expected life of the warrants. The Company currently does not anticipate paying dividends in the foreseeable future and as a result, the expected annual dividend yield is assumed to be 0%.
| F-77 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| 15. SHARE-BASED COMPENSATION AND OTHER BENEFITS |
The components of share-based compensation expense are as follows:
| Year Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Stock options | $ | 7,464 | $ | 2,741 | ||||
| Restricted stock | 5,873 | 5,898 | ||||||
| Warrants | 1,169 | 953 | ||||||
| Total share-based compensation expense | $ | 14,506 | $ | 9,592 | ||||
Equity Incentive Plan
Under the Plan, non-transferable options to purchase Subordinate Voting Shares and restricted Subordinate Voting Shares of the Company may be issued to directors, officers, employees, or consultants of the Company. The Plan authorizes the issuance of up to 15% (plus an additional 2% inducements for hiring employees and senior management) of the number of outstanding shares of common stock (of all classes) of the Company (the “Share Reserve”). Incentive stock options are limited to the Share Reserve as of June 6, 2019. As of December 31, 2021, the maximum number of incentive awards available for issuance under the Plan, including additional awards available for certain new hires, was 7.9 million.
| (a) | Stock Options |
The stock options issued by the Company are options to purchase SVS of the Company. All stock options issued have been issued to employees of certain subsidiaries of the Company under the Company’s Plan. Such options generally expire in ten years from the date of grant and generally vest ratably over three years from the grant date. The options may be net share settled.
Details of the stock option activity are follows:
| Number of Stock Options | Weighted- Average Per Share Exercise Price | ||||||||
| Issued and Outstanding as of December 31, 2020 | 9,573,834 | $ | 2.00 | ||||||
| Granted (1) (2) (3) | 11,486,952 | $ | 4.16 | ||||||
| Exercised (4) | (291,664 | ) | $ | 1.90 | |||||
| Forfeited/expired | (340,002 | ) | $ | 3.23 | |||||
| Issued and Outstanding as of December 31, 2021 | 20,429,120 | $ | 3.20 | ||||||
| Exercisable as of December 31, 2021 | 6,040,093 | $ | 1.97 | ||||||
| (1) | The weighted-average per share grant date fair value was $2.61. |
| (2) | Includes 5,100,000 stock options issued to senior key management. The weighted average exercise price and grant date fair value was $4.12 and $2.57, respectively. |
| (3) | Includes 213,952 stock options issued to independent directors. The weighted average exercise price and fair value was $4.03 and $2.38, respectively. |
| (4) | The weighted-average share price at the date of exercise was $4.92. The Company issued 216,133 SVS and received $208 in cash proceeds from stock options that were exercised during the year ended December 31, 2021. |
| F-78 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
The following table summarizes the issued and outstanding stock options as of December 31, 2021:
| Expiration Year | Stock Options Outstanding |
Exercise Price | Stock Options Exercisable | ||||
| 2028 | 870,000 | $1.00 - $1.35 | 870,000 | ||||
| 2029 | 7,169,668 | $1.26 - $2.75 | 4,766,987 | ||||
| 2030 | 987,500 | $0.91 - $ 3.98 | 329,156 | ||||
| 2031 | 11,401,952 | $3.70 - $6.53 | 73,950 | ||||
| 20,429,120 | 6,040,093 |
As of December 31, 2021, there was $24,942 of total unrecognized compensation expense related to unvested stock options, which is expected to be recognized over a weighted-average period of 16 months. As of December 31, 2021, total stock options issued and outstanding had a weighted-average remaining contractual life of 8.7 years and an intrinsic value of $11,583, and the exercisable stock options had a weighted-average remaining contractual life of 7.4 years and an intrinsic value of $8,013.
In determining the amount of share-based compensation expense related to stock options issued, the Company used the Black-Scholes option-pricing model to establish the measurement date fair value of stock options granted during the period. The following assumptions were applied at the time of grant:
| Year Ended December 31, | |||
| 2021 | 2020 | ||
| Stock price | $3.62 - $6.48 | $0.91 - $3.98 | |
| Risk-free annual interest rate | 0.45% - 1.59% | 0.26% - 1.47% | |
| Expected annual dividend yield | 0% | 0% | |
| Volatility | 72% -78% | 80% - 85% | |
| Expected life of stock options | 5 - 7.5 years | 5 - 6.5 years | |
| Forfeiture rate | 0% | 0% | |
Volatility was estimated by using the Company volatility and a weighting of the average historical volatility of comparable companies from a representative peer group of publicly traded cannabis companies. The expected life in years represents the period of time that stock options issued are expected to be outstanding, using the simplified method. The simplified method represents the Company’s best estimate of the expected term of the options, given the Company’s limited history available. The risk-free rate is based on U.S. Treasury bills with a remaining term equal to the expected life of the options. The Company does not anticipate paying dividends in the foreseeable future, and as a result, the expected annual dividend yield is expected to be 0%.
| F-79 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| (b) | Restricted Stock Grants |
The Company grants restricted SVS to independent directors, management, former owners of acquired businesses or assets, and to consultants and other employees. The restricted SVS are included in the issued and outstanding SVS. The activity for unvested restricted stock grants is as follows:
| Number of Restricted Subordinate Voting Shares | |||||
| Unvested restricted stock as of December 31, 2020 | 6,438,186 | ||||
| Granted (1) | 65,398 | ||||
| Cancelled | (219,479 | ) | |||
| Vested | (3,424,954 | ) | |||
| Unvested restricted stock as of December 31, 2021 | 2,859,151 | ||||
| (1) | The weighted-average grant date fair value of the RSAs was $3.66 and is based on the closing price of the subordinate voting shares of the Company on the grant date. |
Generally, restricted stock awards will vest either one-third on each anniversary of service from the vesting start date or will be fully vested on the completion of one year of full service from the vesting start date, depending on the award. As of December 31, 2021, there was $2,116 of total unrecognized compensation expense related to unvested restricted stock awards, which is expected to be recognized over a weighted-average remaining vesting period of 1.2 years.
(c) Warrants
Refer to Note 14 - Equity for details on warrants issued by the Company for consulting or other services.
401(k) Plan
The Company maintains a 401(k) plan, which is generally available to eligible employees. The Company makes safe harbor matching contributions, subject to a maximum contribution of 4% of the participant’s compensation. The employer matching contributions to the 401(k) plan were $616 and $327 for the years ended December 31, 2021 and 2020, respectively.
| F-80 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| 16. INCOME TAXES |
Details of the Company’s income tax expense (benefit) are as follows:
| Year Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Current tax expense: | ||||||||
| Federal | $ | 25,501 | $ | 9,728 | ||||
| State | 9,234 | 4,452 | ||||||
| Foreign | — | — | ||||||
| $ | 34,735 | $ | 14,180 | |||||
| Deferred tax benefit: | ||||||||
| Federal | $ | (5,477 | ) | $ | (2,650 | ) | ||
| State | (1,724 | ) | (962 | ) | ||||
| Foreign | (3,874 | ) | (3,307 | ) | ||||
| $ | (11,075 | ) | $ | (6,919 | ) | |||
| Change in valuation allowance | 5,965 | 3,362 | ||||||
| Total income tax expense | $ | 29,625 | $ | 10,623 | ||||
The differences between the income tax expense and the expected income taxes based on the statutory tax rate applied to pre-tax earnings (loss) are as follows:
| Year Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Income (loss) before income taxes | $ | 47,104 | $ | (201,892 | ) | |||
| Statutory tax rate | 21.00 | % | 21.0 | % | ||||
| Tax benefit based on statutory rates | $ | 9,892 | $ | (42,397 | ) | |||
| Difference in tax rates | 16,753 | (53,432 | ) | |||||
| IRC Section 280E disallowed expenses | 12,520 | 3,961 | ||||||
| Share-based compensation | 2,361 | 2,099 | ||||||
| Interest expense and debt costs | 3,616 | 1,549 | ||||||
| Gain on fair value of derivative | (50,482 | ) | 83,498 | |||||
| Bargain purchase gain | — | (2,131 | ) | |||||
| Change in valuation allowance | 5,965 | 3,362 | ||||||
| State taxes, net | 1,249 | 1,477 | ||||||
| Change in uncertain tax positions | 26,823 | 11,857 | ||||||
| Other differences | 928 | 780 | ||||||
| Total income tax expense | $ | 29,625 | $ | 10,623 | ||||
| Effective tax rate | 62.9 | % | (5.3 | )% | ||||
| F-81 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Year-end deferred tax assets and liabilities were due to the following:
| Year Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Deferred tax assets: | ||||||||
| Lease liability | $ | 26,261 | $ | 8,364 | ||||
| Net operating losses | 7,941 | 3,837 | ||||||
| Financing fees | 2,289 | 1,562 | ||||||
| Start-up costs | 820 | 881 | ||||||
| Property and equipment | 957 | — | ||||||
| Other deferred tax assets | 1,295 | 172 | ||||||
| $ | 39,563 | $ | 14,816 | |||||
| Deferred tax liabilities: | ||||||||
| Right-of-use assets | $ | (24,406 | ) | $ | (7,362 | ) | ||
| Intangible assets | (17,943 | ) | (1,284 | ) | ||||
| Other deferred tax liabilities | (978 | ) | (497 | ) | ||||
| $ | (43,327 | ) | $ | (9,143 | ) | |||
| Valuation allowance | $ | (11,389 | ) | $ | (5,424 | ) | ||
| Net deferred tax (liabilities) assets (1) | $ | (15,153 | ) | $ | 249 | |||
| (1) | The net deferred tax liabilities are included in non-current income tax liabilities and the net deferred tax assets are included in other non-current assets in the consolidated balance sheets. |
Realization of deferred tax assets associated with the net operating loss carryforwards is dependent upon generating sufficient taxable income prior to their expiration. A valuation allowance to reflect management’s estimate of the temporary deductible differences that may expire prior to their utilization has been recorded at December 31, 2021 and December 31, 2020.
As of December 31, 2021, the Company had $26,697 of non-capital Canadian losses, $1,075 of capital Canadian losses, $3,363 of other foreign losses, $3,560 of state net operating losses which expire in 2038-2041. To the extent that the benefit from these loss carryforwards are not expected to be realized, the Company has recorded a valuation allowance as follows: $26,697 for non-capital Canadian losses, $1,075 for capital Canadian losses, $3,363 for other foreign losses, $1,830 for state net operating losses.
As the Company operates in the legal cannabis industry, the Company is subject to the limits of Internal Revenue Code (“IRC”) Section 280E for US federal income tax purposes as well as state income tax purposes for all states except for California and Colorado. Under IRC Section 280E, the Company is only allowed to deduct expenses directly related to cost of goods sold. This results in permanent differences between ordinary and necessary business expenses deemed non-allowable under IRC Section 280E.
The Company’s tax returns benefited from not applying IRC Section 280E to certain entities of the consolidated group either due to the entity not yet starting operations or because the entity had a separate trade or business that was not medical or recreational cannabis operations. The Company determined that it is not more likely than not these tax positions would be sustained under examination. As a result, the Company has an uncertain tax liability of $41,990 and $13,756 as of December 31, 2021 and 2020, respectively, inclusive of interest and penalties, which is included in non-current income tax liabilities in the consolidated balance sheets. Additionally, there are unrecognized deferred tax benefits of $3,269 and $7,424 as of December 31, 2021 and 2020, respectively. The Company does not expect the unrecognized tax benefits will materially increase or decrease as a result of the 2019 - 2021 tax return positions. As of December 31, 2021, all tax filings remain open for assessment.
| F-82 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
The total amount of interest and penalties recorded in the income statement for the year ended December 31, 2021 were $643 and $2,742. The total amount of interest and penalties recorded within income tax expense for the year ended December 31, 2020, was $45 and $nil.
A reconciliation of the beginning and ending amount of unrecognized tax benefits are as follows:
| Balance at January 1, 2020 | $ | 9,323 | ||
| Additions based on tax positions related to the current year | 11,812 | |||
| Additions for tax positions of prior years | — | |||
| Balance at December 31, 2020 | $ | 21,135 | ||
| Additions based on tax positions related to the current year | 20,368 | |||
| Additions for tax positions of prior years | — | |||
| Balance at December 31, 2021 | $ | 41,503 |
| 17. NON-CONTROLLING INTERESTS |
The components of the Company’s non-controlling interests and related activity are as follows:
| Dalitso | BHILH | Jushi Europe | Agape | Other Non- Material Interests | Total | |||||||||||||||||||
| Balance as of January 1, 2020 | $ | 9,642 | $ | — | $ | — | $ | — | $ | 18 | $ | 9,660 | ||||||||||||
| Acquisitions | — | 4,661 | — | 1,560 | — | 6,221 | ||||||||||||||||||
| Purchase of non-controlling interests | (8,359 | ) | (4,661 | ) | — | — | — | (13,020 | ) | |||||||||||||||
| Cash contribution by partner | — | — | 2,000 | — | — | 2,000 | ||||||||||||||||||
| Transactions with non-controlling interests | — | — | — | — | (6 | ) | (6 | ) | ||||||||||||||||
| (Loss) income | (1,283 | ) | — | (616 | ) | 2 | (11 | ) | (1,908 | ) | ||||||||||||||
| Balance as of December 31, 2020 | $ | — | $ | — | $ | 1,384 | $ | 1,562 | $ | 1 | $ | 2,947 | ||||||||||||
| Purchases of non-controlling interests | — | — | — | (1,562 | ) | — | (1,562 | ) | ||||||||||||||||
| Loss | — | — | (2,771 | ) | — | (1 | ) | (2,772 | ) | |||||||||||||||
| Balance as of December 31, 2021 | $ | — | $ | — | $ | (1,387 | ) | $ | — | $ | — | $ | (1,387 | ) | ||||||||||
Purchases of Non-Controlling Interests
Agape
On January 25, 2021, the Company acquired the remaining 20% of the equity interests of Agape from the non-controlling shareholders for 500,000 SVS for total estimated fair value of $3,425, based on a market price of $6.85 per share on the date of close. As a result of the transaction, the Company recorded a decrease to non-controlling interests of approximately $1,562. The Company now owns 100% of the issued and outstanding shares of Agape.
| F-83 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Dalitso
In November and December 2020, the Company closed on a series of transactions pursuant to which the Company acquired all the remaining 38.24% equity interests of Dalitso for total consideration with an estimated fair value of $14,846. The consideration comprised of 3,294,478 SVS (with a total fair value of $12,053), $381 in cash, and an unsecured convertible promissory note of principal amount $2,412. Refer to “Promissory Notes Payable” in Note 11 - Debt for details of this convertible promissory note. The SVS were valued based on the closing price of the SVS as of the dates of the transactions. The Company now owns 100% of the issued and outstanding shares of Dalitso.
TGSIH/BHILH
In the first quarter of 2020, previous litigation involving a non-controlling interest holder in BHILH/TGSIH was settled resulting in an agreement for the Company to purchase the remaining approximately 25% interest in BHILH/TGSIH held by the non-controlling interest holders. On February 21, 2020, the Company acquired the remaining approximately 25% interest in TGSIH for total consideration with an estimated fair value of $4,661 as of the date of the acquisition. The consideration comprised of $2,000 in cash (of which $150 related to the legal settlement and was expensed), 633,433 Subordinate Voting Shares (fair value $811), and $2,000 in 10% Senior Notes (fair value $1,325) with 950,148 warrants (fair value $675) to acquire Subordinate Voting Shares at an exercise price of ~$1.57 (the exercise price has since been updated to $1.25 as a result of a subsequent down-round). Refer to Note 13 - Derivative Liabilities for additional information. The terms of the Senior Notes are those described in Note 11 - Debt. Refer to Note 13 - Derivative Liabilities for fair value assumptions for warrants issued and bifurcated from the Senior Notes. The SVS were valued based on the closing price of the Subordinate Voting Shares as of the date of the TGSIH NCI Transaction. The Company now owns 100% of TGSIH.
Jushi Europe
In March 2020, the Company finalized its agreement to expand internationally through the establishment of Jushi Europe. The plan for Jushi Europe was to build out the Company’s European presence through a combination of strategic acquisitions, partnerships, and license applications, focused on supplying the highest-quality medical cannabis products to patients throughout Europe. During the first quarter of 2020, $2,000 in cash was contributed by a non-controlling investor partner. The Company owns 51% of Jushi Europe and is exposed, or has rights, to variable returns from Jushi Europe and has the power to govern the financial and operating policies of Jushi Europe through voting control so as to obtain economic benefits, and therefore the Company has consolidated Jushi Europe from the date of acquisition.
On February 22, 2022, Jushi Europe filed a notice of over-indebtedness with the Swiss courts. As a result of the impending proceedings, the Company determined that the assets of Jushi Europe were impaired and recognized an impairment loss of $4,561 for the year ended December 31, 2021, which is included in operating expenses in the consolidated statements of operation and comprehensive income (loss). The impairment loss is comprised of fixed asset impairment charges of $4,451 and other asset write-offs of $110.
| F-84 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| 18. NET INCOME (LOSS) PER SHARE |
The reconciliations of the earnings (loss) and the weighted average number of shares used in the computations of basic and diluted EPS are as follows:
| Year Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Numerator: | ||||||||
| Net income (loss) and comprehensive income (loss) attributable to Jushi shareholders - basic | $ | 20,251 | $ | (210,607 | ) | |||
| Dilutive effect of net income from derivative warrants liability | (104,594 | ) | — | |||||
| Net loss and comprehensive loss attributable to Jushi shareholders - diluted | $ | (84,343 | ) | $ | (210,607 | ) | ||
| Denominator: | ||||||||
| Weighted-average shares of common stock - basic | 170,292,035 | 108,485,158 | ||||||
| Dilutive effect of derivative warrants | 31,318,216 | — | ||||||
| Weighted-average shares of common stock - diluted | 201,610,251 | 108,485,158 | ||||||
| Net income (loss) per common share attributable to Jushi: | ||||||||
| Basic | $ | 0.12 | $ | (1.94 | ) | |||
| Diluted | $ | (0.42 | ) | $ | (1.94 | ) | ||
On August 9, 2021, all of the 149,000 previously issued and outstanding Super Voting Shares and all of the 4,000,000 previously outstanding Multiple Voting Shares were converted into Subordinate Voting Shares in accordance with their terms as described in Jushi Holdings Inc.’s Articles of Incorporation. Refer to Note 14 - Equity. The number of basic and diluted weighted-average shares outstanding for the years ended December 31, 2021 and 2020, assumes the conversion of the Multi Voting Share and Super Voting Shares into Subordinate Voting Shares as of the beginning of the year. Other than voting rights, the Multi Voting Shares and Super Voting Shares had the same rights as the Subordinate Voting Shares and therefore all these shares are treated as the same class of common stock for purposes of the earnings (loss) per share calculations.
The weighted-average shares outstanding for the year ended December 31, 2021 includes unvested restricted stock. The weighted-average shares outstanding for the year ended December 31, 2020 excludes unvested restricted stock awards as they are not considered participating securities under ASC 260 Earnings Per Share, because although the holders have dividend rights regardless of vesting, they are not obligated to participate in losses until the shares are vested. Shares issued to employees for which a corresponding non-recourse promissory note receivable with the employee is outstanding, are not considered participating securities and are not included in the weighted average shares outstanding.
| F-85 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
The computations of net income (loss) per share for the years ended December 31, 2021 and 2020 exclude any adjustments relating to convertible promissory notes, which are a liability classified, share-settled obligation (refer to Note 11 - Debt), because the shares to be delivered are not participating securities as they do not have voting rights and are not entitled to participate in dividends until they are issued. For purposes of computing diluted earnings (loss) per share for the years ended December 31, 2021, and 2020, outstanding stock options, non-derivative warrants and the convertible notes are considered anti-dilutive because the Company was in a net loss position, or was in a net loss position after adjusting for the effect of the derivative warrants liability. The following summarizes equity instruments that may, in the future, have a dilutive effect on earnings (loss) per share, but were excluded from consideration in the computation of diluted net loss per share for the years ended December 31, 2021 and 2020, because the impact of including them would have been anti-dilutive:
| December 31, 2021 | December 31, 2020 | |||||||
| Stock options | 20,429,120 | 9,573,834 | ||||||
| Warrants (on an as-converted basis) | 29,156,048 | 78,782,136 | ||||||
| Convertible promissory notes (on an as-converted basis) | 910,000 | 930,000 | ||||||
| 50,495,168 | 89,285,970 | |||||||
| 19. OPERATING EXPENSES |
The major components of operating expenses are as follows:
| Year Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Salaries, wages and employee related expenses | $ | 58,228 | $ | 21,781 | ||||
| Other general and administrative expenses (1) | 32,652 | 18,558 | ||||||
| Share-based compensation expense | 14,506 | 9,592 | ||||||
| Depreciation and amortization expense | 5,805 | 4,154 | ||||||
| Goodwill impairment | 1,783 | — | ||||||
| Asset impairment | 4,561 | — | ||||||
| Acquisition and deal costs | 1,624 | 810 | ||||||
| Total operating expenses | $ | 119,159 | $ | 54,895 | ||||
| (1) | Other general and administrative expenses are primarily comprised of rent and related expenses, professional fees and legal expenses, marketing and selling expenses, insurance costs, administrative and application fee, software and technology costs, travel, entertainment and conferences and other. |
| 20. OTHER INCOME (EXPENSE) |
The components of other expense, net are as follows:
| Year Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| Gains on business combinations | $ | — | $ | 10,149 | ||||
| Gains (losses) on investments and financial assets | 1,216 | (2,473 | ) | |||||
| Losses on debt redemptions/extinguishments/modifications | (3,815 | ) | (1,853 | ) | ||||
| Gains (losses) on legal settlements | 10,350 | (2,217 | ) | |||||
| Other | 558 | 96 | ||||||
| Other income, net | $ | 8,309 | $ | 3,702 | ||||
| F-86 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| 21. RELATED PARTY TRANSACTIONS |
The Company had the following related party transactions:
| Year Ended December 31, | As of December 31, | |||||||||||||||
| 2021 | 2020 | 2021 | 2020 | |||||||||||||
| Nature of transaction | Related Party Income (Expense) $ | Related Party Prepaid/Receivable (Payable) $ | ||||||||||||||
| Management services agreements (1)(2) | $ | (42 | ) | $ | (248 | ) | $ | — | $ | 7 | ||||||
| Directors’ fees (3) | (36 | ) | (200 | ) | (13 | ) | (50 | ) | ||||||||
| 10% Senior Notes - interest expense and principal amount (4)(5) | (4 | ) | (2,305 | ) | (1,194 | ) | (25,475 | ) | ||||||||
| Loans to senior key management - interest charged and principal plus accrued interest outstanding (6) | 90 | 21 | — | 2,470 | ||||||||||||
| (1) | Includes fees paid to entities controlled by the Company’s Chief Executive Officer, James Cacioppo, for shared costs of administrative services, the provision of financial and research-related advice, and sourcing and assisting in mergers, acquisitions and capital transactions. These amounts are included in operating expenses within the consolidated statements of operations and comprehensive income (loss). |
| (2) | Excludes expense from previously issued warrants, which is included in stock-based compensation expense. For the years ended December 31, 2021 and 2020, total expense for warrants issued in connection with these management services agreements was $106 and $359. In addition, the Company incurred expense of $193 in connection with warrants issued in connection with a consulting agreement with a significant investor and former director of the board (Denis Arsenault) during the year ended December 31, 2020. The Company did not issue any additional warrants in connection with these related party consulting and/or management services agreements during the years ended December 31, 2021 and 2020. |
| (3) | Excludes expense from restricted stock awards, which is included in stock-based compensation expense. RSA and stock option expense relating to the Company’s board of independent directors were $615 and $483 for the years ended December 31, 2021 and 2020, respectively. For the year ended December 31, 2021, the majority of the board of directors’ fees were paid in the form of restricted stock or stock options. Refer to Note 15 - Share-Based Compensation and Other Benefits. Joseph Max Cohen resigned from the board of directors during the first quarter of 2021 and Marina Hahn was appointed to the board of directors during the second quarter of 2021. |
| (4) | For the year ended December 31, 2020, interest expense includes amounts related to certain senior key management, directors and other employees as well as a significant investor. Interest expense for the year ended December 31, 2021 cannot be reliably determined as the majority of the senior notes are publicly traded since December 2020 and the majority of the notes for related parties were sold and/or redeemed during the year ended December 31, 2021. Principal amounts outstanding as of December 31, 2021 and 2020 are also estimates for this reason. |
| (5) | Refer to “Senior Notes” in Note 11 - Debt for information on the Senior Notes redemptions which occurred in January and February of 2021. Of the total amount of Senior Notes redeemed in January 2021, $3,072 was estimated to be related to related parties including certain senior management, a significant shareholder and employees. In February 2021, certain senior key management and a significant investor sold their remaining principal amounts of publicly traded 10% Senior Notes totaling $19,219. |
| (6) | In connection with tax elections related to the issuance of certain restricted stock to key management personnel during the year ended December 31, 2020, the Company paid taxes totaling $2,450 on behalf of these employees, for which promissory notes were issued in October of 2020, and which amounts, including accrued interest, were included in other non-current assets as of December 31, 2020. Refer to Note 9 - Other Non-Current Assets. The promissory notes bore interest at 5% compounded annually, which was payable on maturity. In January 2021, an executive received a loan from the Company of $174 for withholding tax requirements for RSAs issued to the executive, which was repaid in full via payroll deductions. In April 2019, the Company entered into promissory notes with certain executives for the purchase of restricted stock, pursuant to which those executives borrowed an aggregate of $1,813 at a rate of 2.89% per annum, compounded annually. As these loans were non-recourse loans under the accounting guidance they were not reflected in the consolidated balance sheet or table above. As of December 31, 2021, all these balances plus accrued interest have been settled. The balances including accrued interest were settled as part of the executive’s regular pay and bonus, severance or via shares repurchased by the Company. During the year ended December 31, 2021, the Company received 471,757 shares from key management personnel in full settlement of principal amount $2,007 outstanding promissory notes and related interest. |
Refer to “Other Debt” in Note 11 - Debt for details of other loans from related parties.
| F-87 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| 22. COMMITMENTS AND CONTINGENCIES |
Contingencies
Although the possession, cultivation and distribution of cannabis for medical use is permitted in certain states, cannabis is classified as a Schedule-I controlled substance under the U.S. Controlled Substances Act and its use remains a violation of federal law. The Company’s operations are subject to a variety of local and state regulations. Failure to comply with one or more of those regulations could result in fines, restrictions on its operations, or losses of permits that could result in the Company ceasing operations. While management believes that the Company is in material compliance with applicable local and state regulations as of December 31, 2021, marijuana regulations continue to evolve and are subject to differing interpretations. As a result, the Company could be subject to regulatory fines, penalties or restrictions at any time. Since federal law criminalizing the use of cannabis preempts state laws that legalize its use, strict enforcement of federal law regarding cannabis would likely result in the Company’s inability to proceed with the Company’s business plans. In addition, the Company’s assets, including real property, cash and cash equivalents, equipment, inventory and other goods, could be subject to asset forfeiture because cannabis is still federally illegal.
Refer to Note 16 - Income Taxes for certain tax related contingencies and to Note 7 - Acquisitions for an acquisition-related contingent consideration liability.
Claims and Litigation
Any proceeding that may be brought against the Company could have a material adverse effect on the Company’s business plans, financial condition and results of operations. From time to time, the Company may be involved in litigation relating to claims arising out of operations in the normal course of business. Except as disclosed below, as of December 31, 2021, there were no pending or threatened lawsuits that could reasonably be expected to have a material effect on the Company’s financial results. There are also no proceedings in which any of the Company’s directors, officers or affiliates is an adverse party or has a material interest adverse to the Company’s interest.
SFN Litigation
On March 19, 2018, the Company acquired a majority stake in TGS National Holdings LLC which controlled TGS National Franchise, LLC (“TGS NF”), a franchisor. During 2018, San Felasco Nurseries, Inc. (“SFN”) terminated franchise agreements between it and TGS NF. SFN then sold its business to a third-party. TGS NF contended the termination of the franchise agreements and sale to the third party were wrongful and in late 2018 initiated arbitration seeking to recover its monetary damages. In May 2020, Jushi FL SPV, LLC was substituted for TGS NF as the claimant in the arbitration. The final hearing in the arbitration was held in May 2021. In July 2021, three arbiters of the American Arbitration Association found that SFN improperly terminated its franchise agreements with Jushi FL SPV, LLC without cause and in bad faith. The parties confidentially agreed to payment terms. Pursuant to a separate contract, a third party was entitled to receive 25% of the recovery of the arbitration, net of all fees and costs related to SFN. During the year ended December 31, 2021, the Company received the settlement payment, and after deducting expenses, fees and the 25% due to a third party, recognized a net gain on legal settlement of $10,248 which is included in gains on legal settlements within other income (expense), net in the consolidated statements of operations and comprehensive income (loss).
| F-88 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Refer to Note 17 - Non-Controlling Interests for discussion of the bankruptcy of Jushi Europe.
Commitments
In addition to the contractual obligations outlined in Note 11 - Debt and Note 12 - Lease Obligations, the Company has the following commitments as of December 31, 2021:
(i) Acquisitions
NuLeaf
In November 2021, the Company entered into a definitive agreement to acquire NuLeaf, Inc. together with its subsidiaries and related companies (collectively, “NuLeaf”), a Nevada-based vertically integrated operator. NuLeaf currently operates two high-performing adult-use and medical retail dispensaries in Las Vegas, NV and Lake Tahoe, NV, in addition to a 27,000 sq. ft. cultivation facility in Sparks, NV, as well as a 13,000 sq. ft. processing facility in Reno, NV. Additionally, NuLeaf owns a third licensed retail dispensary located directly on Las Vegas Boulevard. Refer to Note 24 - Subsequent Events
Apothecarium
In September 2021, the Company entered into a definitive agreement to acquire 100% of the equity interest of an entity operating an adult-use and medical retail dispensary under the name The Apothecarium in Las Vegas, NV (“Apothecarium”). The Apothecarium acquisition, together with the prior acquisition of FBS Nevada, a holder of medical and adult-use cannabis cultivation, processing, and distribution licenses, will enable the Company to become vertically integrated in Nevada, as well as provide significant branding exposure for Jushi’s high-quality product lines. Refer to Note 24 - Subsequent Events.
(ii) Property and Construction Commitments
In connection with various license applications, the Company may enter into conditional leases or other property commitments which will be executed if the Company is successful in obtaining the applicable license and/or resolving other contingencies related to the license or application.
In addition, the Company expects to incur capital expenditures for leasehold improvements and construction of buildouts of certain locations, including for properties for which the lease is conditional on obtaining the applicable related license or for which other contingencies exist. If the Company were to be unsuccessful in obtaining a particular license or certain other conditions are not met, the previously capitalized improvements and buildouts relating to that license may need to be expensed in future periods in the statements of operations and comprehensive income (loss).
| F-89 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
Concentration Risk
The Company has a geographical concentration of revenues from external customers as follows:
| Year Ended December 31, | |||||||||
| 2021 | 2020 | ||||||||
| Pennsylvania | 49 | % | 55 | % | |||||
| Illinois | 37 | % | 41 | % | |||||
| 23. FINANCIAL INSTRUMENTS |
The following table sets forth the Company’s financial assets and liabilities, subject to fair value measurements on a recurring basis by level within the fair value hierarchy as of December 31, 2021:
| Level 1 | Level 2 | Level 3 | Total | |||||||||||||
| Financial assets: | ||||||||||||||||
| Non-current equity investment | $ | — | $ | — | $ | 1,500 | $ | 1,500 | ||||||||
| $ | — | $ | — | $ | 1,500 | $ | 1,500 | |||||||||
| Financial liabilities: | ||||||||||||||||
| Derivative liabilities | $ | — | $ | — | $ | 92,435 | $ | 92,435 | ||||||||
| Contingent consideration liabilities | $ | — | $ | — | $ | 8,223 | $ | 8,223 | ||||||||
| $ | — | $ | — | $ | 100,658 | $ | 100,658 | |||||||||
The following table sets forth the Company’s financial assets and liabilities, subject to fair value measurements on a recurring basis by level within the fair value hierarchy as of December 31, 2020:
| Level 1 | Level 2 | Level 3 | Total | |||||||||||||
| Financial assets: | ||||||||||||||||
| Investments in securities-shares | $ | 6,026 | $ | — | $ | — | $ | 6,026 | ||||||||
| Investments in securities-warrants | — | 1,908 | — | $ | 1,908 | |||||||||||
| Non-current equity investment | — | — | 1,500 | $ | 1,500 | |||||||||||
| $ | 6,026 | 1,908 | $ | 1,500 | $ | 9,434 | ||||||||||
| Financial liabilities: | ||||||||||||||||
| Derivative liabilities | $ | — | $ | — | $ | 205,361 | $ | 205,361 | ||||||||
| $ | — | $ | — | $ | 205,361 | $ | 205,361 | |||||||||
The carrying amounts of certain financial instruments, including cash and cash equivalents, accounts receivable, accounts payable and certain accrued expenses, and certain other assets and liabilities held at amortized cost, approximate their fair values due to the short-term nature of these instruments. The non-current equity investment approximates its fair value at December 31, 2021 and 2020. The carrying amounts of the promissory notes approximate their fair values as the effective interest rates are consistent with market rates. The fair value of the 10% Senior Notes as of December 31, 2021 and 2020 approximated the principal amount.
There were no transfers between hierarchy levels during the years ended December 31, 2021 and 2020.
| F-90 |
JUSHI HOLDINGS INC. Notes to the Consolidated Financial Statements December 31, 2021 and 2020 (Amounts Expressed in Thousands of United States Dollars, Unless Otherwise Stated) |
 |
| 24. SUBSEQUENT EVENTS |
The Company has evaluated subsequent events through May 16, 2022, which is the date these consolidated financial statements were approved by the board of directors of the Company.
Private Placements
In January 2022, the Company closed non-brokered private placement offerings for an aggregate 3,717,392 SVS at a price of $3.68 per share to an existing investor group for aggregate gross proceeds of $13,680.
Acquisitions
In March and April 2022, the Company closed on the Apothecarium and the NuLeaf acquisitions, respectively. The Company drew down $25,000 from the Acquisition Facility to fund the cash portion of these transactions. For the NuLeaf acquisition, the Company paid upfront consideration comprised of $15,750 in cash, subject to working capital adjustments, issued 4,662,384 SVS (with a grant date fair value of $2.87 each), issued $15,750 in an unsecured five-year note, and may pay up to $10,000 in potential earn-out consideration based on receiving certain regulatory approval. For the Apothecarium acquisition, the Company issued 527,704 SVS (with a grant date fair value of $3.79 each), issued a $10,000 five-year note, and paid $6,000 in cash, subject to adjustments, and may pay up to $2,000 in potential earn out consideration based on the achievement of certain financial metrics. In addition, the Company issued 101,082 SVS for broker fees in connection with the acquisition.
Other than the information already disclosed in Note 22 - Commitments and Contingencies, the Company is still in the process of gathering and preparing additional disclosure information for these acquisitions, including preliminary purchase price allocations and pro forma financial information. Due to the timing of the acquisitions, the Company is not able to provide additional information at this time.
| 25. CORRECTION OF ERROR IN PREVIOUSLY ISSUED FINANCIAL STATEMENTS |
In the process of preparing the Company’s first quarter 2022 financial statements, the Company identified a misstatement related to the classification of interest payments on its finance leases in the consolidated statement of cash flows for the year end December 31, 2021. Consequently, the Company restated the classification of interest payments which resulted in an increase in net cash flows used in operating activities from $7,928 (as previously reported) to $14,304 (as restated) and a corresponding increase in net cash flows provided by financing activities from $131,287 (as previously reported) to $137,663 (as restated) for the year ended December 31, 2021. In addition, the Company restated the supplemental cash flow information for cash paid for interest (excluding capitalized interest) from $7,422 (as previously reported) to $13,798 (as restated). These corrections did not have an impact on the net change in cash and cash equivalents and restricted cash during the period.
| F-91 |
Subordinate Voting Shares

PROSPECTUS
August 12, 2022